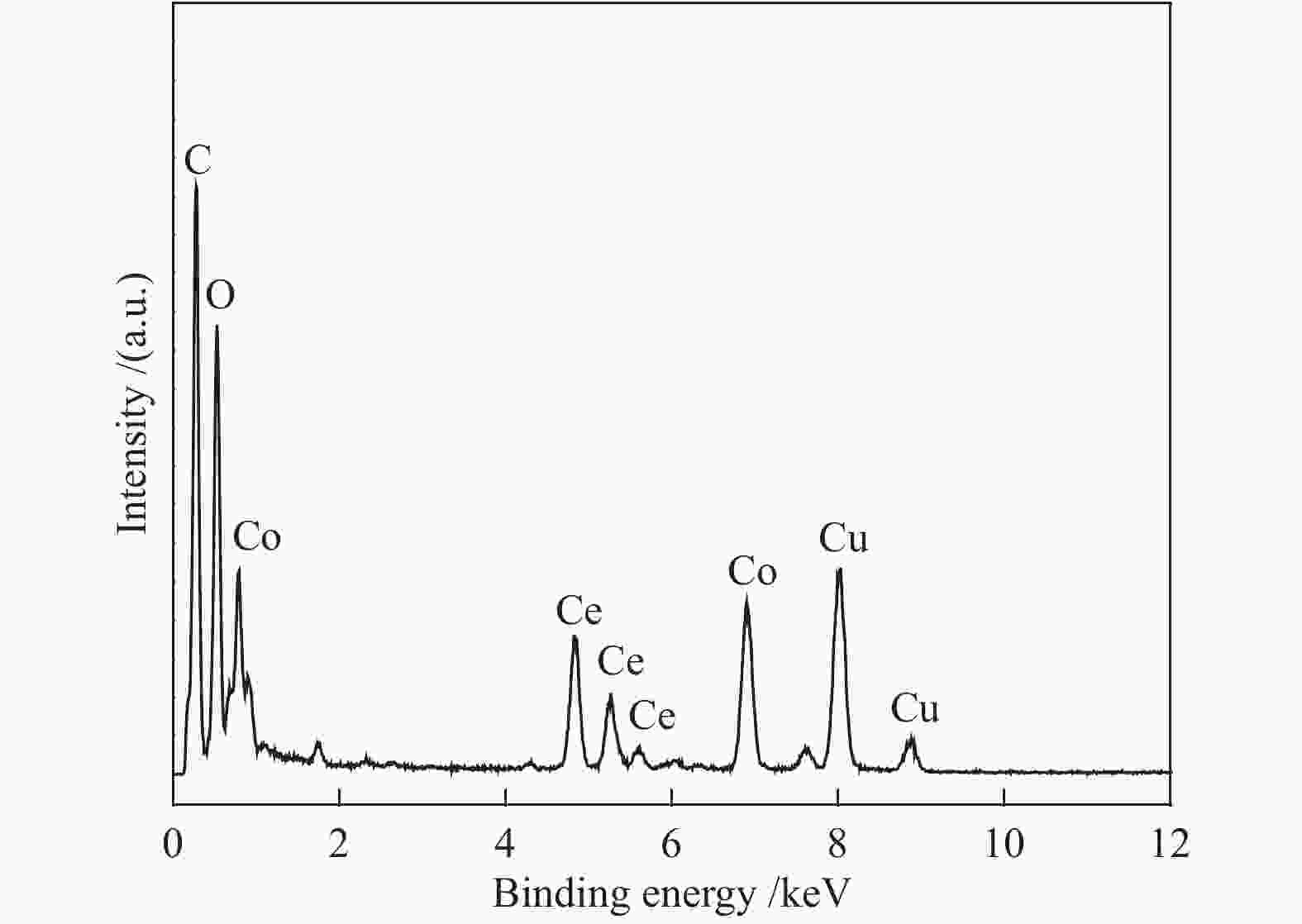
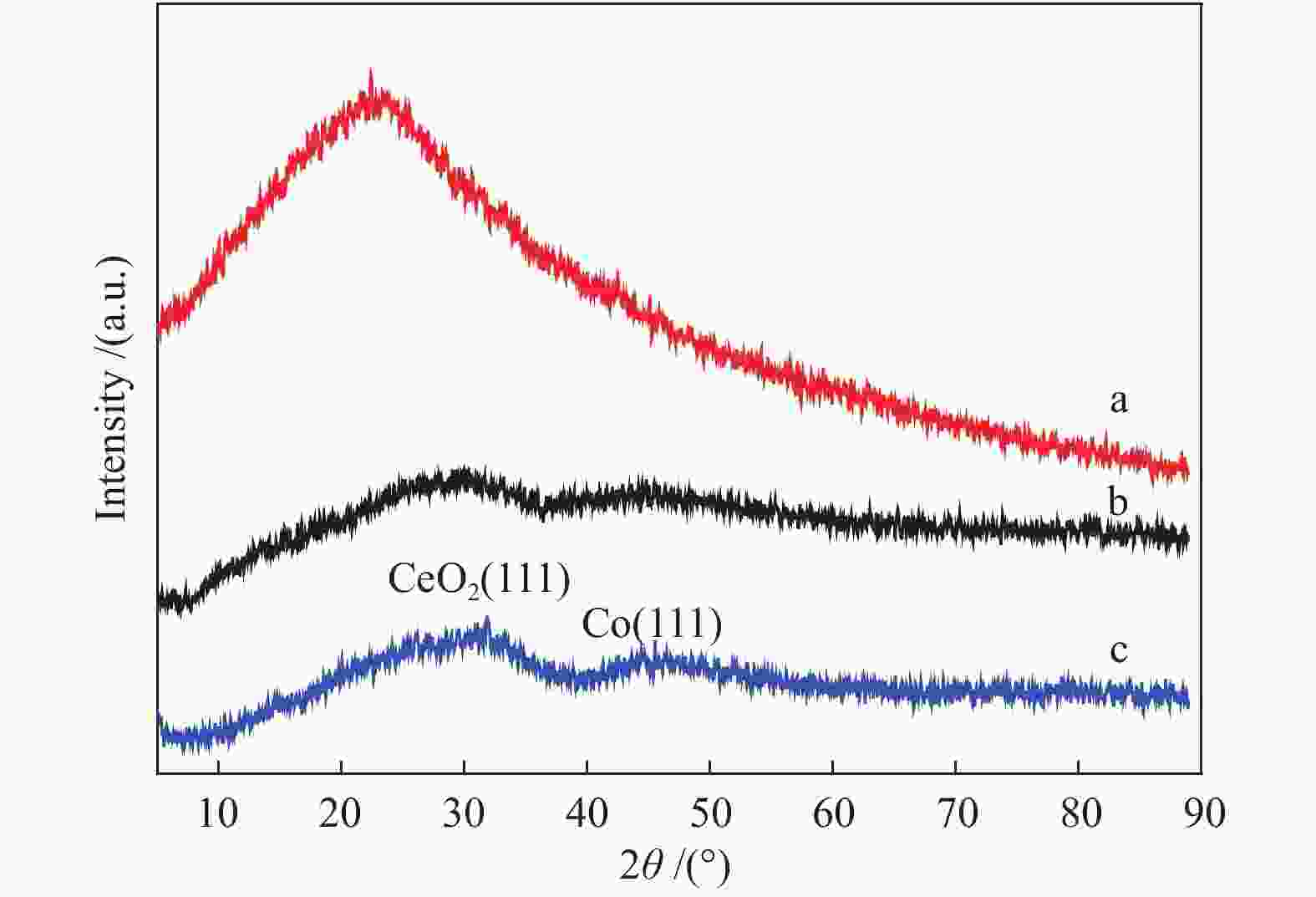
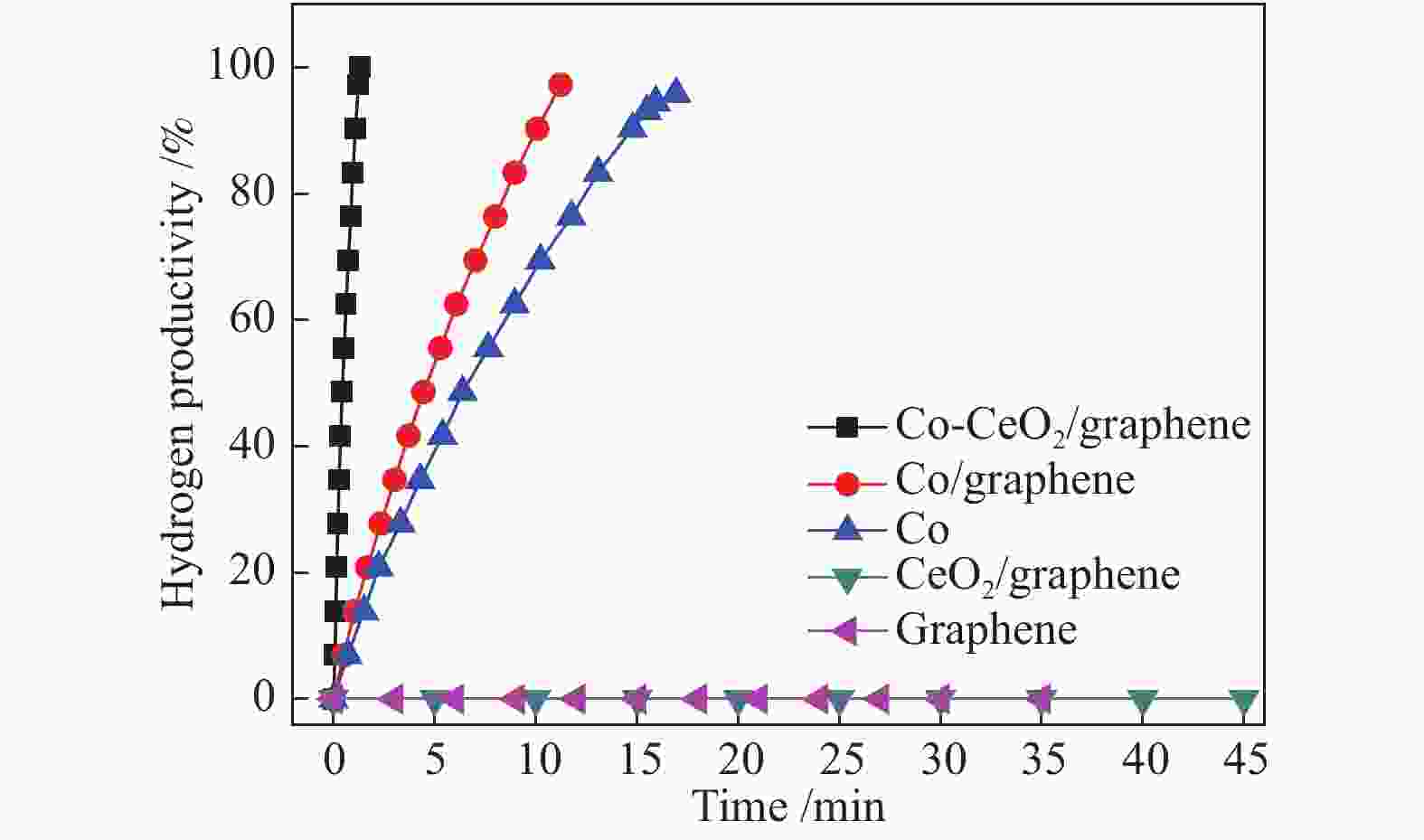
Preparation of graphene-supported Co-CeOx nanocomposites as a catalyst for the hydrolytic dehydrogenation of ammonia borane
-
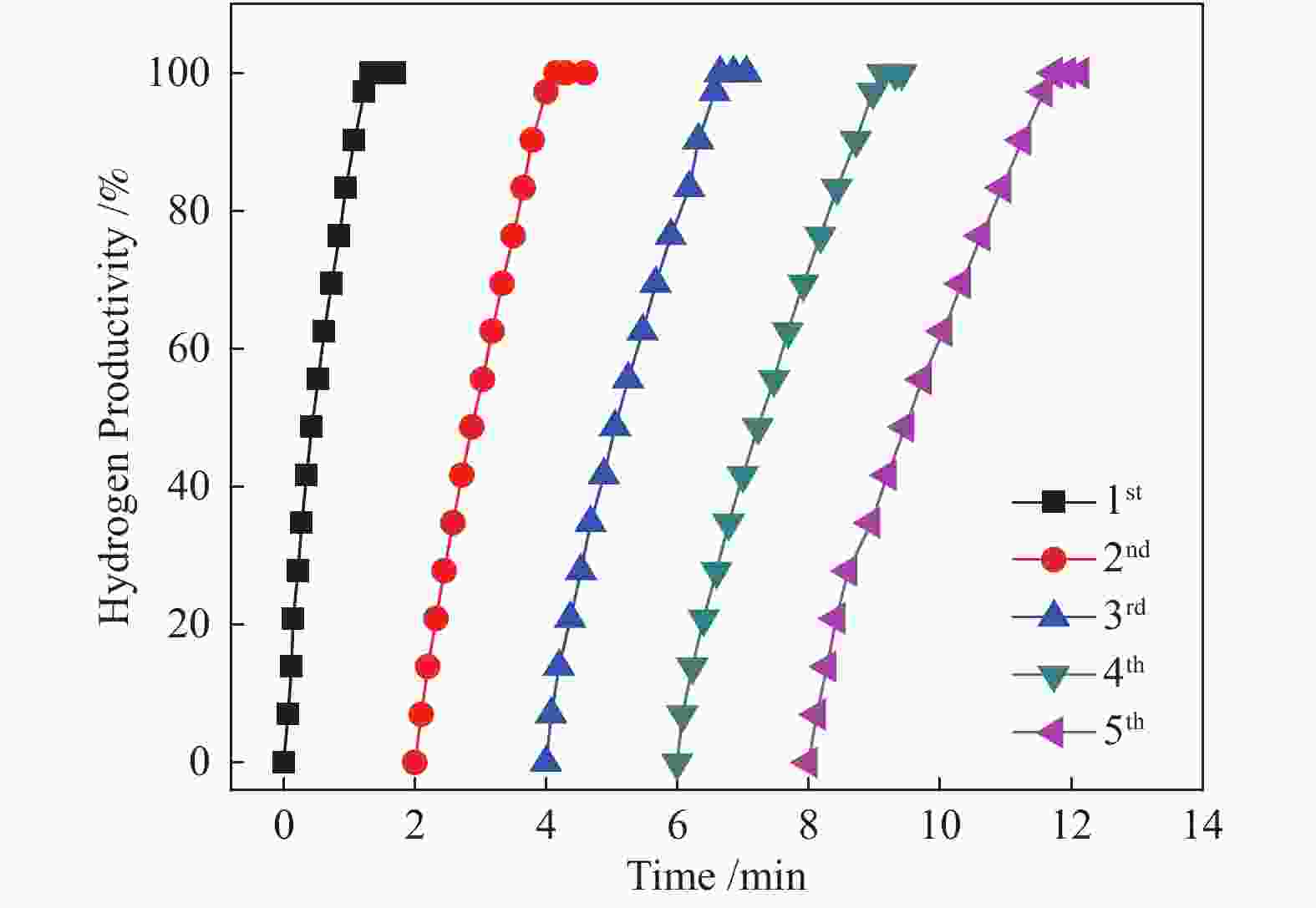
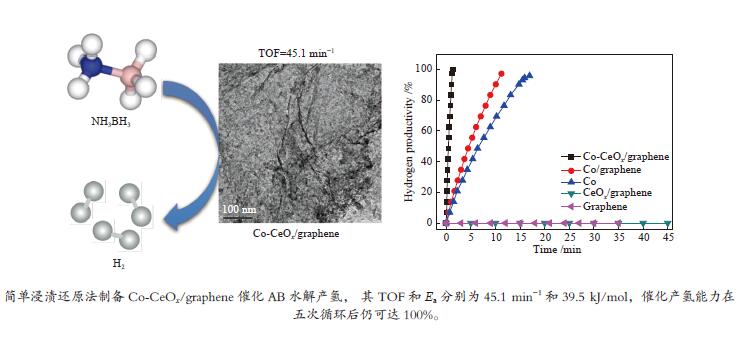
摘要: 高效、低成本催化剂是实现以氨硼烷(NH3BH3, AB)为化学储氢材料水解产氢的关键。本文采用简单的浸渍还原法,制备了石墨烯负载的Co-CeOx 纳米复合物(Co-CeOx /graphene)催化剂,对其氨硼烷水解产氢的催化性能进行了研究。结果表明,Co与CeOx 之间存在强的协同电子效应,同时高分散的纳米Co-CeOx 粒子与石墨烯之间存在强的金属--载体相互作用,因而所制备的Co-CeOx /graphene催化剂对氨硼烷水解具有良好的催化活性和循环稳定性。Co-CeOx /graphene催化剂上氨硼烷水解的活化能( Ea )为39.5 kJ/mol,反应转化频率(TOF)为45.1 min−1,分别为纯Co及Co/graphene催化剂上的12倍与9倍之多,高于大多数已报道的非贵金属催化剂。Abstract: Low-cost and high-performance catalyst is crucial for hydrogen generation via the hydrolysis of ammonia borane (NH3BH3, AB) as a chemical hydrogen storage material. In this work, Co-CeOx nanocomposites supported on graphene (Co-CeOx/graphene) were prepared through a facile impregnation and chemical reduction method and used as a catalyst in the hydrolytic dehydrogenation of ammonia borane. The results indicate that the as-prepared Co-CeOx/graphene nanocomposite exhibits superior catalytic activity and recycling stability in the hydrolysis of ammonia borane, owing to the ultra-fine size of Co-CeOx particles, the strong synergistic electronic effect between Co and CeOx, as well as the strong metal-support interaction between Co-CeOx and graphene. For the hydrolysis of ammonia borane over the optimized Co-CeOx/graphene catalyst, the turnover frequency (TOF) reaches 45.1 min−1, with the activation energy (Ea) of 39.5 kJ/mol; such a TOF value is 12 times and 9 times higher than those over Co and Co/graphene, respectively, also much higher than those reported for most noble-metal-free catalysts.
-
Key words:
- hydrolytic dehydrogenation /
- ammonia borane /
- catalyst /
- Co-CeOx /
- graphene
-
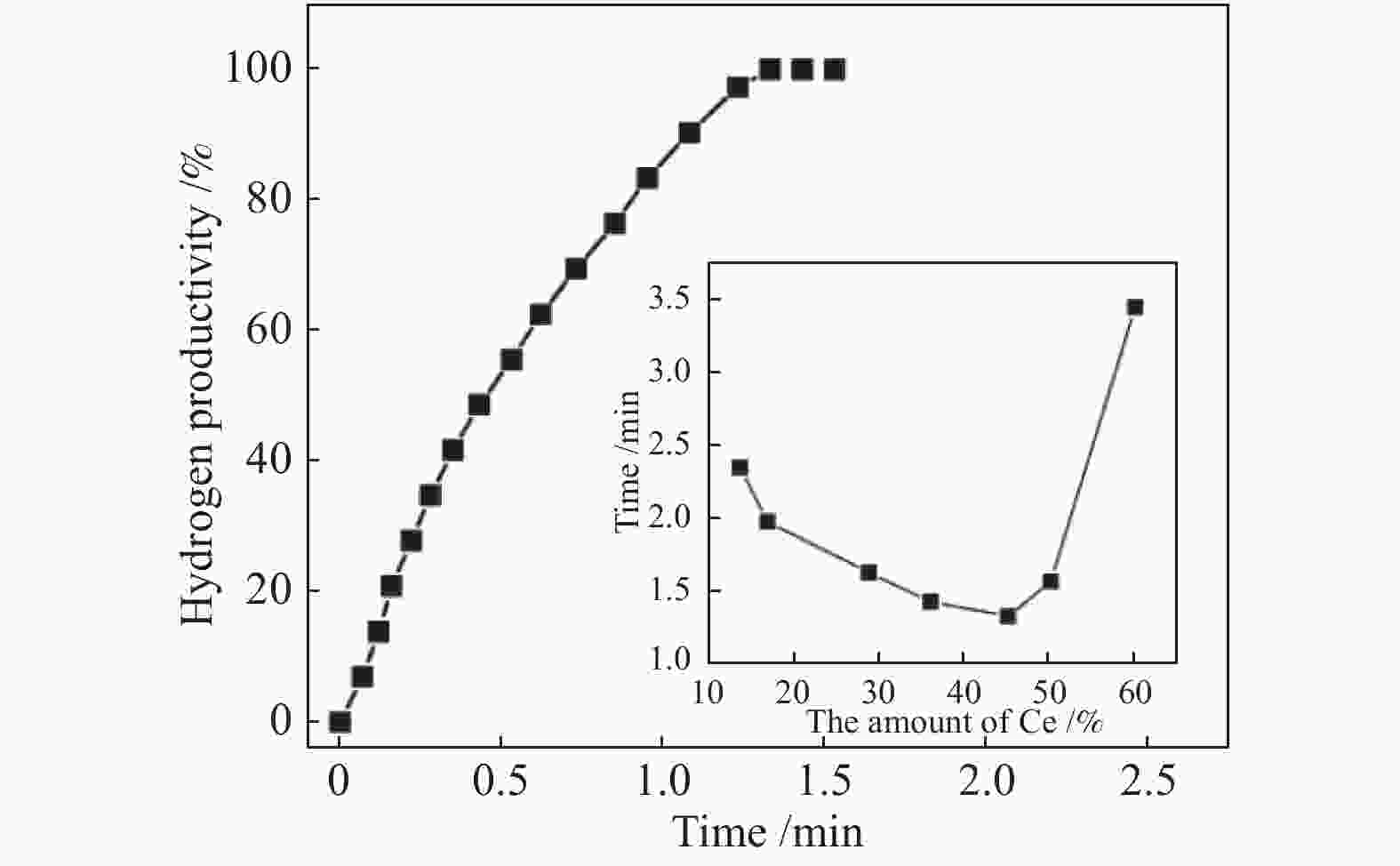
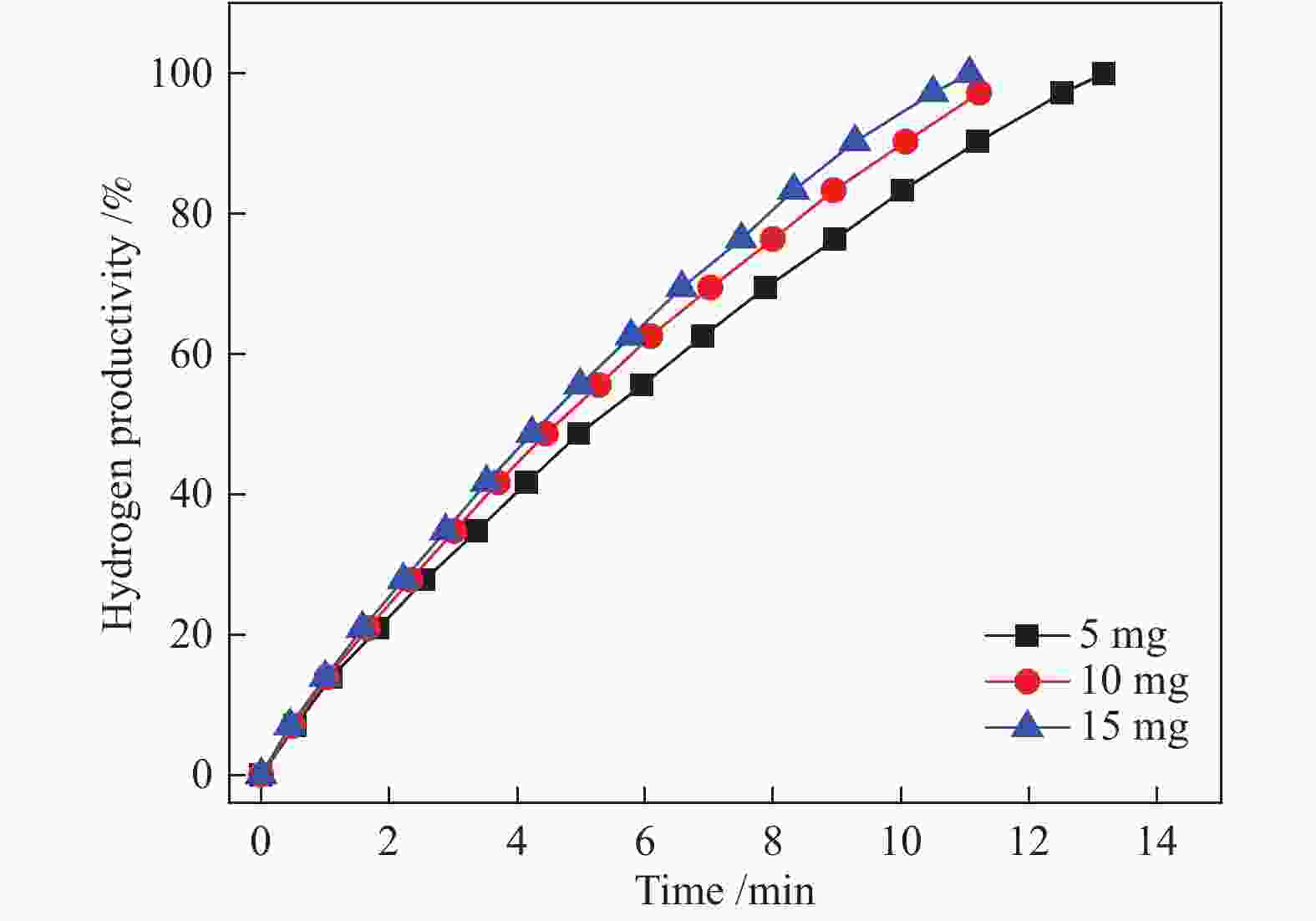
图 6 Co-CeOx/graphene催化剂在298 K下催化氢硼烷水溶液(200 mmol/L, 5 mL)释放氢的生产率与反应时间的关系(Co/AB = 0.05, Ce = 45%);插图显示了Co-CeOx/graphene在不同的Ce含量下催化AB水解脱氢的反应时间
Figure 6 Hydrogen productivity vs. reaction time forhydrolysis of aqueous ammonia borane(200 mmol/L, 5 mL) catalyzed by the Co-CeOx/graphene catalyst at 298 K (Co/AB = 0.05, Ce = 45%); the inset shows the reaction time for the hydrolytic dehydrogenation of AB catalyzed by Co-CeOx/graphene with different molar contents of CeOx
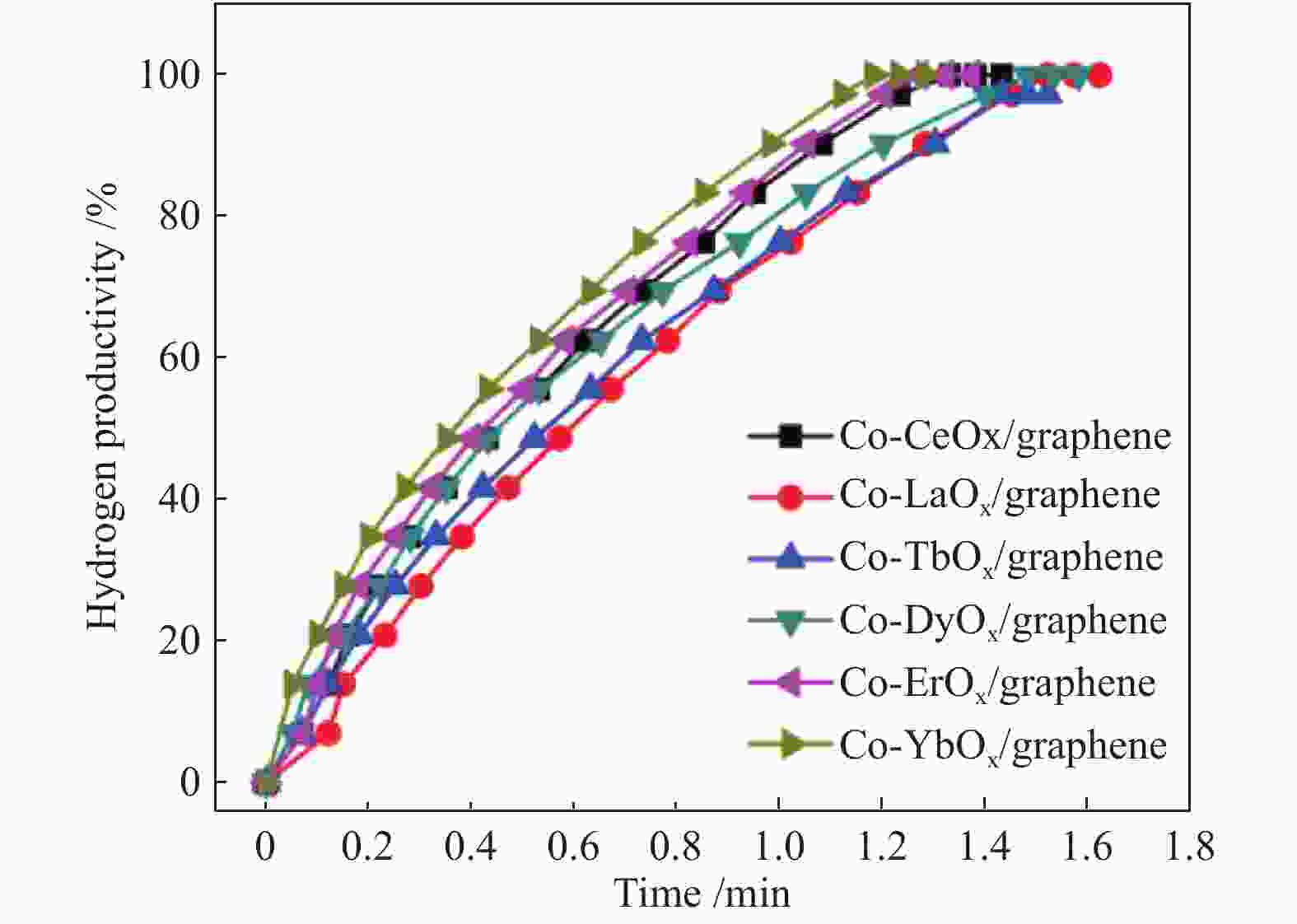
图 7 不同稀土元素的Co-ReOx/graphene催化剂催化氢硼烷水溶液(200 mmol/L, 5 mL)释放氢的生产率与反应时间的关系(Co/AB = 0.05, Re = Ce、La、Tb、Er、Dy and Yb, 45% of Re)
Figure 7 Hydrogen generation from the hydrolysis of ammonia borane (200 mmol/L, 5 mL) catalyzed by Co-ReOx /graphene (Re = Ce, La, Tb, Er, Dy and Yb, 45% of Re) under ambient atmosphere at room temperature (Co/AB = 0.05)
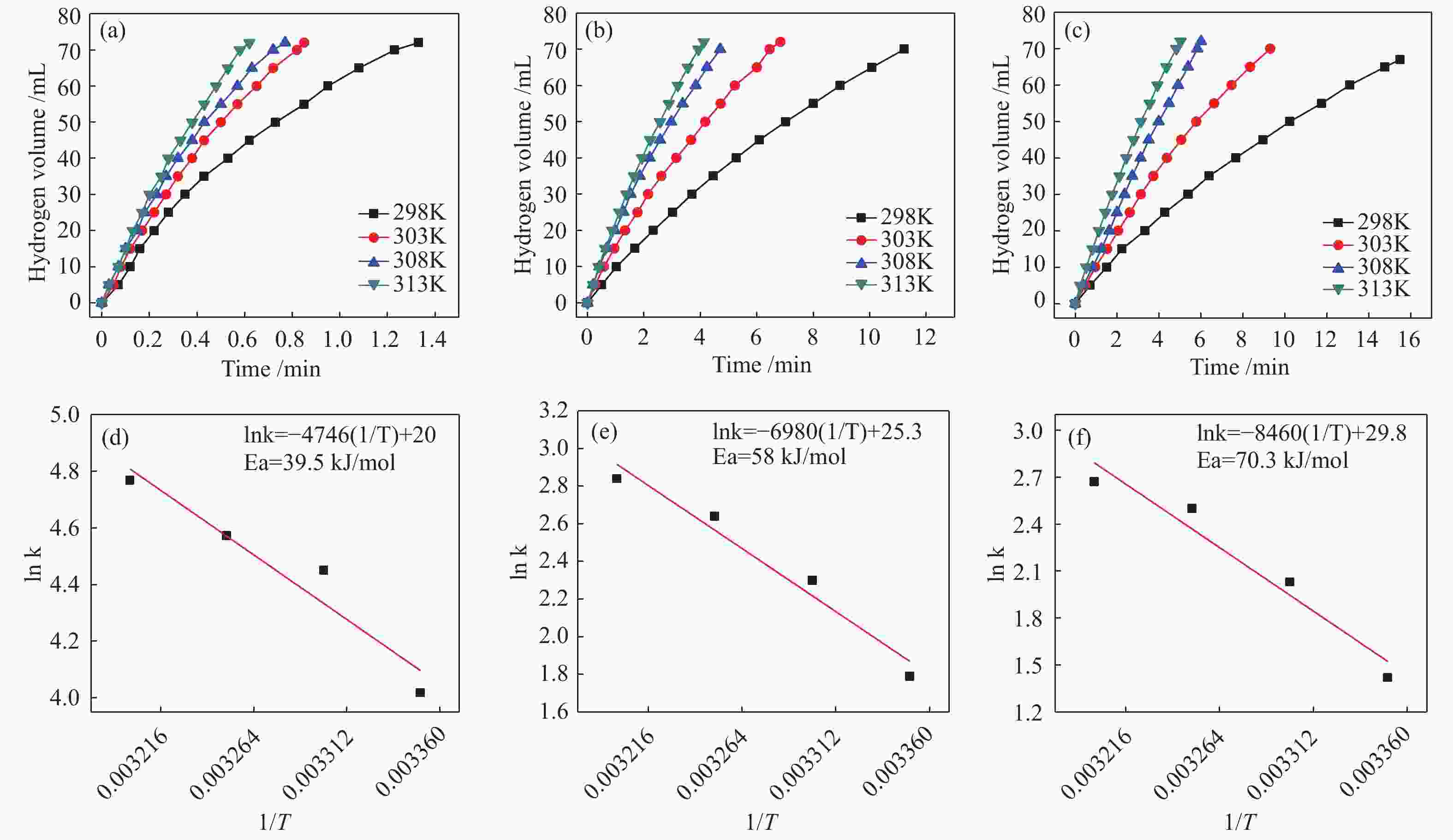
图 9 (a) Co-CeOx/graphene, (b) Co/graphene, (c) Co催化剂在298−313 K下催化氨硼烷溶液(5 mmol/L, 5 mL)放氢量与时间的关系(Co/AB = 0.05, Ce = 45%);(d), (e), (f)分别为(a), (b), (c)对应的阿伦尼乌斯曲线图
Figure 9 Evolution plots of hydrogen generated vs. time for the hydrolysis of ammonia borane (200 mmol/L, 5 mL, Co/AB=0.05) catalyzed by Co-CeOx /graphene (Ce = 45%) (a), Co/graphene (b) and Co (c) at different temperatures (298–313 K); Graphs (d), (e) and (f) are the Arrhenius plots obtained from the Graphs (a), (b) and (c), respectively
表 1 室温下水溶液中AB水解制氢各种非贵金属催化剂的催化活性
Table 1 Catalytic performances of various non-noble metal catalysts for hydrogen generation from the hydrolysis of aqueous AB at room temperature
Catalyst TOF/
$({{\bf{mo} }{ {\bf{l} }_{ { {\rm{H} }_{_2} } } }\cdot{\bf{mol} }_{ {\bf{metal} } }^{\bf{ - 1}}\cdot{\bf{mi} }{ {\bf{n} }^{\bf{ - 1}} }} )$Ea/
(kJ·mol−1)Ref. Co/MIL-101-1-U 51.4 31.3 [34] CoNi/RGO 19.5 39.9 [22] Ag@CoNi/graphene 99.3 36.15 [35] Co-(CeOx)0.91/NGH 79.5 31.82 [36] Co/graphene 13.80 32.75 [37] Ag@Co/graphene 102.4 20.03 [38] Ag@Ni /graphene 77 49.56 [38] Cu@CoNi/graphene 15.46 58.41 [39] Co0.75B0.25 7.24 40.85 [40] Co0/CeO2 7.0 43 [33] Ni-CeOx/graphene 68.2 28.9 [41] Co/γ-Al2O3 2.3 62 [42] Co-CeOx/graphene 45.1 39.5 this work Co/graphene 4.75 58.0 this work Co 3.68 70.3 this work -
[1] 肖国鹏, 乔韦军, 王丽宝, 张磊, 张健, 王宏浩. LaNiO3的焙烧温度对甲醇水蒸气重整制CuO/LaNiO3催化剂的影响[J]. 燃料化学学报,2020,48(2):213−220. doi: 10.3969/j.issn.0253-2409.2020.02.011XIAO Guo-peng, QIAO Wei-jun, WANG Li-bao, ZHANG Lei, ZHANG Jian, WANG Hong-hao. Effect of calcination temperature of LaNiO3 on CuO/LaNiO3 catalyst for hydrogen production via methanol steam reforming[J]. J Fuel Chem Technol,2020,48(2):213−220. doi: 10.3969/j.issn.0253-2409.2020.02.011 [2] ZHANG J, DONG Y N, LIU Q X, ZHOU M, MI G, DU X G. Hierarchically alloyed Pd-Cu microarchitecture with tunable shapes: Morphological engineering, and catalysis for hydrogen evolution reaction of ammonia borane[J]. Int J Hydrog Energy,2019,44(57):30226−30236. doi: 10.1016/j.ijhydene.2019.09.213 [3] 卢培静, 蔡夫锋, 张军, 刘予宇, 孙予罕. B改性CuZnAlOx催化剂对甲醇水蒸气重整制氢性能的研究[J]. 燃料化学学报,2019,47(7):791−798. doi: 10.3969/j.issn.0253-2409.2019.07.003LU Pei-jing, CAI Fu-feng, ZHANG Jun, LIU Yu-yu, SUN Yu-han. Hydrogen production from methanol steam reforming over B-modified CuZnAlOx catalysts[J]. J Fuel Chem Technol,2019,47(7):791−798. doi: 10.3969/j.issn.0253-2409.2019.07.003 [4] WU H, WU M, WANG B Y, YONG X, LIU Y S, LI B J, LIU B Z, LU S Y. Interface electron collaborative migration of Co-Co3O4/carbon dots: Boosting the hydrolytic dehydrogenation of ammonia borane[J]. J Energy Chem,2020,48(9):43−53. [5] AKBAYRAK S, ÖZKAR S. Ammonia borane as hydrogen storage materials[J]. Int J Hydrog Energy,2018,43(40):18592−18606. doi: 10.1016/j.ijhydene.2018.02.190 [6] FENG Y F, WANG H Z, CHEN X D, LV F, LI Y Z, ZHU Y M, XU C J, ZHANG X B, LIU H R, LI H. Simple synthesis of Cu2O-CoO nanoplates with enhanced catalytic activity for hydrogen production from ammonia borane hydrolysis[J]. Int J Hydrog Energy,2020,45(35):17164−17173. doi: 10.1016/j.ijhydene.2020.04.257 [7] ZHANG H, GU X J, LIU P L, SONG J, CHENG J, SU H Q. Highly efficient visible-light-driven catalytic hydrogen evolution from ammonia borane using non-precious metal nanoparticles supported by graphitic carbon nitride[J]. J Mater Chem A,2017,5(5):2288−2296. doi: 10.1039/C6TA08987A [8] SANYAL U, DEMIRCI U B, JAGIRDAR B R, MIELE P. Hydrolysis of ammonia borane as a hydrogen source: Fundamental issues and potential solutions towards implementation[J]. ChemSusChem,2011,4(12):1731−1739. doi: 10.1002/cssc.201100318 [9] PENG S G, LIU J C, ZHANG J, WANG F Y. An improved preparation of graphene supported ultrafine ruthenium(0) NPs: Very active and durable catalysts for H2 generation from methanolysis of ammonia borane[J]. Int J Hydrog Energy,2015,40(34):10856−10866. doi: 10.1016/j.ijhydene.2015.06.113 [10] YURDERI M, BULUT A, ERTAS I E, ZAHMAKIRAN M, KAYA M. Supported copper-copper oxide nanoparticles as active, stable and low-cost catalyst in the methanolysis of ammonia-borane for chemical hydrogen storage[J]. Appl Catal B: Environ,2016,186:212−212. doi: 10.1016/j.apcatb.2016.01.028 [11] ZHOU L M, MENG J, LI P, TAO Z L, MAI L Q, CHEN J. Ultrasmall cobalt nanoparticles supported on nitrogen-doped porous carbon nanowires for hydrogen evolution from ammonia borane[J]. Mater Horiz,2017,4(2):268−273. doi: 10.1039/C6MH00534A [12] CHEN W Y, LI D L, WANG Z J, QIAN G, SUI Z J, DUAN X Z, ZHOU X G, YEBOAH I, CHEN D. Reaction mechanism and kinetics for hydrolytic dehydrogenation of ammonia borane on a Pt/CNT catalyst[J]. AIChE J,2017,63(1):60−65. doi: 10.1002/aic.15389 [13] TONBUL Y, AKBAYRAK S, ÖZKAR S. Magnetically separable rhodium nanoparticles as catalysts for releasing hydrogen from the hydrolysis of ammonia borane[J]. J Colloid Interface Sci,2019,553:581−587. doi: 10.1016/j.jcis.2019.06.038 [14] AKBAYRAK S, TONBUL, Y, ÖZKAR S. Ceria supported rhodium nanoparticles: Superb catalytic activity in hydrogen generation from the hydrolysis of ammonia borane[J]. Appl Catal B: Environ,2016,198:162−170. doi: 10.1016/j.apcatb.2016.05.061 [15] ZOU H T, ZHANG S L, HONG X L, YAO Q L, LUO Y, LU Z H. Immobilization of Ni-Pt nanoparticles on MIL-101/rGO composite for hydrogen evolution from hydrous hydrazine and hydrazine borane[J]. J Alloy Compd,2020,835:155426. doi: 10.1016/j.jallcom.2020.155426 [16] YAO Q L, SHI W M, FENG G, LU Z H, ZHANG X L, TAO D J, KONG D J, CHEN X S. Ultrafine Ru nanoparticles embedded in SiO2 nanospheres: Highly efficient catalysts for hydrolytic dehydrogenation of ammonia borane[J]. J Power Sources,2014,257:293−299. doi: 10.1016/j.jpowsour.2014.01.122 [17] DU X Q, TAN S Y, CAI P, LUO W, CHENG G Z. A RhNiP/rGO hybrid for efficient catalytic hydrogen generation from an alkaline solution of hydrazine[J]. J Mater Chem A,2016,4(38):14572−14576. doi: 10.1039/C6TA05917A [18] ZHANG H, GU X J, SONG J. Co, Ni-based nanoparticles supported on graphitic carbon nitride nanosheets as catalysts for hydrogen generation from the hydrolysis of ammonia borane under broad-spectrum light irradiation[J]. Int J Hydrog Energy,2020,45(41):21273−211286. doi: 10.1016/j.ijhydene.2020.05.178 [19] LIANG Z J, XIAO X Z, YU X Y, HUANG X, JIANG Y Q, FAN X L, CHEN L X. Non-noble trimetallic Cu-Ni-Co nanoparticles supported on metal-organic frameworks as highly efficient catalysts for hydrolysis of ammonia borane[J]. J Alloy Compd,2018,741:501−508. doi: 10.1016/j.jallcom.2017.12.151 [20] WANG Q T, ZHANG F F, DU F L, LIU T. A cost effective cobalt nickel nanoparticles catalyst with exceptional performance for hydrolysis of ammonia-borane[J]. J Nanosci Nanotechnol,2017,17(12):9333−9338. doi: 10.1166/jnn.2017.14331 [21] YANG Y W, FENG G, LU Z H, HU N, ZHANG F, CHEN X S. In situ synthesis of reduced graphene oxide supported Co nanoparticles as efficient catalysts for hydrogen generation from NH3BH3[J]. Acta Phys-Chim Sin,2014,30(6):1180−1186. doi: 10.3866/PKU.WHXB201404141 [22] YANG Y W, ZHANG F, WANG H L, YAO Q L, CHEN X S, LU Z H. Catalytic hydrolysis of ammonia borane by cobalt nickel nanoparticles supported on reduced graphene oxide for hydrogen generation[J]. J Nanomater,2014,1−9. [23] ZHANG Z J, LU Z H, TAN H L, CHEN X S, YAO Q L. CeOx-modified RhNi nanoparticles grown on rGO as highly efficient catalysts for complete hydrogen generation from hydrazine borane and hydrazine[J]. J Mater Chem A,2015,3(46):23520−23529. doi: 10.1039/C5TA06197K [24] DU X Q, LIU C, DU C, CAI P, CHENG G Z, LUO W. Nitrogen-doped graphene hydrogel-supported NiPt-CeOx nanocomposites and their superior catalysis for hydrogen generation from hydrazine at room temperature[J]. Nano Res,2017,10(8):2856−2865. doi: 10.1007/s12274-017-1494-6 [25] CAO N, LUO W, CHENG G Z. One-step synthesis of graphene supported Ru nanoparticles as efficient catalysts for hydrolytic dehydrogenation of ammonia borane[J]. Int J Hydrog Energy,2013,38(27):11964−11972. doi: 10.1016/j.ijhydene.2013.06.125 [26] UZUNDURUKAN A, DEVRIM Y. Carbon nanotube-graphene hybrid supported platinum as an effective catalyst for hydrogen generation from hydrolysis of ammonia borane[J]. Int J Hydrog Energy,2019,44(49):26773−26782. doi: 10.1016/j.ijhydene.2019.08.153 [27] FERNANDE R, PATEL N, MIOTELLO A, JAISWAL R, KOTHARI D C. Dehydrogenation of ammonia borane with transition metal-doped Co-B alloy catalysts[J]. Int J Hydrog Energy,2012,37(3):2397−2406. doi: 10.1016/j.ijhydene.2011.10.119 [28] YAO Q L, LU Z H, WEI H, CHEN X S, ZHU J. Highly Pt-like activity of Ni-Mo/graphene catalyst for hydrogen evolution from hydrolysis of ammonia borane[J]. J Mater Chem A,2016,4(22):8579−8583. doi: 10.1039/C6TA02004F [29] YAN J M, ZHANG X B, HAN S, SHIOYAMA H, XU Q. Iron-nanoparticle-catalyzed hydrolytic dehydrogenation of ammonia borane for chemical hydrogen storage[J]. Angew Chem Int Ed,2008,47(12):2287−2289. doi: 10.1002/anie.200704943 [30] MCINTYRE N S, JOHNSTON D D, COATSWORTH L L, DAVIDSON R D. X‐ray photoelectron spectroscopic studies of thin film oxides of cobalt and molybdenum[J]. Surf Interface Anal,1990,15(4):265−272. doi: 10.1002/sia.740150406 [31] ZHU F F, CHEN G Z, SUN S X, SUN X. In situ growth of Au@CeO2 core-shell nanoparticles and CeO2 nanotubes from Ce(OH)CO3 nanorods[J]. J Mater Chem A,2013,1(2):288−294. doi: 10.1039/C2TA00293K [32] YAO Q L, SHI Y, ZHANG X L, CHEN X S, LU Z H. Facile synthesis of platinum–cerium(IV) oxide hybrids arched on reduced graphene oxide catalyst in reverse micelles with high activity and durability for hydrolysis of ammonia borane[[J]. Chem-Asian J,2016,11(22):3251−3257. doi: 10.1002/asia.201601147 [33] AKBAYRAK S, TANEROĞLU O, ÖZKAR S. Nanoceria supported cobalt(0) nanoparticles: a magnetically separable and reusable catalyst in hydrogen generation from the hydrolysis of ammonia borane[J]. New J Chem,2017,41(14):6546−6552. doi: 10.1039/C7NJ01035D [34] LIU P L, GU X J, KANG K, ZHANG H, CHENG J, SU H Q. Highly efficient catalytic hydrogen evolution from ammonia borane using the synergistic effect of crystallinity and size of noble-metal-free nanoparticles supported by porous metal-organic frameworks[J]. ACS Appl Mater Interfaces,2017,9(12):10759−10767. doi: 10.1021/acsami.7b01161 [35] YANG L, SU J, MENG X Y, LUO W, CHENG G Z. In situ synthesis of graphene supported Ag@CoNi core-shell nanoparticles as highly efficient catalysts for hydrogen generation from hydrolysis of ammonia borane and methylamine borane[J]. J Mater Chem A,2013,1(34):10016−10023. doi: 10.1039/c3ta11835e [36] MEN Y N, SU J, HUANG C Z, LIANG L J, CAI P, CHENG G Z, LUO W. Three-dimensional nitrogen-doped graphene hydrogel supported Co-CeOx nanoclusters as efficient catalysts for hydrogen generation from hydrolysis of ammonia borane[J]. Chin Chem Lett,2018,29:1671−1674. doi: 10.1016/j.cclet.2018.04.009 [37] YANG L, CAO N, DU C, DAI H M, HU K, LUO W, CHENG G Z. Graphene supported cobalt(0) nanoparticles for hydrolysis of ammonia borane[J]. Mater Lett,2014,115:113−116. doi: 10.1016/j.matlet.2013.10.039 [38] YANG L, LUO W, CHENG G Z. Graphene-supported Ag-based core-shell nanoparticles for hydrogen generation in hydrolysis of ammonia borane and methylamine borane[J]. Appl Mater Interfaces,2013,5(16):8231−8240. doi: 10.1021/am402373p [39] MENG X Y, YANG L, CAO N, DU C, HU K, SU J, LUO W, CHENG G Z. Graphene-supported trimetallic core-shell Cu@CoNi nanoparticles for catalytic hydrolysis of amine borane[J]. Chem Plus Chem,2014,79(2):325−332. doi: 10.1002/cplu.201300336 [40] KANTÜRK F A. Dehydrogenation characteristics of ammonia borane via boron-based catalysts (Co-B, Ni-B, Cu-B) under different hydrolysis conditions[J]. Int J Hydrog Energy,2013,38(22):9186−9197. doi: 10.1016/j.ijhydene.2013.05.081 [41] YAO Q L, LU Z H, YANG Y W, CHEN Y Z, CHEN X S, JIANG H L. Facile synthesis of graphene-supported Ni-CeOx nanocomposites as highly efficient catalysts for hydrolytic dehydrogenation of ammonia borane[J]. Nano Res,2018,11:4412−4422. doi: 10.1007/s12274-018-2031-y [42] XU Q, CHANDRA M. Catalytic activities of non-noble metals for hydrogen generation from aqueous ammonia-borane at room temperature[J]. J Power Sources,2006,163(1):364−370. doi: 10.1016/j.jpowsour.2006.09.043 -




 下载:
下载: