Application of density functional theory on the NO-char heterogeneous reduction mechanism in the presence of CO2
-
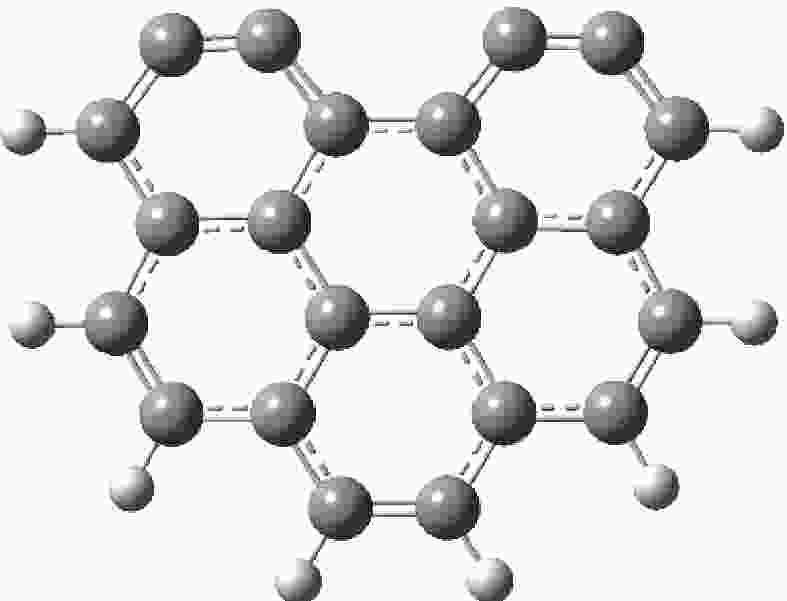
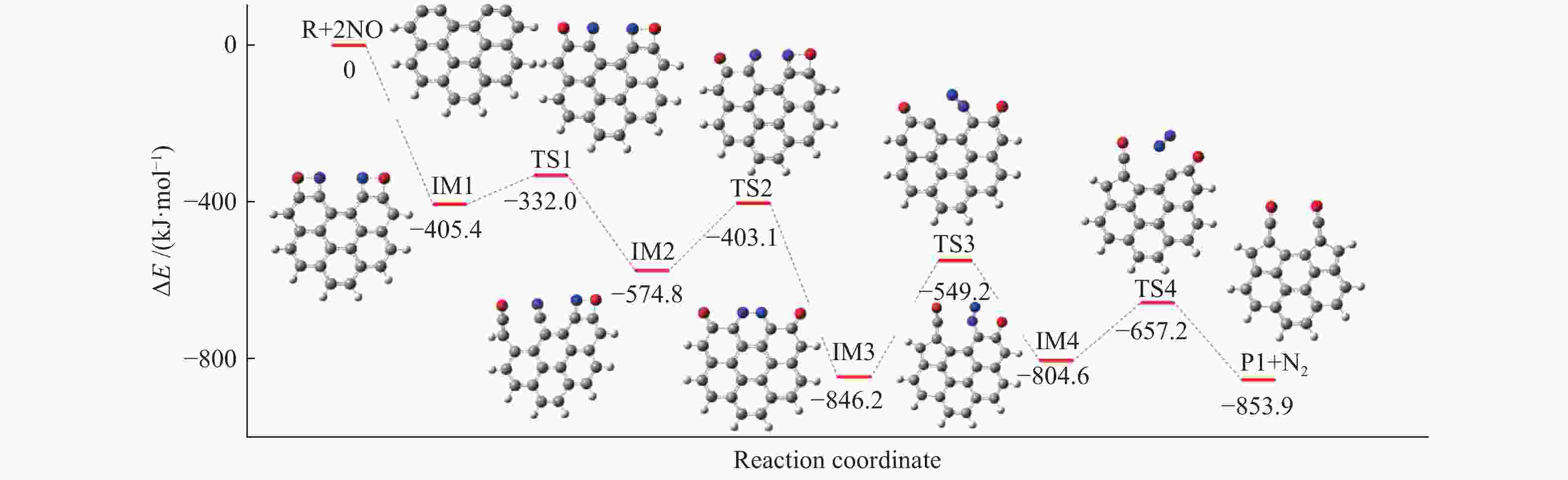
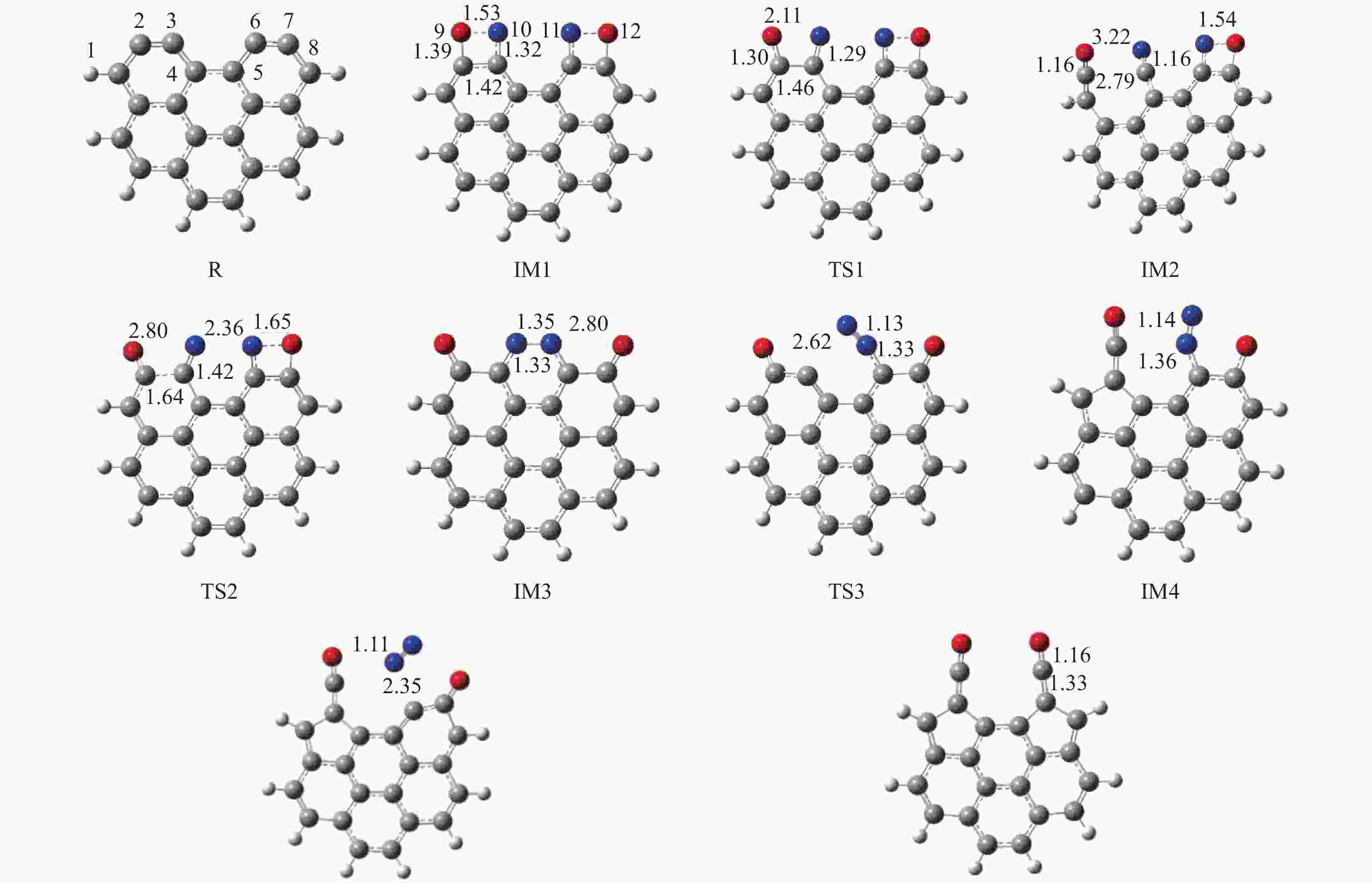
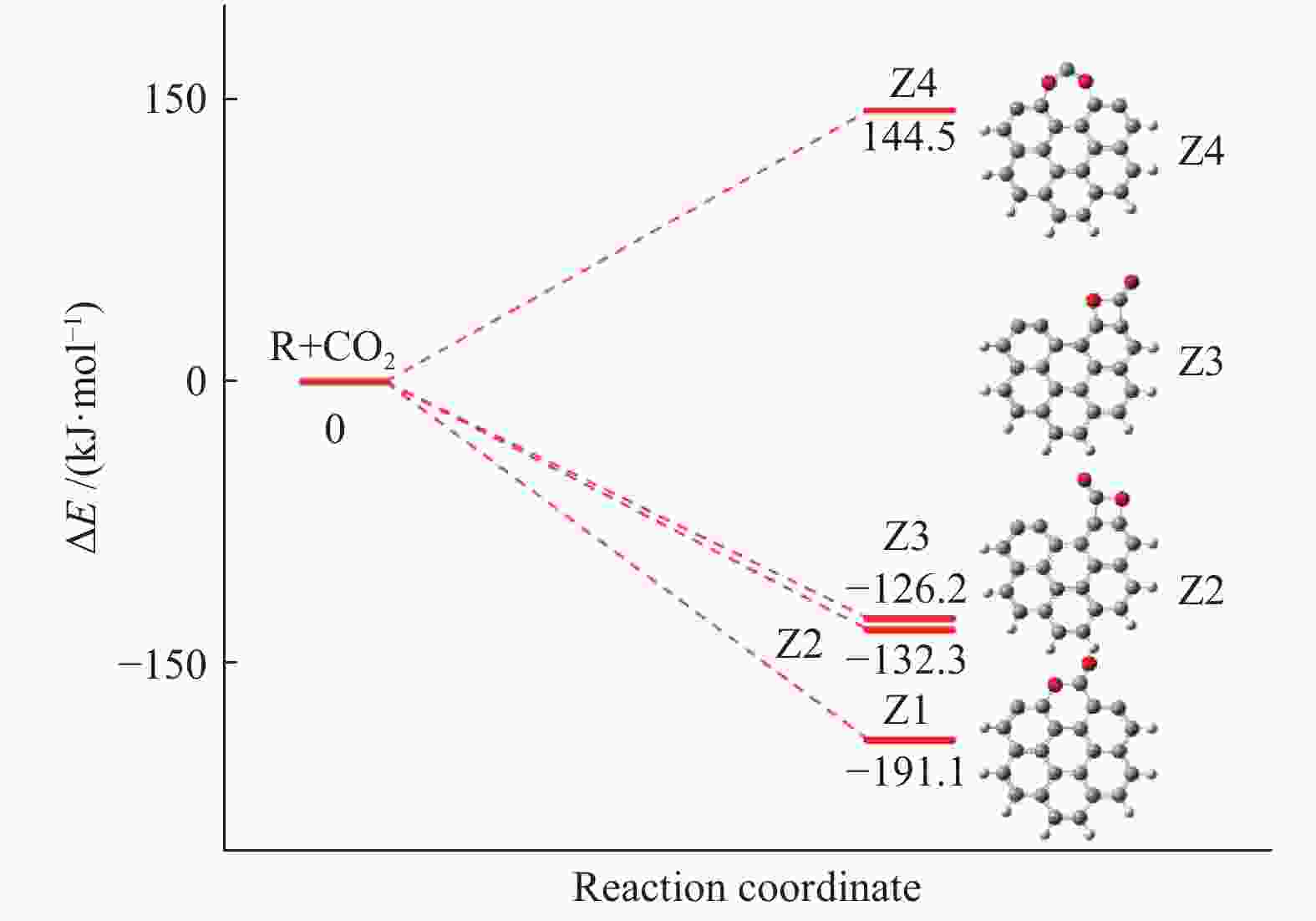
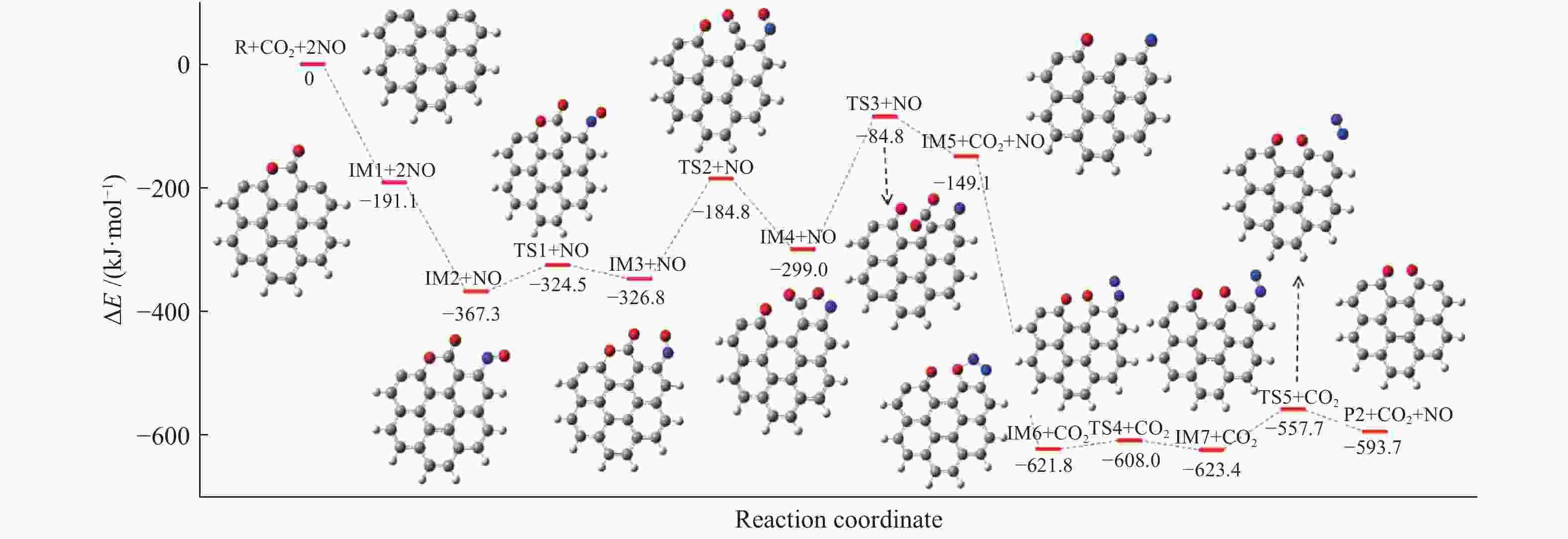
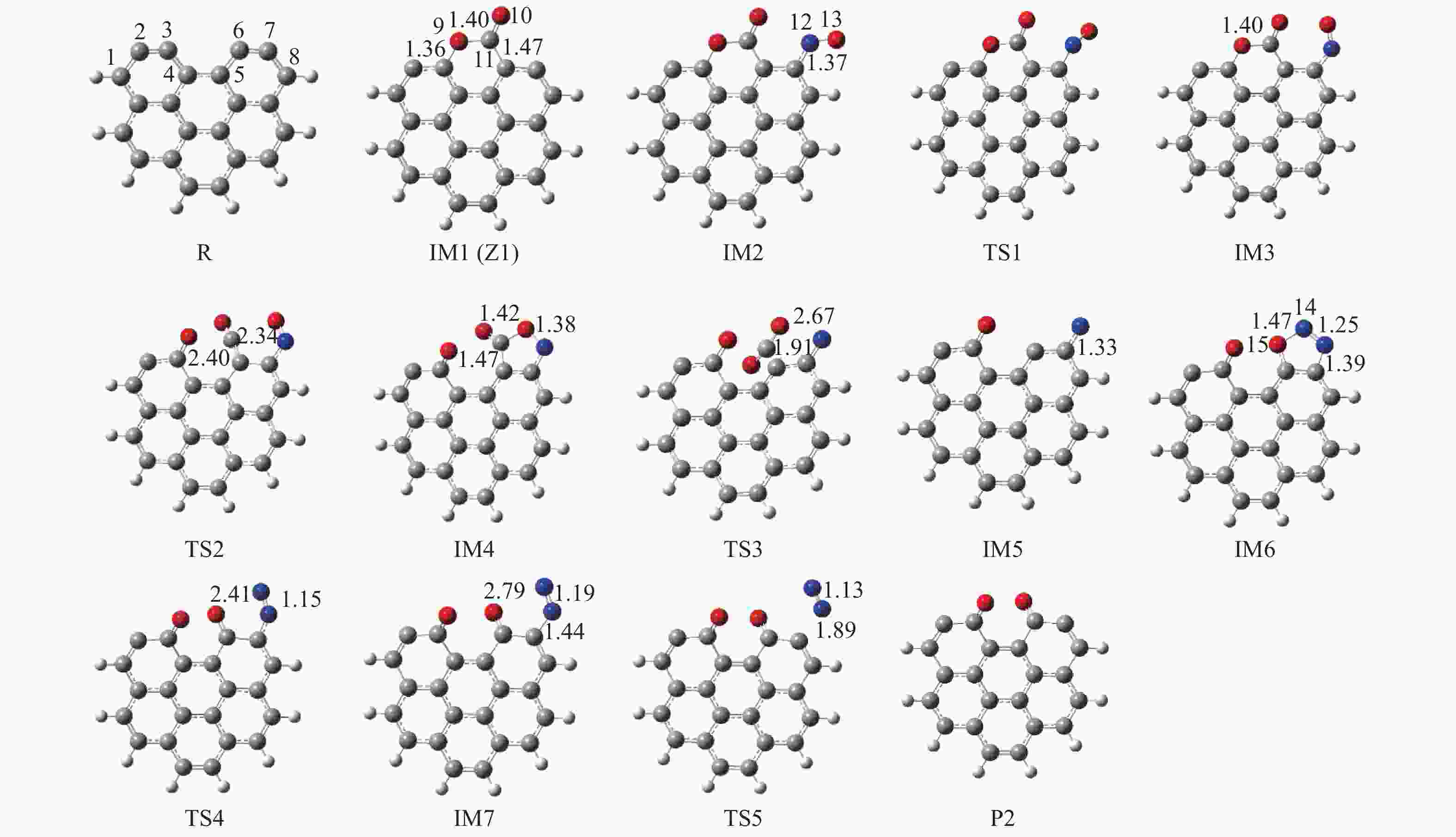
摘要: 为深入理解CO2对NO异相还原的影响,本研究基于密度泛函理论,对CO2参与下的煤焦-NO异相还原反应机理进行研究,并选取armchair苯环模型模拟焦炭表面。结构优化采用B3LYP-D3/6-31G(d)方法,单点能计算采用B3LYP-D3/def2-TZVP方法。研究表明,CO2吸附后形成的羰基与吸附态NO反应生成CO2,继而CO2脱附为后续NO吸附及N2脱附提供邻近的碳活性位点。热力学研究表明,无CO2参与条件下,反应放热853.9 kJ/mol,决速步能垒为297.0 kJ/mol;CO2参与条件下,反应放出593.7 kJ/mol的热量,决速步能垒为214.1 kJ/mol。动力学研究表明,在298.15–1800 K的温度下,CO2参与条件下的反应速率常数大于无CO2参与条件下的反应速率常数。综合热力学和动力学研究结果发现,CO2对NO的异相还原反应具有促进作用。Abstract: In order to obtain the mechanism of the effect of CO2 on the NO heterogeneous reduction, density functional theory (DFT) was adopted to investigate the interactions between char and NO with the participation of CO2. The armchair configuration composed with several aromatic ring clusters was selected as the carbonaceous surfaces. Geometric optimizations were carried out at the B3LYP-D3/6-31G(d) level. Energies of optimized geometries were calculated at the B3LYP-D3/def2-TZVP level. The results show that, the surface carbonyl groups produced by the adsorption of CO2 combine with the adsorbed NO to desorb CO2, thereby providing adjacent carbon active sites for subsequent NO adsorption and N2 desorption. Thermodynamic studies show that the exothermic heat of this reaction is 853.9 kJ/mol, and the highest energy barrier is 297.0 kJ/mol without the participation of CO2, but the exothermic heat of this reaction is 593.7 kJ/mol, and the highest energy barrier is 214.1 kJ/mol with the participation of CO2. Kinetic studies show that over the temperature range of 298.15–1800 K, the reaction rate constants of rate-limiting steps are calculated with conventional transition state theory. The rate constant with the participation of CO2 is higher than that without the participation of CO2. In summary, CO2 plays a promoting role in interacting with NO and char and reducing energy barrier to form N2 directly.
-
Key words:
- CO2 /
- NO /
- heterogeneous reduction /
- density functional theory
-
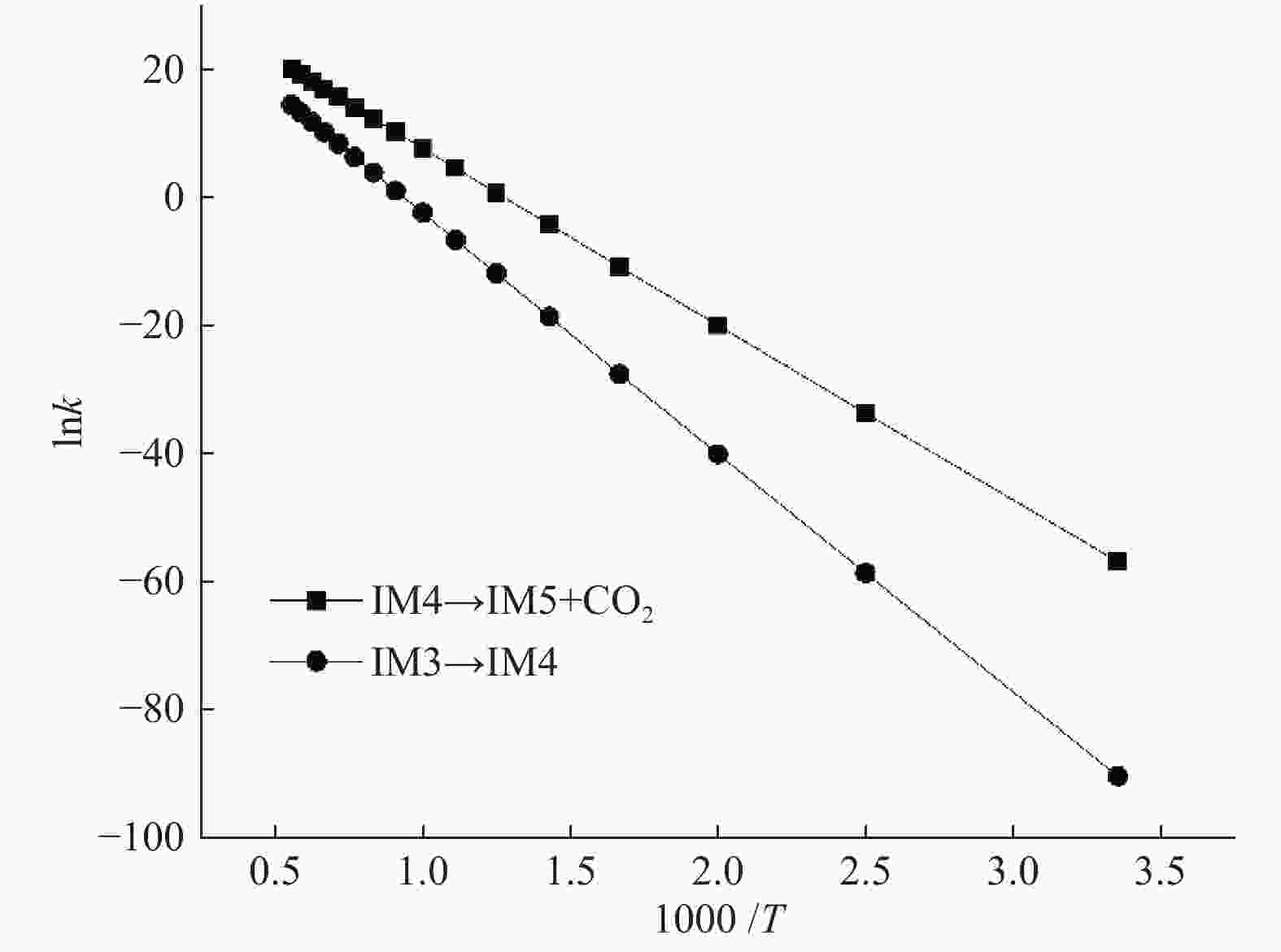
表 1 拟合所得动力学反应参数
Table 1 Fitted kinetic parameters of Arrhenius expressions
Reaction A/s−1 Ea/(kJ·mol−1) IM4→IM5+CO2 2.06 × 1015 228.8 IM3→IM4 1.83 × 1015 311.5 -
[1] 毛洪钧, 李悦宁, 林应超, 王婷, 李维尊, 鞠美庭, 朱复东. 生物质锅炉氮氧化物排放控制技术研究进展[J]. 工程科学学报,2019,41(1):4−14.MAO Hong-jun, LI Yue-ning, LIN Ying-chao, WANG Ting, LI Wei-zun, JU Mei-ting, ZHU Fu-dong. Overview of advances in emission control technologies for nitric oxides from biomass boilers[J]. Chin J Eng,2019,41(1):4−14. [2] BURCH T E, CHEN W, LESTER T W, STERLING A M. Interaction of fuel nitrogen with nitric oxide during reburning with coal[J]. Combust Flame,1994,98(4):391−401. doi: 10.1016/0010-2180(94)90177-5 [3] CHEN W, LONG M. Effect of heterogeneous mechanisms during reburning of nitrogen oxide[J]. Fuel Energy Abstr,1997,42(7):1968−1976. [4] LIU H, HAMPARTSOUMIAN E, GIBBS B M. Evaluation of the optimal fuel characteristics for efficient NO reduction by coal reburning[J]. Fuel,1997,76(11):985−993. doi: 10.1016/S0016-2361(97)00114-2 [5] 张秀霞. 焦炭燃烧过程中氮转化机理与低NOx燃烧技术的开发[D]. 杭州: 浙江大学, 2012.ZHANG Xiu-xia. Nitrogen conversion mechanism during char combustion and develepment of low NOx technology[D]. Hangzhou: Zhejiang University, 2012. [6] CHAMBRION P, ORIKASA H, SUZUKI T, KYOTANI T, TOMITA A. A study of the C-NO reaction by using isotopically labelled C and NO[J]. Fuel,1997,76(6):493−498. doi: 10.1016/S0016-2361(96)00224-4 [7] ILLÁN-GÓMEZ M J, LINARES-SOLANO A, RADOVIC L R, DE LECEA C S-M. NO reduction by activated carbons. 7. some mechanistic aspects of uncatalyzed and catalyzed reaction[J]. Energy Fuels,1996,10(1):158−168. doi: 10.1021/ef950066t [8] PARK D-C, DAY S J, NELSON P F. Nitrogen release during reaction of coal char with O2, CO2, and H2O[J]. Proc Combust Inst,2005,30(2):2169−2175. doi: 10.1016/j.proci.2004.08.051 [9] ZHAO Y J, FENG D D, LI B W, WANG P X, TAN H P, SUN S Z. Effects of flue gases (CO/CO2/SO2/H2O/O2) on NO-char interaction at high temperatures[J]. Energy,2019,174:519−525. [10] LIAO X J, SHAO J A, ZHANG S H, LI X P, CHEN H P. Effects of CO2 and CO on the reduction of NO over calcined limestone or char in oxy-fuel fluidized bed combustion[J]. IET Renew Power Gener,2019,13(10):1633−1640. doi: 10.1049/iet-rpg.2018.6277 [11] 李相鹏, 张世红, 廖新杰, 杨海平, 陈汉平. CFB富氧燃烧中CO2对煤焦与NO还原作用的影响[J]. 工程热物理学报,2016,37(12):2703−2709.LI Xiang-peng, ZHANG Shi-hong, LIAO Xin-jie, YANG Hai-ping, CHEN Han-ping. Effect of CO2 on coal char-NO redcution process in CFB oxy–fuel combustion[J]. J Eng Thermo,2016,37(12):2703−2709. [12] 刘一, 李相鹏, 邵敬爱, 王贤华, 张世红, 陈汉平. CO2气氛下煤焦对NO还原作用的试验研究[J]. 电站系统工程,2015,31(6):10−12+6.LIU Yi, LI Xiang-peng, SHAO Jing-ai, WANG Xian-hua, ZHANG Shi-hong, CHEN Han-ping. Experimental investigation of char effect on NO reduction under CO2 atmosphere[J]. Power Syst Eng,2015,31(6):10−12+6. [13] AARNA I, SUUBERG E M. The role of carbon monoxide in the NO-carbon reaction[J]. Energy Fuels,1999,13(6):1145−1153. doi: 10.1021/ef9900278 [14] SUUBERG E M, LILLY W D, AARNA I. Kinetics and mechanisms of NOx-char reduction. Quarterly technical progress report, August 1, 1995–October 31, 1995[R]. United States: Brown University, 1996. [15] 吕刚, 陆继东, 刘智湘, 薛锦添, 谢新华, 曾阔, 胡芝娟. 分解炉内不同CO2体积分数下的煤粉及煤焦还原NO特性[J]. 燃烧科学与技术,2012,18(1):50−55.LV Gang, LU Ji-dong, LIU Zhi-xiang, XUE Jin-tian, XIE Xin-hua, ZENG Kuo, HU Zhi-juan. NO reduction by coal and char at different CO2 concentrations in Cement Precalciner[J]. J Combust Sci Technol,2012,18(1):50−55. [16] 张永春, 张军. O2/CO2燃烧方式下煤焦−NO反应特性研究[J]. 热力发电,2015,44(07):12−17. doi: 10.3969/j.issn.1002-3364.2015.07.012ZHANG Yong-chun, ZHANG Jun. Characteristics of NO-char reaction in O2/CO2 atmosphere[J]. Therm Power Gener,2015,44(07):12−17. doi: 10.3969/j.issn.1002-3364.2015.07.012 [17] KYOTANI T, TOMITA A. Analysis of the reaction of carbon with NO/N2O using ab initio molecular orbital theory[J]. J Phys Chem B,1999,109:3434−3441. [18] MONTOYA A, TRUONG T N, SAROFIM A F. Application of density functional theory to the study of the reaction of NO with char-bound nitrogen during combustion[J]. J Phys Chem A,2000,104(36):8409−8417. doi: 10.1021/jp001045p [19] SENDT K, HAYNES B S. Density functional study of the chemisorption of O2 on the zigzag surface of graphite[J]. Combust Flame,2005,143(4):629−643. doi: 10.1016/j.combustflame.2005.08.026 [20] SENDT K, HAYNES B S. Density functional study of the chemisorption of O2 across two rings of the armchair surface of graphite[J]. J Phys Chem C,2007,111(14):5465−5473. doi: 10.1021/jp067363r [21] SENDT K, HAYNES B S. Density functional study of the reaction of O2 with a single site on the zigzag edge of graphene[J]. Proc Combust Inst,2011,33(2):1851−1858. doi: 10.1016/j.proci.2010.06.021 [22] MONTOYA A, MONDRAGÓN F, TRUONG T N. CO2 adsorption on carbonaceous surfaces: A combined molecular modeling and experimental study[J]. ACS Div Fuel Chem, Prepr,2001,46:217−219. [23] MONTOYA A, MONDRAGÓN F, TRUONG T N. Formation of CO precursors during char gasification with O2, CO2 and H2O[J]. Fuel Process Technol,2002,77–78:125−130. [24] ROBERTS M J, EVERSON R C, DOMAZETIS G, NEOMAGUS H W J P, JONES J M, VAN SITTERT C G C E, OKOLO G N, VAN NIEKERK D, MATHEWS J P. Density functional theory molecular modelling and experimental particle kinetics for CO2-char gasification[J]. Carbon,2015,93:295−314. doi: 10.1016/j.carbon.2015.05.053 [25] ZHU Z H, LU G Q, FINNERTY J, YANG R T. Electronic structure methods applied to gas–carbon reactions[J]. Carbon,2003,41(4):635−658. doi: 10.1016/S0008-6223(02)00380-9 [26] CHEN N, YANG R T. Ab initio molecular orbital calculation on graphite selection of molecular system and model chemistry[J]. Carbon,1998,36(7):1061−1070. [27] JIAO A Y, ZHANG H, LIU J X, SHEN J, JIANG X M. The role of CO played in the nitric oxide heterogeneous reduction: A quantum chemistry study[J]. Energy,2017,141:1538−1546. [28] ZHANG H, JIANG X M, LIU J X. Updated effect of carbon monoxide on the interaction between NO and char bound nitrogen: A combined thermodynamic and kinetic study[J]. Combust Flame,2020,220:107−118. doi: 10.1016/j.combustflame.2020.06.032 [29] 田向红. 焦炭氧化的密度泛函理论研究[D]. 郑州; 郑州大学, 2019.TIAN Xiang-hong. Study on coke oxidation with density functional theory[J]. Zhengzhou: Zhengzhou University, 2019. [30] STEPHENS P J, DEVLIN F J, CHABALOWSKI C F, FRISCH M J. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields[J]. J Phys Chem,1994,98(45):11623−11627. doi: 10.1021/j100096a001 [31] ZHU Z H, FINNERTY J, LU G Q, YANG R T. A comparative study of carbon gasification with O2 and CO2 by density functional theory calculations[J]. Energy Fuels,2002,16(6):1359−1368. doi: 10.1021/ef0200020 [32] ZHANG H, JIANG X M, LIU J X, SHEN J. Application of density functional theory to the nitric oxide heterogeneous reduction mechanism in the presence of hydroxyl and carbonyl groups[J]. Energ Convers Manage,2014,83:167−176. [33] GRIMME S, ANTONY J, EHRLICH S, KRIEG H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu[J]. J Chem Phys,2010,132:154104. [34] 钟俊, 高正阳, 丁艺, 余岳溪, 杨维结. Zigzag煤焦表面异相还原N2O反应[J]. 煤炭学报,2017,42(11):3028−3034.ZHONG Jun, GAO Zheng-yang, DING Yi, YU Yue-xi, YANG Wei-jie. Heterogeneous reduction reaction of N2O by char based on zigzag carbonaceous model[J]. J China Coal Soc,2017,42(11):3028−3034. [35] GONZALEZ C, SCHLEGEL H B. Reaction path following in mass-weighted internal coordinates[J]. J Phys Chem,1990,94(14):5523−5527. doi: 10.1021/j100377a021 [36] FRISCH M J, TRUCKS G W, SCHLEGEL H B, SCUSERIA G E. Gaussian 09 Rev. D. 01[M]. Wallingford, CT. 2009. [37] 傅献彩. 物理化学[M]. 5版. 北京: 高等教育出版社, 2005.FU Xian-cai. Physical Chemistry[M]. 5th ed. Beijing: Higher Education Press, 2005. [38] CHEN P, GU M Y, CHEN G, LIU F S, LIN Y Y. DFT study on the reaction mechanism of N2O reduction with CO catalyzed by char[J]. Fuel,2019,192(9):1682−1706. -




 下载:
下载: