Experimental study on dry reforming of methane by a plasma catalytic hybrid system
-
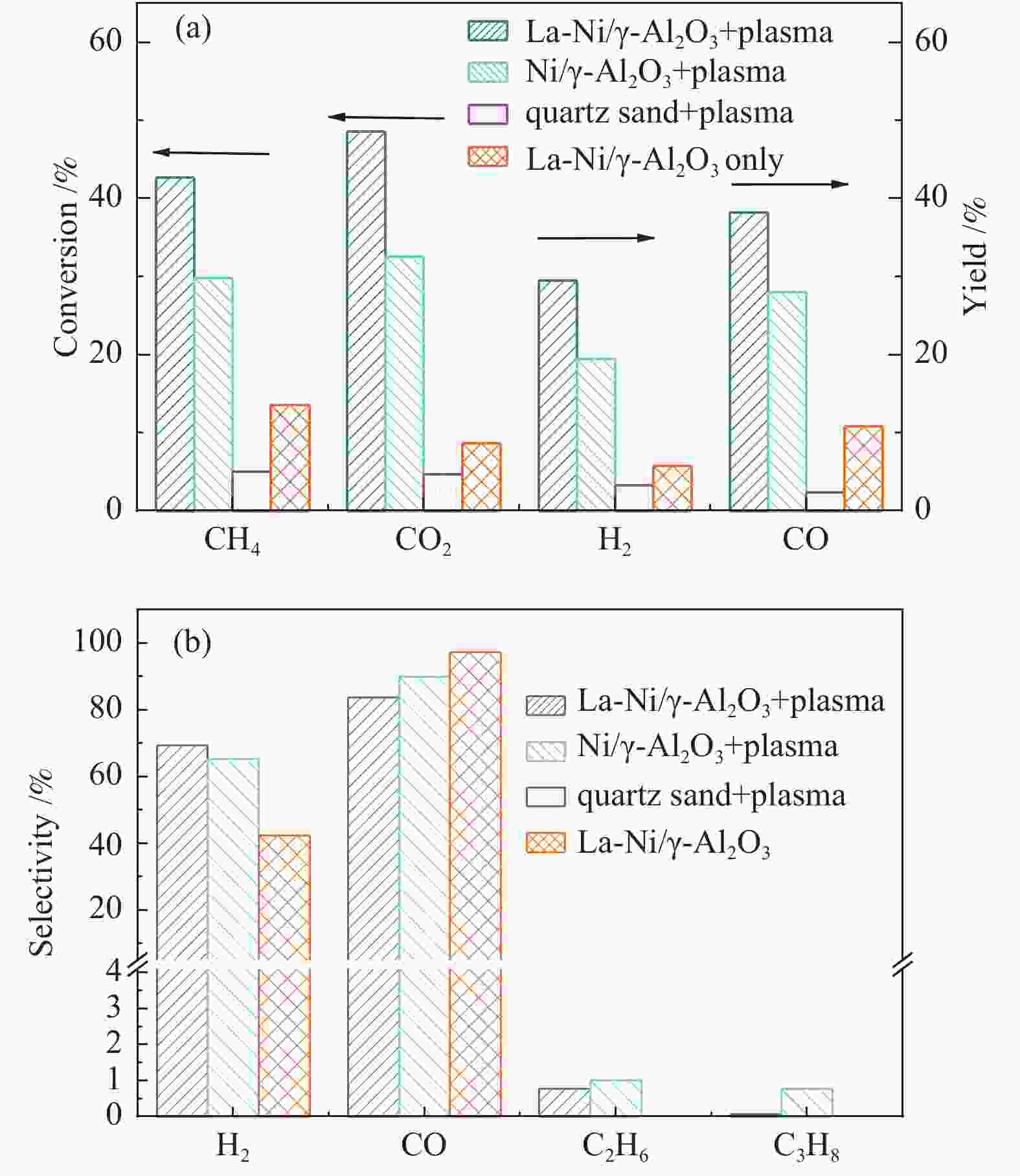
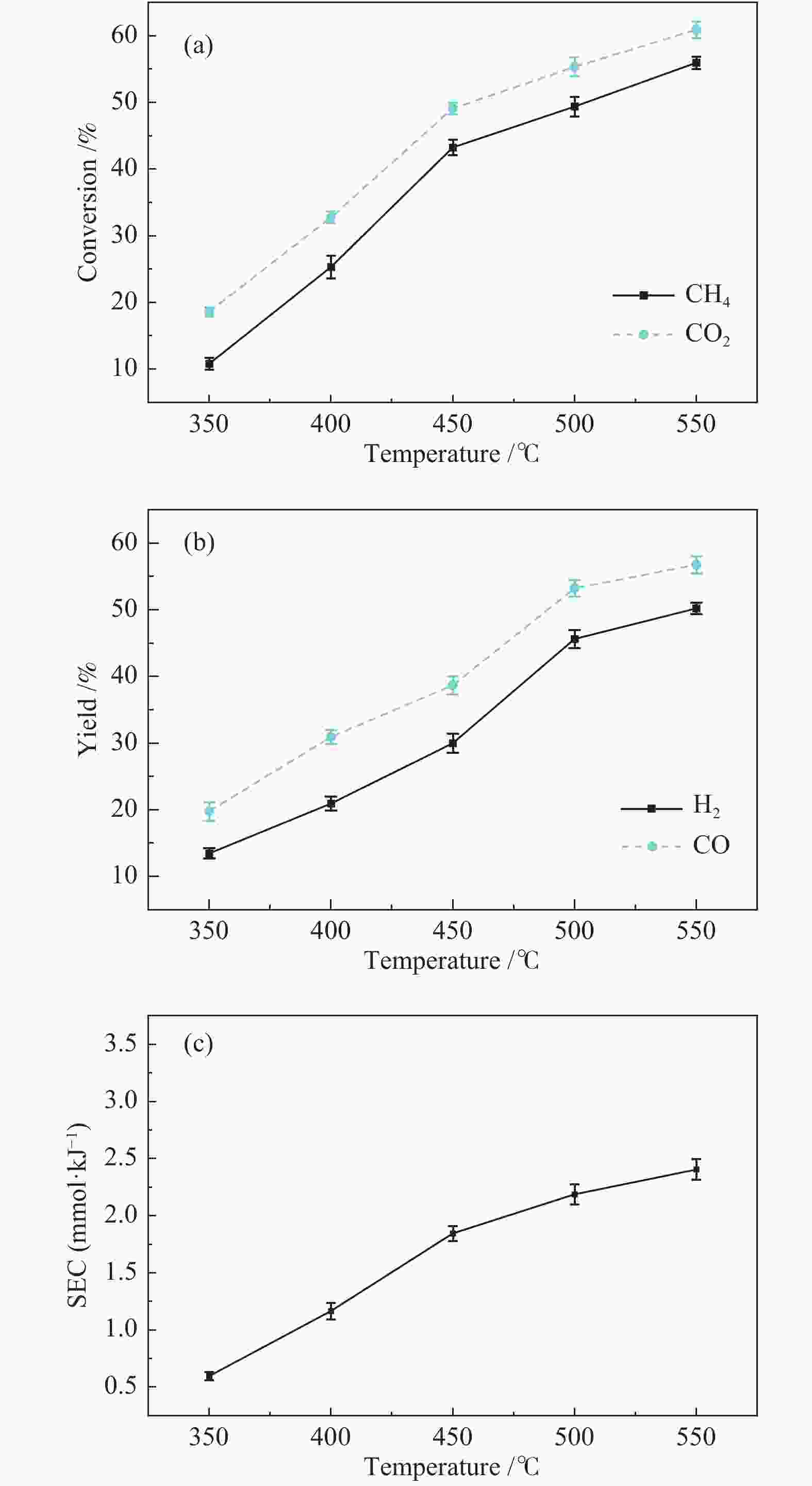
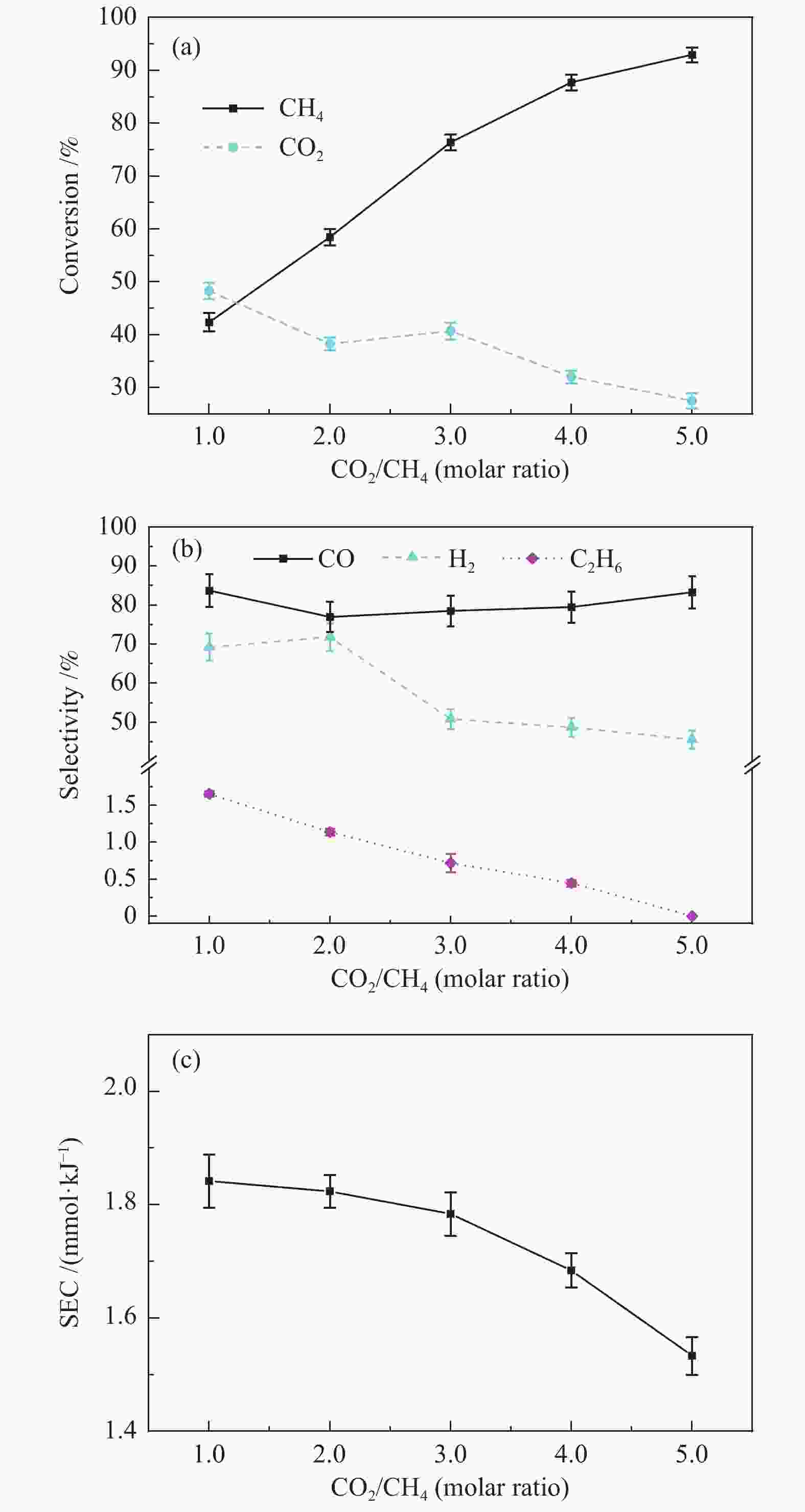
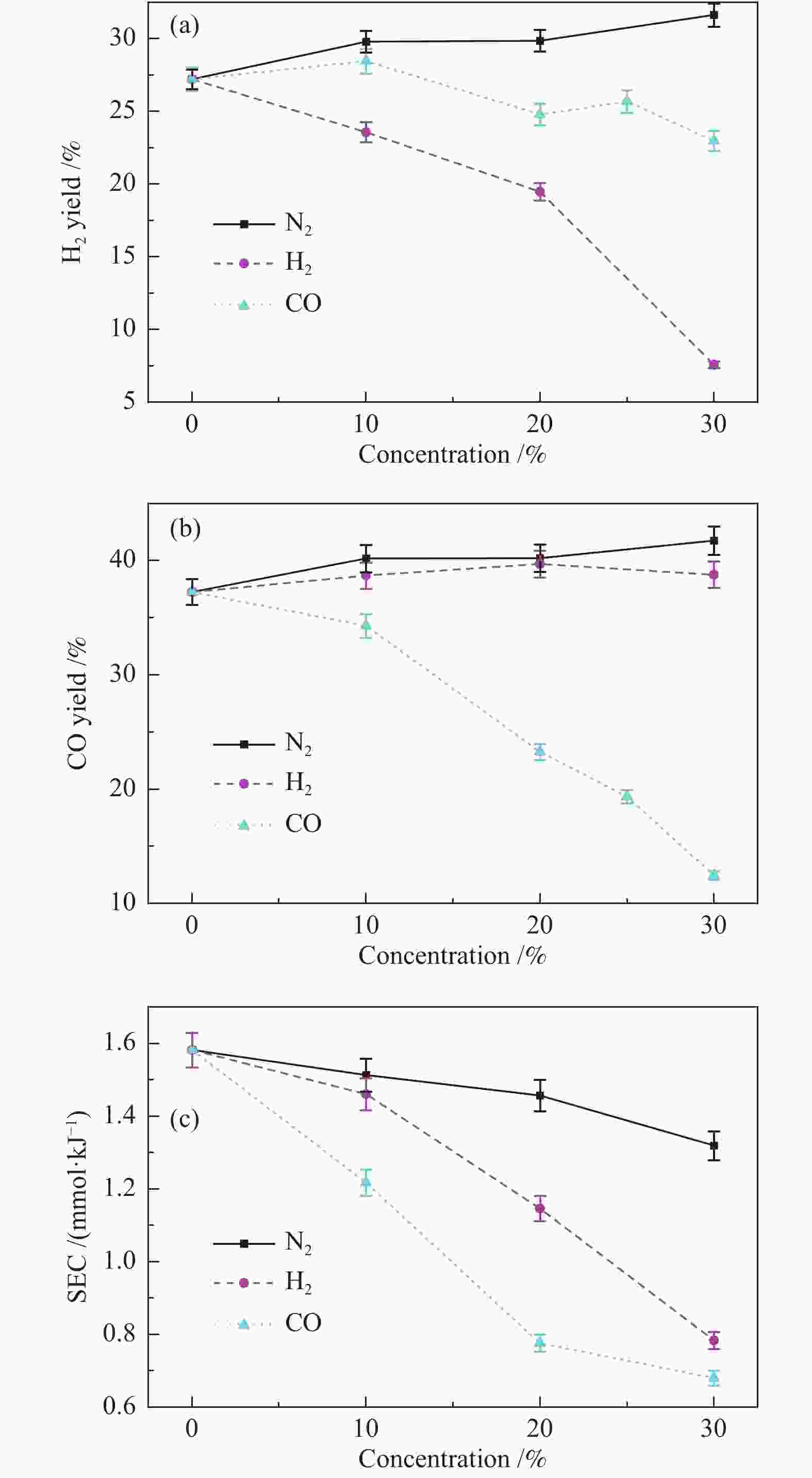
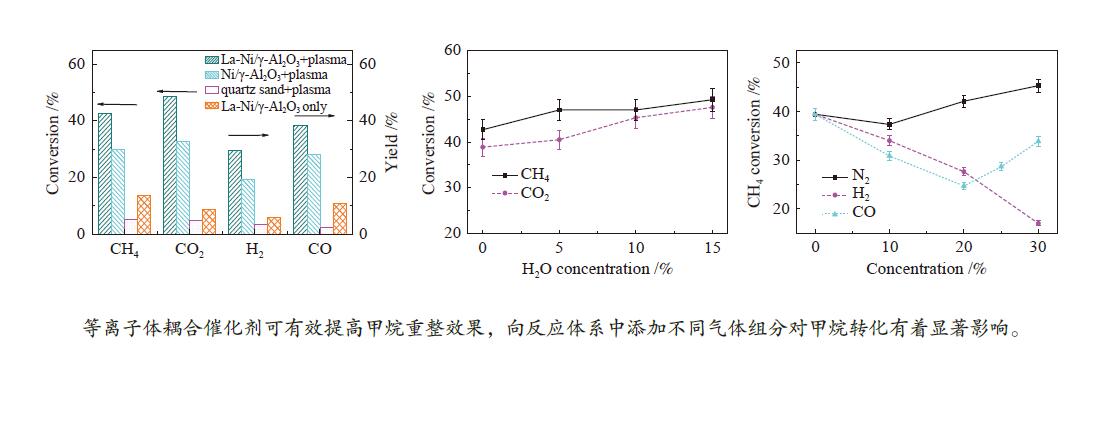
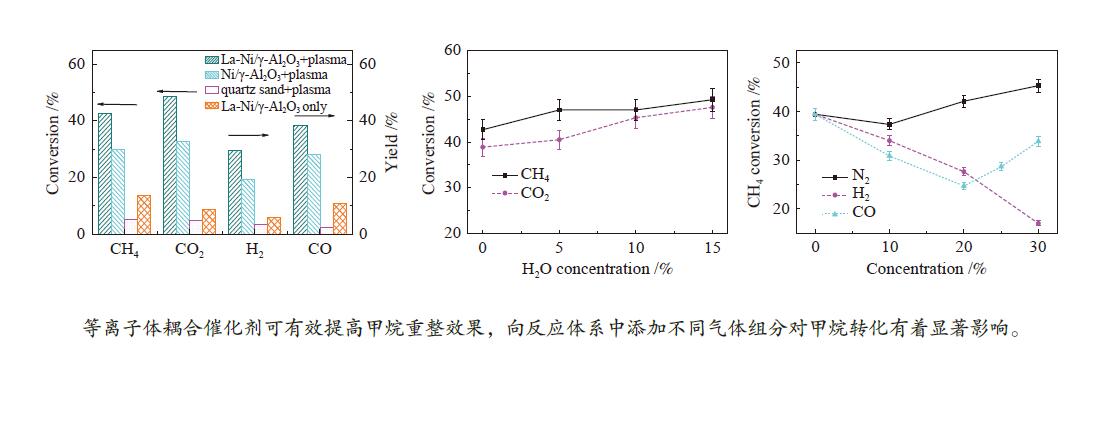
摘要: 本文利用等离子体耦合催化剂的方式进行CH4干重整(Dry Reforming of Methane,DRM),重点考察了反应温度、CO2/CH4物质的量比、合成气主要气体组分浓度(N2、H2、CO、H2O)对CH4转化率及等离子体催化能量效率的影响。结果表明,以La-Ni/γ-Al2O3为催化剂,当反应温度450 ℃,CO2/CH4物质的量比为1.0时,CH4转化率为41.57%;提高CO2/CH4物质的量比可提高CH4转化率,当CO2/CH4物质的量比为5.0时,等离子体催化CH4干重整过程的CH4转化率可达92.82%。温度和CO2/CH4物质的量比对CH4转化率影响显著,气体组分的变化改变了体系中的激发态粒子,不仅直接影响到CH4转化率,还影响着催化剂表面积炭。向反应体系中添加N2、H2O可提高CH4转化率,并抑制积炭;而添加H2、CO后CH4转化率显著降低。研究结果可望为生物质气化合成化工品的工艺开发提供基础数据和参考依据。Abstract: In this paper, the plasma coupled catalyst was used for the dry reforming of methane (DRM). The impacts of the reaction temperature, the molar ratio of CO2/CH4, and the concentration of the main gas components (N2, H2, CO, H2O) on the conversion rate of CH4 and the energy efficiency of plasma catalysis were observed. The results shows that the conversion rate of CH4 is 41.57%, with La-Ni/γ-Al2O3 as the catalyst, the reaction temperature being 450℃ and the molar ratio of CO2/CH4 being 1.0, and without other gases. The conversion rate of CH4 increases with the increase of the molar ratio of CO2/CH4. The conversion rate of CH4 is 92.82% respectively during DRM by plasma catalysis, with the molar ratio of CO2/CH4 being 5.0. The reaction temperature and the molar ratio of CO2/CH4 have a significant effect on CH4 conversion. The excited particles in the system are influenced by the changes of gas composition, which affect not only CH4 conversion directly, but also the carbon deposition on the surface of catalyst. The addition of N2 and H2O in the reaction system not only improves conversion of CH4 but also inhibits carbon deposition. The addition of H2 and CO reduces the conversion of CH4 significantly. The results are expected to provide basic data and reference for the development of the synthetic process of chemicals through biomass gasification.
-
Key words:
- syngas components /
- plasma catalysis /
- dry reforming of methane
-
表 1 生物质富氧-水蒸气气化的产气组成
Table 1 Gas composition of biomass oxygen-steam gasification
表 2 介质阻挡放电甲烷干重整
Table 2 Research progress of dry reforming of methane by dielectric barrier discharge
Experimental conditions Conversion rates/% Yield/% Energy efficiency
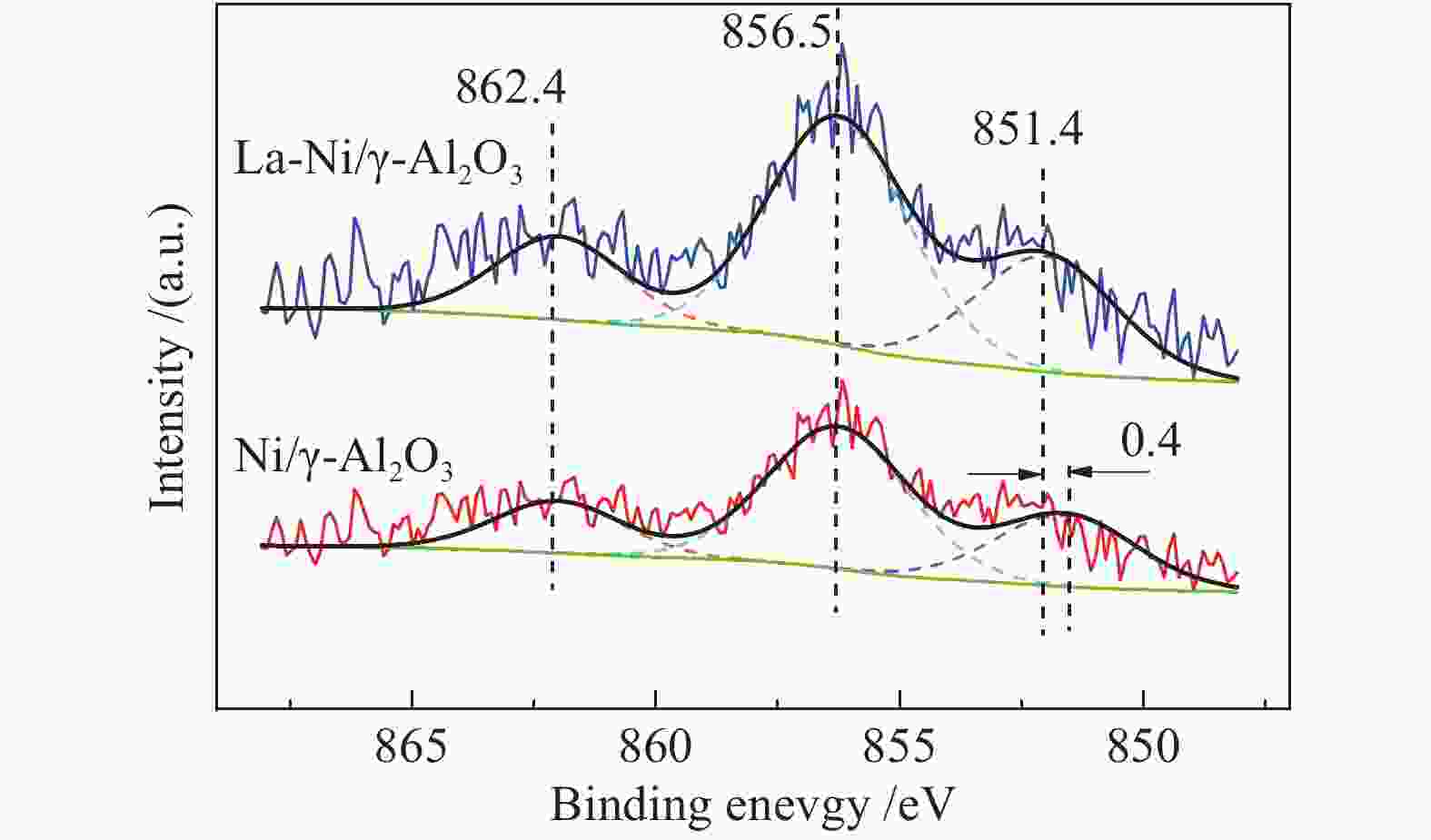
SEC/(mmol·kJ−1)Literature Power/W CO2/CH4 (mol ratio) catalyst CH4 CO2 H2 CO 45 1:1 Pt/Al2O3 32.5 22.1 − − 0.205 [15] 140 1:1 Al2O3 79 71 21 32 0.084 [16] 100 3:1 Ni/La2O3-MgAl2O4 78.8 68.1 30.4 37 0.125 [17] 200 1:1 Ni/ZrO2 52.8 60.8 52.1 63.5 − [18] 100 1:1 Ni/La2O3-MgAl2O4 86.0 84.5 42 40 0.26 [19] 表 3 Ni/γ-Al2O3和La-Ni/γ-Al2O3催化剂XPS光谱Ni 2p3/2区峰面积
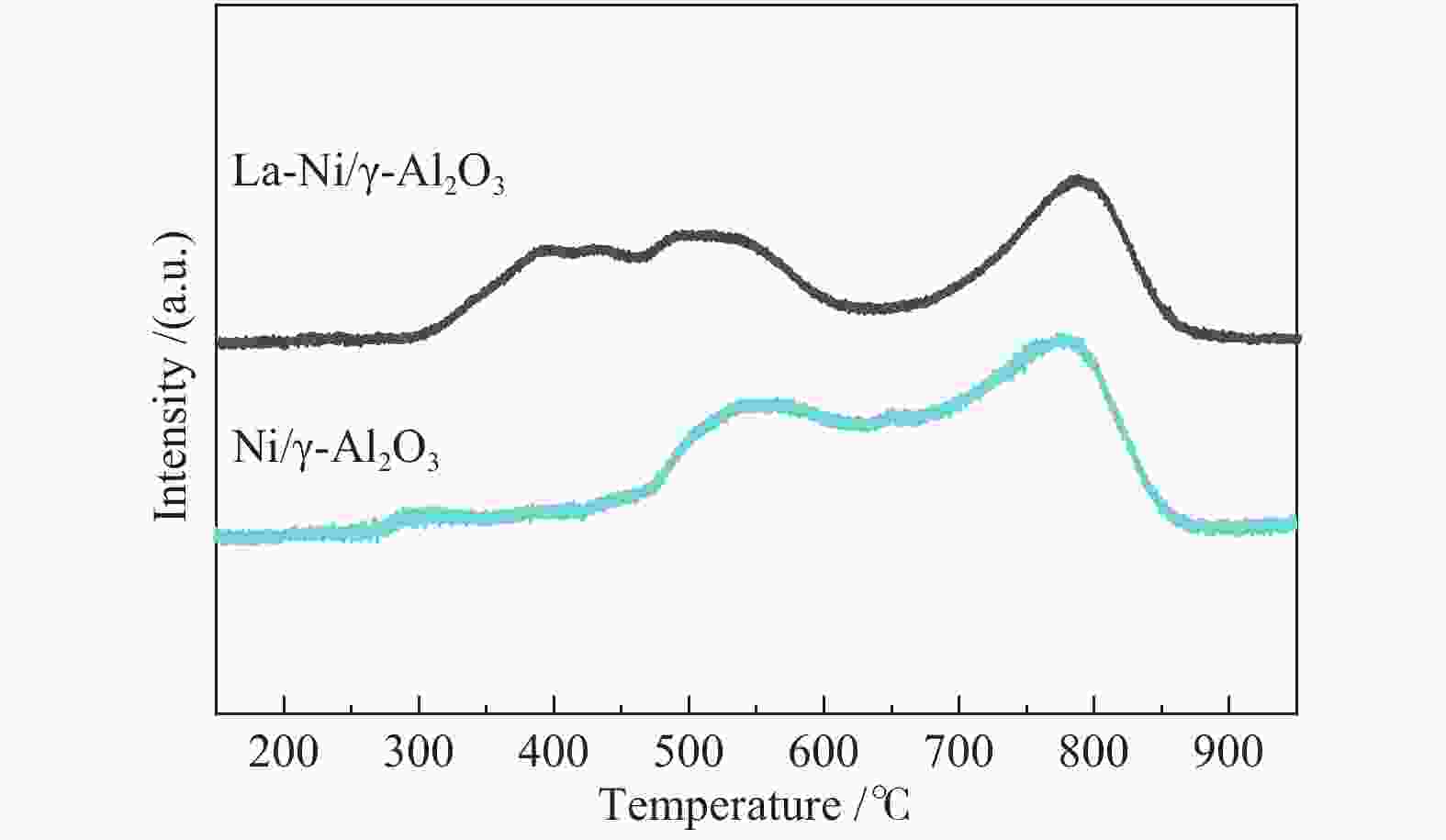
Table 3 Peak areas of Ni 2p3/2 region in XPS spectra of Ni/γ- Al2O3 and La Ni/γ- Al2O3 catalysts
Catalyst Peak area /% 851.4 eV 856.5 eV 862.4 eV Ni/γ-Al2O3 10.7 65.7 23.6 La-Ni/γ-Al2O3 27.5 53.8 28.7 表 4 不同催化剂的比表面积和孔容积
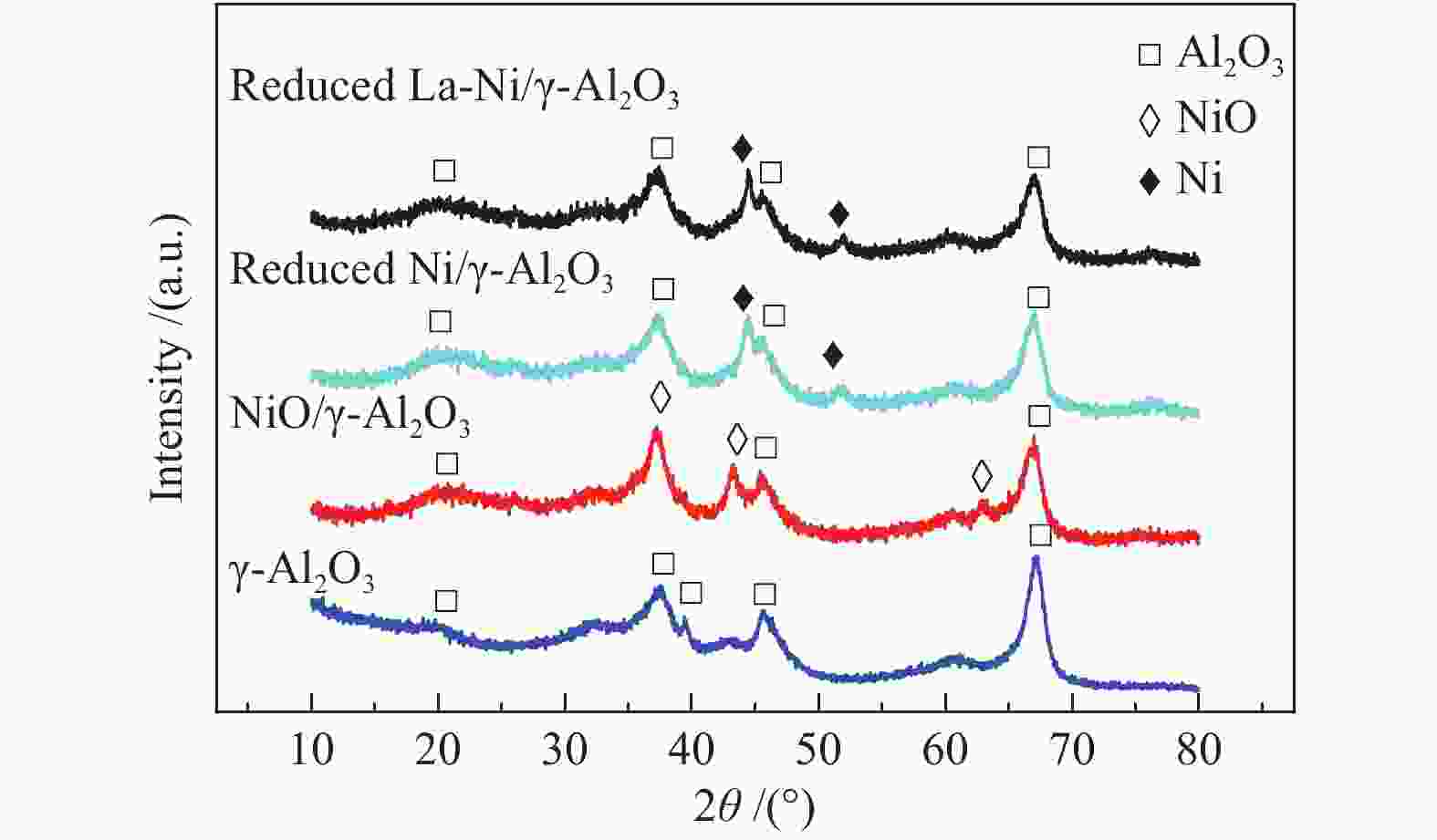
Table 4 Specific surface area and pore volume of different catalysts
Packing
materialsSpecific surface
area/(cm2·g−1)Pore volume/
(cm3·g−1)γ-Al2O3 110.31 0.281 Ni/γ-Al2O3 112.76 0.259 La-Ni/γ-Al2O3 97.99 0.243 表 5 不同气氛下反应20 min后催化剂的积炭量
Table 5 Carbon deposition on catalyst after 20 min reaction under different atmosphere
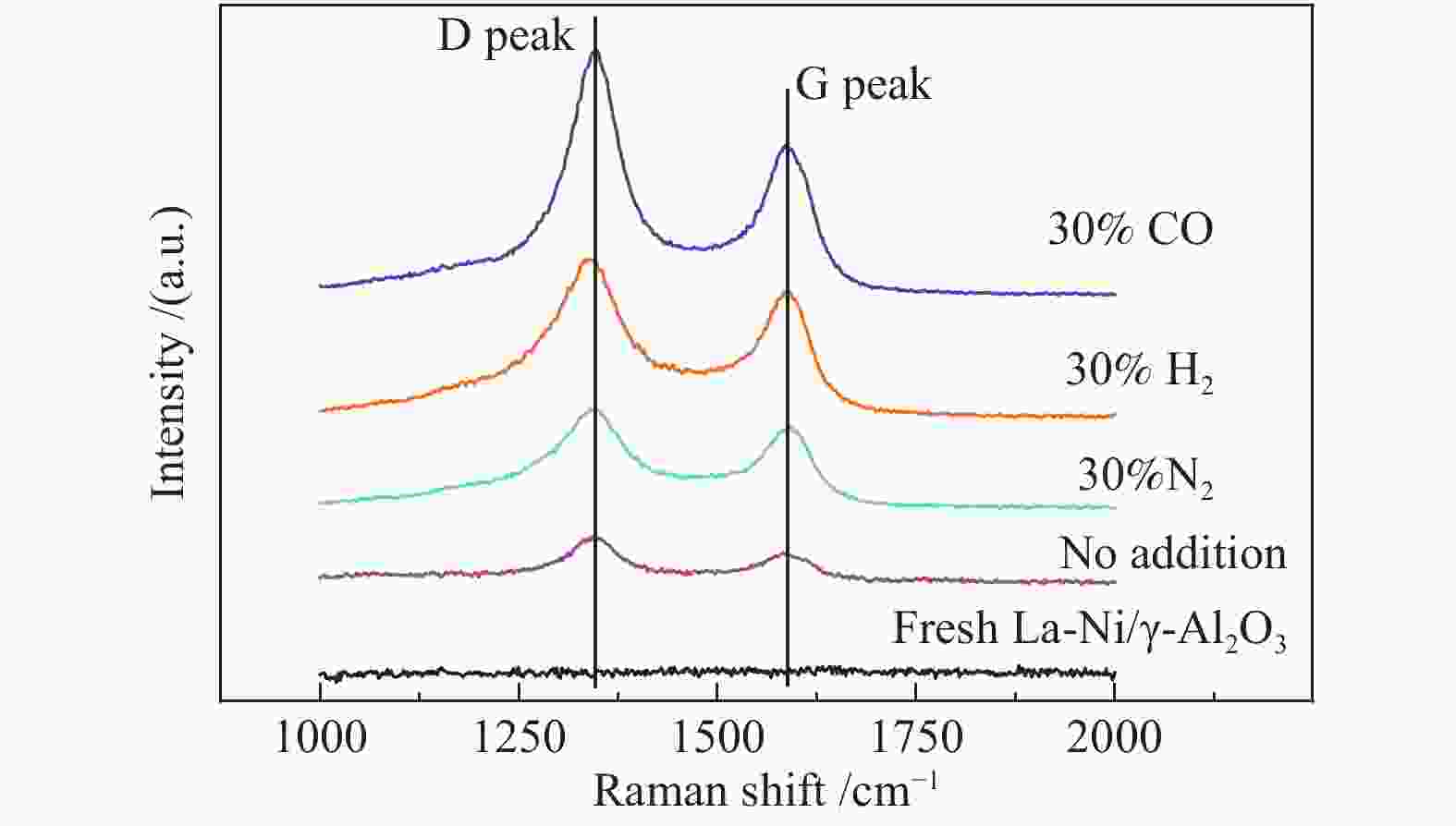
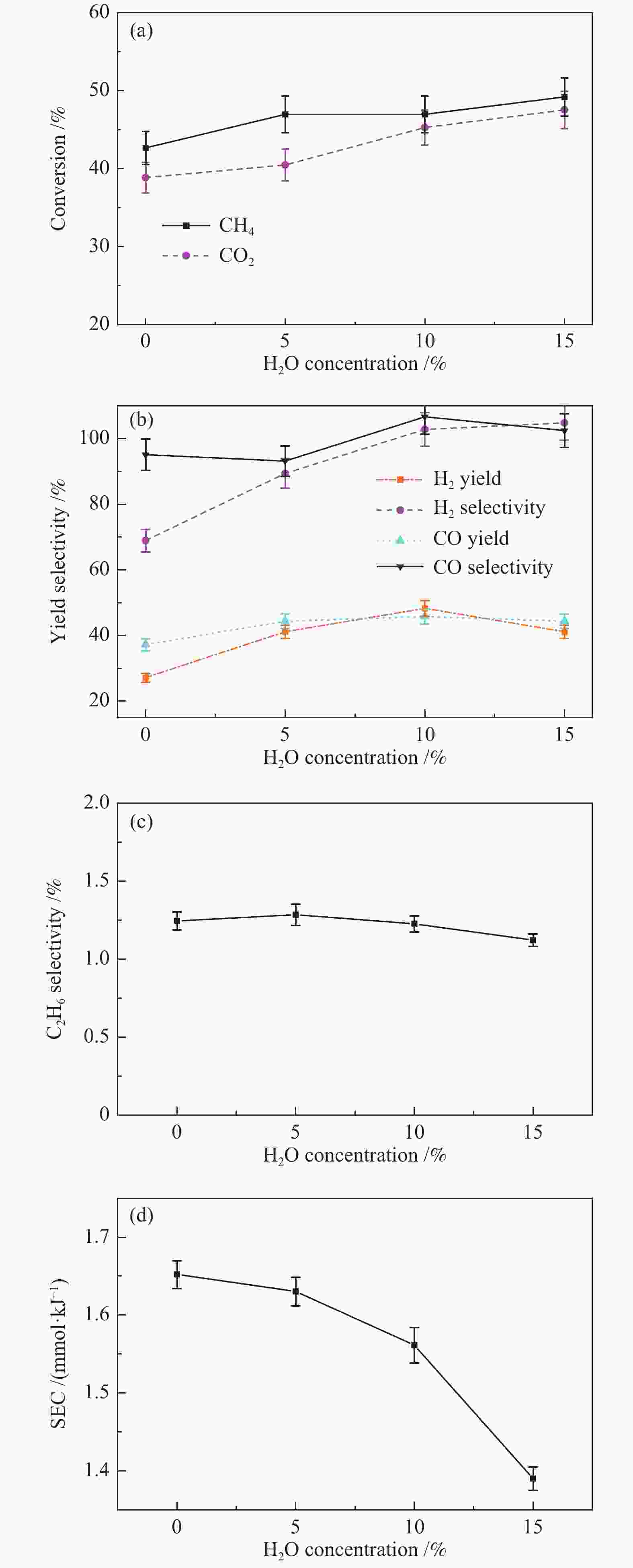
Process Carbon deposition w/% No addition 1.65 30% N2 addition 1.67 30% H2 addition 1.30 30% CO addition 7.69 20% CO addition 4.62 15% H2O addition 1.20 -
[1] DESHMUKH R, JACOBSON A, CHAMBERLIN C, KAMMEN D. Thermal gasification or direct combustion? Comparison of advanced cogeneration systems in the sugarcane industry[J]. Biomass Bioenergy,2013,55(55):163−174. [2] 徐彬. 低温等离子体协同催化脱除焦油典型组分的研究[D]. 北京: 中国科学院大学, 2019.XU Bin. Research on the removal of tar model compounds using the combination of non-thermal plasma and catalysts[D]. Beijing: University of Chinese Academy of Sciences, 2019. [3] 苏德仁, 周肇秋, 谢建军, 郎林, 阴秀丽, 吴创之. 生物质流化床富氧-水蒸气气化制备合成气研究[J]. 农业机械学报,2011,42(3):100−104.SU De-ren, ZHOU Zhao-qiu, XIE Jian-jun, LANG Lin, YIN Xiu-li, WU Chuang-zhi. Biomass oxygen enriched-steam gasification in an atmospheric fluidized bed for syngas production[J]. Trans Chin Soc Agric Mach,2011,42(3):100−104. [4] NIU Y, HAN F, CHEN Y, LYU Y, WANG L. Experimental study on steam gasification of pine particles for hydrogen-rich gas[J]. J Energy Inst,2017,90(5):715−724. doi: 10.1016/j.joei.2016.07.006 [5] 谢思凡, 胡建军, 张全国, 王伟, 党钾涛, 赵淑蘅. 秸秆热解催化重整制备合成气实验研究及模型预测[J]. 可再生能源,2020,38(9):1149−1156.XIE Si-fan, HU Jian-jun, ZHANG Quan-guo, WANG Wei, DANG Jia-tao, ZHAO Shu-heng. Experimental study and model prediction of syngas from straw catalytic reforming[J]. Renewable Energy Resour,2020,38(9):1149−1156. [6] 王嘉琦, 王秋颖, 朱桐慧, 朱小梅, 孙冰. 甲烷重整制氢的研究现状分析[J]. 现代化工,2020,40(7):15−20.WANG Jia-qi, WANG Qiu-ying, ZHU Tong-hui, ZHU Xiao-mei, SUN Bing. A review on research status of hydrogen production by methane reforming[J]. Mod Chem Ind,2020,40(7):15−20. [7] 莫文龙, 马凤云, 刘月娥, 刘景梅, 钟梅, 艾沙·努拉洪. 制备方法对Ni-Al2O3催化剂在CO2-CH4重整反应中催化性能的影响[J]. 燃料化学学报,2015,43(9):1083−1091.MO Wen-long, MA Feng-yun, LIU Yue-e, LIU Jing-mei, ZHONG Mei, AISHA·nulahong. Effect of preparation methods on the catalytic performance of Ni-Al2O3 for CO2-CH4 reforming[J]. J Fuel Chem Technol,2015,43(9):1083−1091. [8] AKRI M, EL KASMI A, BATIOT-DUPEYRAT C, QIAO B. Highly active and carbon-resistant nickel single-atom catalysts for methane dry reforming[J]. Catalysts,2020,10(6):630. doi: 10.3390/catal10060630 [9] 方琼, 吴东, 沈卫华, 朱志庆, 方云进. La改性NiMgAl催化剂对甲烷干气重整反应的影响[J]. 现代化工,2020,40(1):86−90.FANG Qiong, WU Dong, SHEN Wei-hua, ZHU Zhi-qing, FANG Yun-jin. Effect of La-promoting on performance of NiMgAl catalyst in dry[J]. Mod Chem Ind,2020,40(1):86−90. [10] 赵健, 周伟, 汪吉辉, 马建新. 甲烷干重整研究进展[J]. 天然气化工(C1化学与化工),2011,36(6):53−60+65.ZHAO Jian, ZHOU Wei, WANG Ji-hui, Ma Jian-xin. Research progress of methane dry reforming[J]. Nat Gas Ind,2011,36(6):53−60+65. [11] 张安杰, 丁天英, 刘云, 石川. 介质阻挡放电等离子体Cu-Ni/γ-Al2O3催化剂体系在甲烷二氧化碳重整反应中的协同作用[J]. 分子催化,2011,25(1):11−16.ZHANG An-jie, DING Tian-ying, LIU Yun, SHI Chuan. Reforming of methane over a combined system of dielectric-barrier discharge plasma and Cu-Ni/γ-Al2O3 catalyst[J]. J Mol Catal,2011,25(1):11−16. [12] ALAWI N M, SUNARSO J, PHAM G H, BARIFCANI A, NGUYEN M H, LIU S. Comparative study on the performance of microwave-assisted plasma DRM in nitrogen and argon atmospheres at a low microwave power[J]. J Ind Eng Chem,2020,85:118−129. doi: 10.1016/j.jiec.2020.01.032 [13] LIU L, ZHANG Z, DAS S, KAWI S. Reforming of tar from biomass gasification in a hybrid catalysis-plasma system: A review[J]. Appl Catal B: Environ,2019,250:250−272. doi: 10.1016/j.apcatb.2019.03.039 [14] ZHU F, ZHANG H, YAN X, YAN J, NI M, LI X, TU X. Plasma-catalytic reforming of CO2-rich biogas over Ni/γ-Al2O3 catalysts in a rotating gliding arc reactor[J]. Fuel,2017,199:430−437. doi: 10.1016/j.fuel.2017.02.082 [15] ANDERSEN J A, CHRISTENSEN J M, ØSTBERG M, BOGAERTS A, JENSEN A D. Plasma-catalytic dry reforming of methane: Screening of catalytic materials in a coaxial packed-bed DBD reactor[J]. Chem Eng J,2020,397:125519. doi: 10.1016/j.cej.2020.125519 [16] KHOJA A H, TAHIR M, AMIN N A S. Dry reforming of methane using different dielectric materials and DBD plasma reactor configurations[J]. Energy Convers Manage,2017,144:262−274. [17] KHOJA A H, TAHIR M, AMIN N A S. Process optimization of DBD plasma dry reforming of methane over Ni/La2O3-MgAl2O4 using multiple response surface methodology[J]. Int J Hydrog Energy,2019,44(23):11774−11787. doi: 10.1016/j.ijhydene.2019.03.059 [18] HU X, JIA X, ZHANG X, LIU Y, LIU C. Improvement in the activity of Ni/ZrO2 by cold plasma decomposition for dry reforming of methane[J]. Catal Commun,2019,128:105720. doi: 10.1016/j.catcom.2019.105720 [19] KHOJA A H, TAHIR M, AMIN N A S. Evaluating the performance of a Ni catalyst supported on La2O3-MgAl2O4 for dry reforming of methane in a packed bed dielectric barrier discharge plasma reactor[J]. Energy Fuels,2019,(33):11630−11647. [20] 何小强, 莫文龙, 覃松, 马凤云. 铝源对Ni/Al2O3催化剂结构及其CO2-CH4重整性能的影响[J]. 燃料化学学报,2020,48(2):221−230.HE Xiao-qiang, MO Wen-long, QIN Song, MA Feng-yun. Effect of aluminum source on the structure and performance of Ni /Al2O3 catalysts in CO2-CH4 reforming[J]. J Fuel Chem Technol,2020,48(2):221−230. [21] 刘少文, 涂文艳, 包传平. La2O3助剂对Ni/γ-Al2O3/堇青石结构化催化剂催化加氢合成间苯二胺性能的影响[J]. 化工进展,2012,31(1):122−125+162.LIU Shao-wen, TU Wen-yan, BAO Chuan-ping. Effect of La2O3 on the properties of Ni/γ-Al2O3/cordierite structured catalysts for hydrogenation of MDN to MPDA[J]. Chem Ind Eng Prog,2012,31(1):122−125+162. [22] RENCHUN Y, ZHIHUA Z, JUNSHENG W, XIAOGANG L, LUHAI W. Hydrotreating performance of La-modified Ni/Al2O3 catalysts prepared by hydrothermal impregnation method[J]. Kinet Catal,2015,56(2):222−225. doi: 10.1134/S0023158415020123 [23] MENG F, LI X, LI M, CUI X X, LI Z. Catalytic performance of CO methanation over La-promoted Ni/Al2O3 catalyst in a slurry-bed reactor[J]. Chem Eng J,2016,1548−55. [24] 孙路石, 孔继红, 向军, 胡松. La和Ce改性Ni/Al2O3催化剂上生物质气燃烧[J]. 华中科技大学学报(自然科学版),2010,38(9):54−57.SUN Lu-shi, SUN Ji-hong, XIANG Jun, HU Song. Catalytic combustion of gasified biomass over Ni/Al2O3 catalyst modified by La and Ce[J]. J Huaz Univ Sci Tenol (Nat Sci Ed),2010,38(9):54−57. [25] WANG H, HAN J, BO Z, QIN L, WANG Y, YU F. Non-thermal plasma enhanced dry reforming of CH4 with CO2 over activated carbon supported Ni catalysts[J]. Mol Catal,2019,475:110486. doi: 10.1016/j.mcat.2019.110486 [26] NOZAKI T, OKAZAKI K. Non-thermal plasma catalysis of methane: Principles, energy efficiency, and applications[J]. Catal Today,2013,211:29−38. doi: 10.1016/j.cattod.2013.04.002 [27] TU X, WHITEHEAD J C. Plasma-catalytic dry reforming of methane in an atmospheric dielectric barrier discharge: Understanding the synergistic effect at low temperature[J]. Appl Catal B: Environ,2012,125:439−448. doi: 10.1016/j.apcatb.2012.06.006 [28] ZHENG X G, YAN S Y, DONG L C, LI S B, CHEN H M, WEI S A. Experimental and kinetic investigation of the plasma catalytic dry reforming of methane over perovskite LaNiO3 nanoparticles[J]. Fuel Process Technol,2015,137:250−258. doi: 10.1016/j.fuproc.2015.02.003 [29] SERRANO-LOTINA A, DAZA L. Influence of the operating parameters over dry reforming of methane to syngas[J]. Int J Hydrogen Energ,2014,39(8):4089−4094. doi: 10.1016/j.ijhydene.2013.05.135 [30] SHAPOVAL V, MAROTTA E. Investigation on plasma-driven methane dry reforming in a self-triggered spark reactor[J]. Plasma Process Polym,2015,12(8):808−816. doi: 10.1002/ppap.201400177 [31] LIU S, MEI D, WANG L, TU X. Steam reforming of toluene as biomass tar model compound in a gliding arc discharge reactor[J]. Chem Eng J,2017,307:793−802. doi: 10.1016/j.cej.2016.08.005 [32] 刘潇钰. 滑动弧等离子体催化甲烷水蒸汽重整制氢研究[D]. 大连: 大连理工大学, 2019.LIU Xiao-yu. Steam methane reforming (SMR) for hydrogen production by gliding arc plasma catalysis[D]. Dalian: Dalian University of Technology, 2019. [33] 任盼盼. 新型镍基甲烷水蒸气-二氧化碳双重整催化剂的制备与性能研究[D]. 大连: 大连理工大学, 2016.REN Pan-pan. Study on the preparation and catalytic properties of novel Ni-based catalysts towards steam-CO2 dual reforming of methane[D]. Dalian: Dalian University of Technology, 2016. [34] WANG Y F, TSAI C H, CHANG W Y, KUO Y M. Methane steam reforming for producing hydrogen in an atmospheric-pressure microwave plasma reactor[J]. Int J Hydrogen Energy,2010,35(1):135−140. doi: 10.1016/j.ijhydene.2009.10.088 [35] WANG Q, SHI H, YAN B, JIN Y, CHENG Y. Steam enhanced carbon dioxide reforming of methane in DBD plasma reactor[J]. Int J Hydrogen Energy,2011,36(14):8301−8306. doi: 10.1016/j.ijhydene.2011.04.084 [36] 李智. 滑动弧等离子体催化CH4-CO2-H2O重整制合成气[D]. 大连: 大连理工大学, 2019.LI Zhi. Reforming of CH4-CO2-H2O for syngas in a gliding arc plasma catalytic reactor[D]. Dalian: Dalian University of Technology, 2019. -




 下载:
下载: