Effect of thermal treatment temperature on catalytic performance of Pt/TiO2 nanobelt composite for HCHO oxidation
-
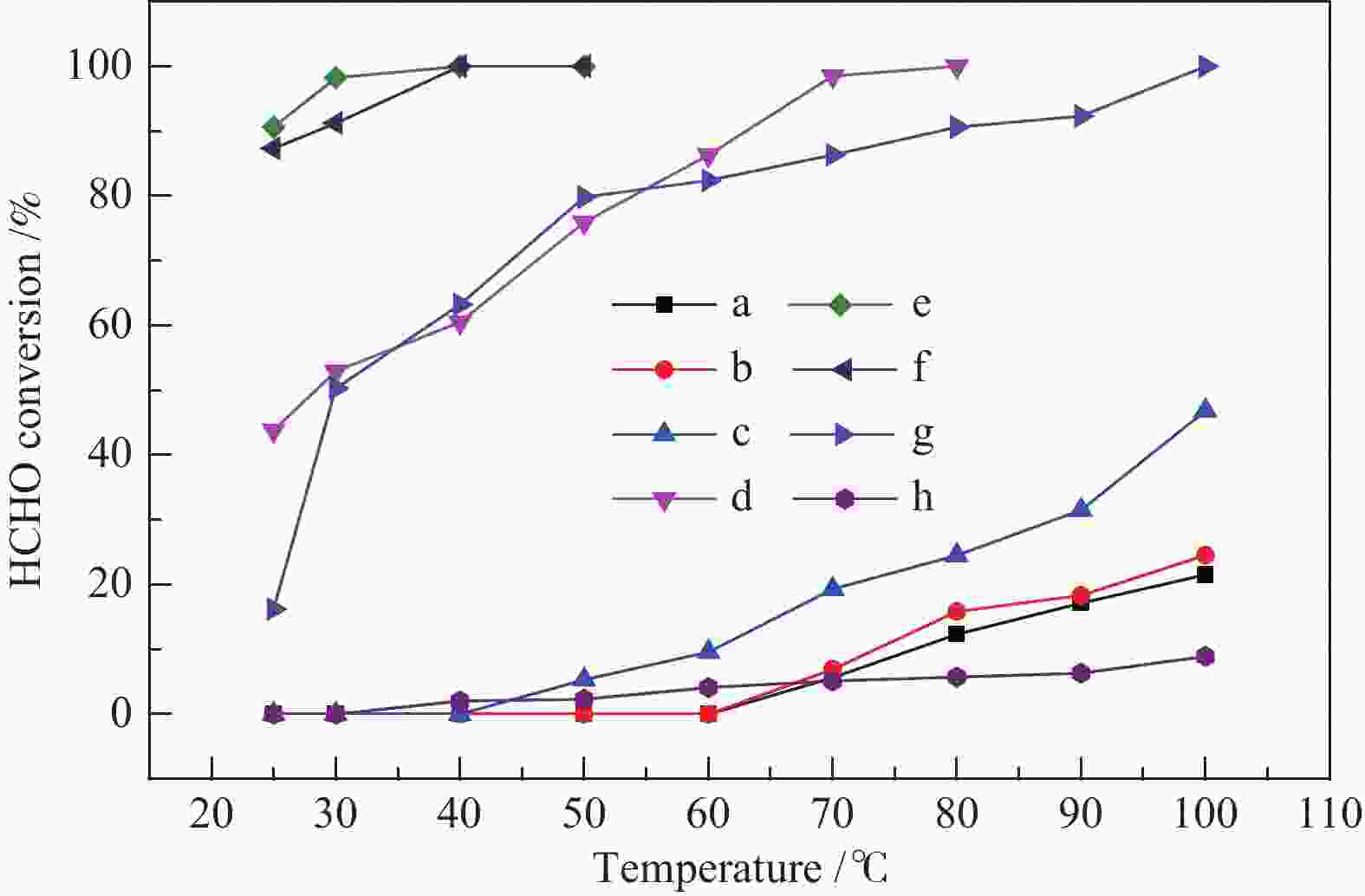
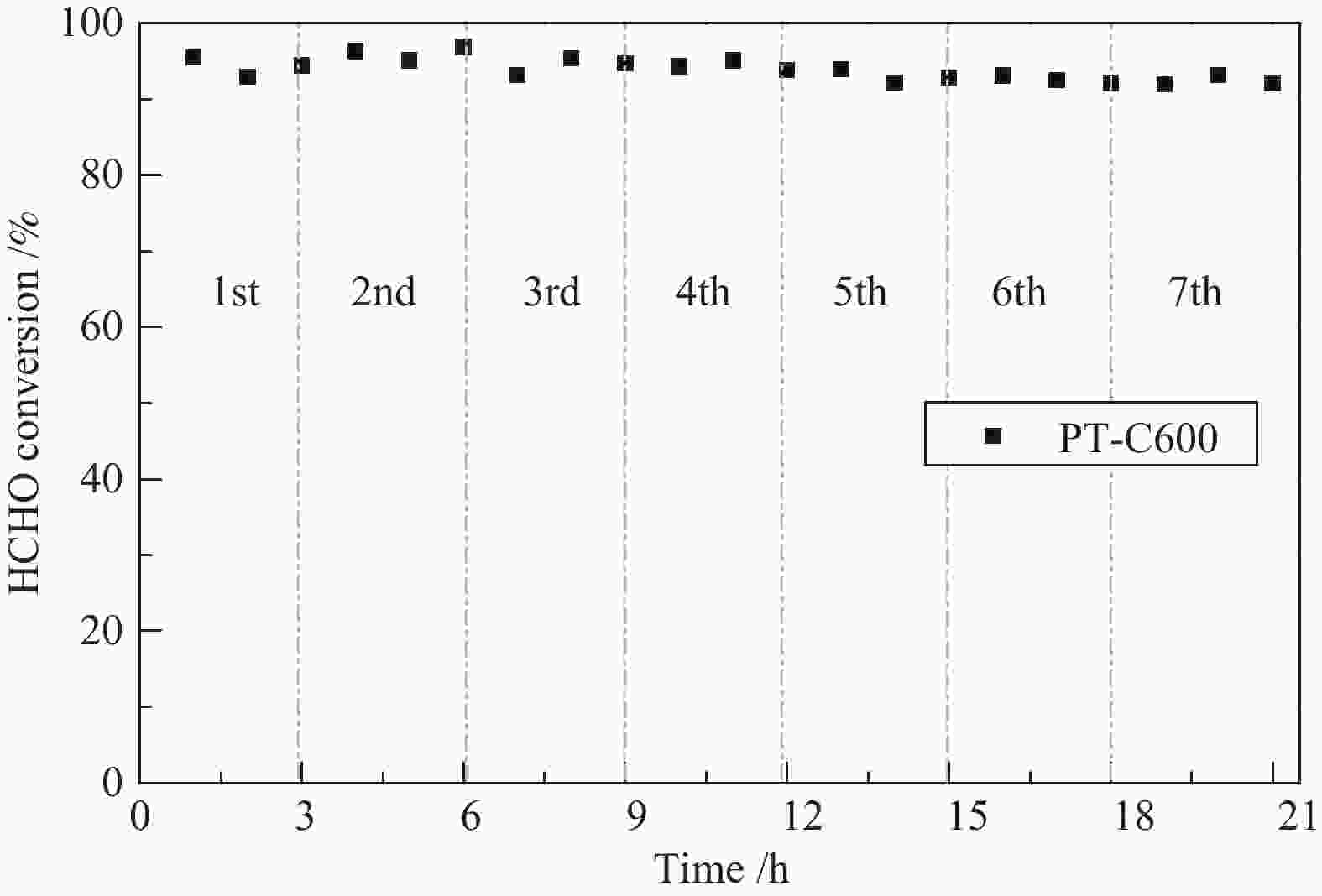
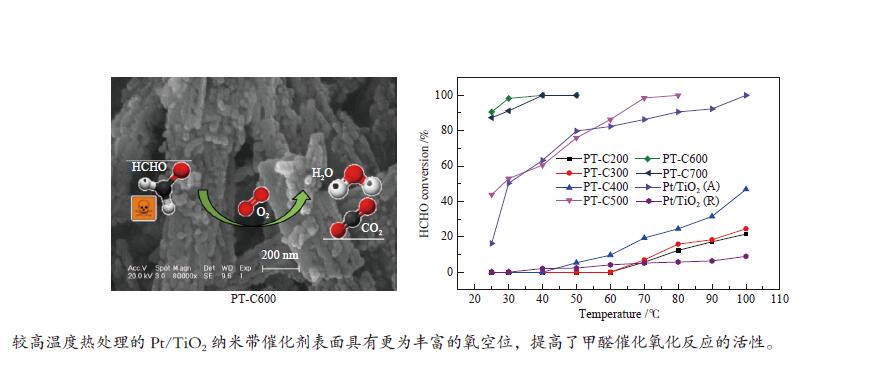
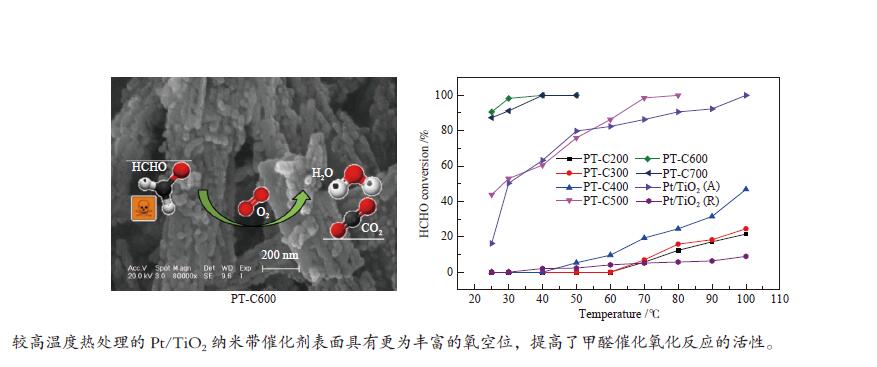
摘要: 采用水热合成法并结合酸处理制备出TiO2纳米带,进行不同温度热处理,在TiO2纳米带上沉积Pt纳米颗粒(NPs),制备出一系列Pt/TiO2纳米带复合物,用于甲醛室温催化氧化。通过各种技术对催化剂进行表征,结果表明,热处理温度对TiO2纳米带的相组成和表面结构,以及表面的氧空位和羟基的数量有较大的影响。在室温下,600 ℃热处理的TiO2纳米带负载Pt催化剂具有更为丰富的氧空位,有利于吸附氧的活化,并形成较多的Ti-(OH)x-Pt物种,表现出较高的催化活性,在25 ℃,相对湿度为55%时,甲醛的转化率为91.6%。Abstract: TiO2 nanobelts were prepared by hydrothermal synthesis and acid treatment, then calcination at different temperatures. And subsequently Pt nanoparticles were deposited on the TiO2 nanobelts. Pt/TiO2 catalytic properties were investigated in the oxidation of formaldehyde. These catalysts were characterized by various techniques and the characterization results showed that the applied thermal treatment temperature greatly influenced the phase composition and surface structure of TiO2 nanobelts, as well as the number of oxygen vacancies and hydroxyl groups on the surface. The Pt/TiO2 nanobelts thermally treated at 600 °C had more oxygen vacancies, which were conducive to the activation of adsorbed oxygen, formed more Ti-(OH)x-Pt species, and showed higher catalytic activity. At 25 °C and relative humidity of 55%, the conversion of formaldehyde reached 91.6%.
-
Key words:
- formaldehyde /
- phase structure /
- thermal treatment temperature /
- oxygen vacancies /
- catalytic oxidation
-
表 1 Pt/TiO2催化剂的物化性质和Pt金属分散度
Table 1 Physical properties and Pt dispersion of the Pt/TiO2 catalyst
Pt/TiO2
catalystABET/
(m2·g−1)Pore volume /
(m3·g−1)Pore size /
nmPt
dispersion/%PT-C200 81 0.22 8.9 42.2 PT-C300 77 0.22 9.3 40.4 PT-C400 70 0.21 9.7 38.2 PT-C500 64 0.18 11.2 37.2 PT-C600 56 0.16 12.0 37.1 PT-C700 51 0.15 12.5 34.3 表 2 PT-C300和PT-C600催化剂的XPS分析
Table 2 XPS analysis results for the catalysts of PT-C300 and PT-C600
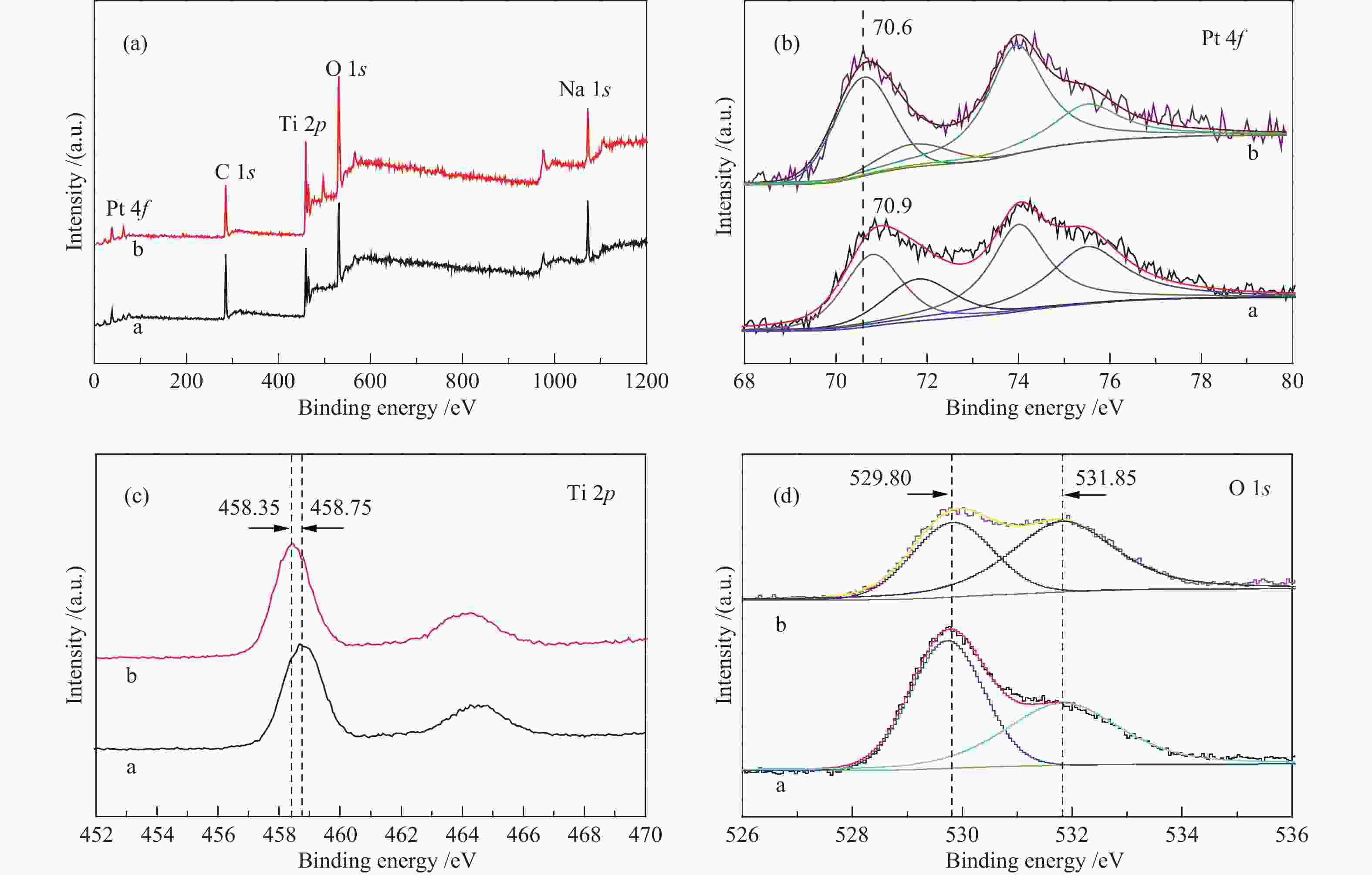
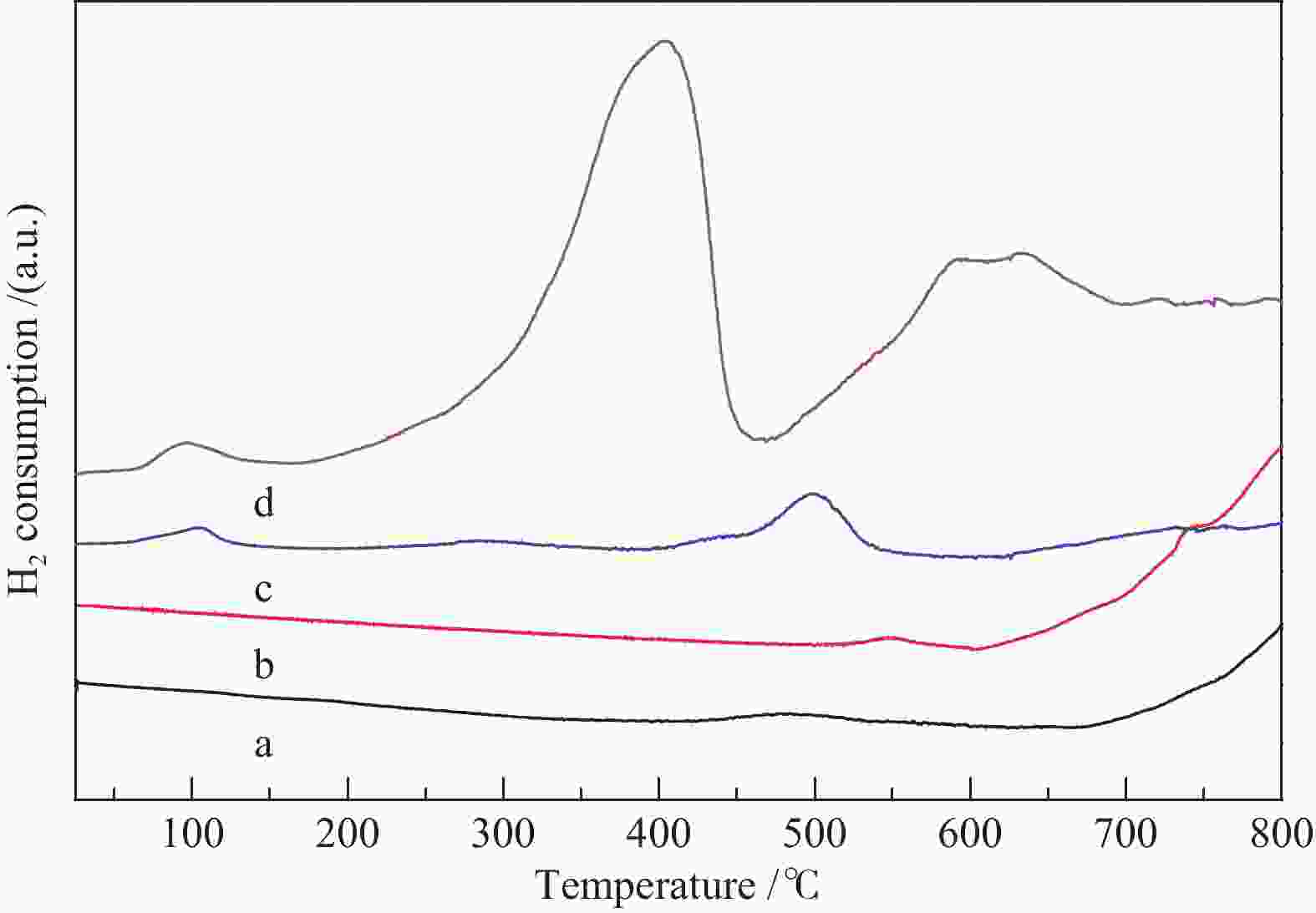
Catalyst BE (eV) Surface atom ratio Pt 4f7/2(Pt 4f5/2) OII (OI) Ti 2p OII /(OI+ OII) Pt/Ti Pt2+/Pt0 PT-C300 70.90(74.10) 531.80(529.75) 458.75 0.462 0.041 0.767 PT-C600 70.60(74.00) 531.85(529.80) 458.35 0.618 0.036 0.361 OⅡ/(OⅠ+ OⅡ) and Pt2+/Pt0 were calculated from the corresponding areas of fitted peaks done by XPSPEAK 4.1 with Shirley background -
[1] TANG X J, BAI Y, DUONG A, SMITH M T, LI L Y, ZHANG L P. Formaldehyde in China: Production, consumption, exposure levels, and health effects[J]. Environ Int,2009,35(8):1210−1224. doi: 10.1016/j.envint.2009.06.002 [2] CHI C C, CHEN W D, GUO M, WENG M L, YAN G, SHEN X Y. Law and features of TVOC and formaldehyde pollution in urban indoor air[J]. Atmos Environ,2016,132(5):85−90. [3] JHON Q T, SEBASTIEN Y, BELLAT J P, GIRAUDON J M, LAMONIER J F. Formaldehyde: Catalytic oxidation as a promising soft way of elimination[J]. ChemSusChem,2013,6:578−592. doi: 10.1002/cssc.201200809 [4] HUANG H B, XU Y, FENG Q Y, LEUNG D Y C. Low temperature catalytic oxidation of volatile organic compounds: A review[J]. Catal Sci Technol,2015,5(2):2649−2669. [5] NIE L H, YU J G, JARONIEC M, TAO F. Room-temperature catalytic oxidation of formaldehyde on catalysts[J]. Catal Sci Technol,2016,6(11):3649−3669. doi: 10.1039/C6CY00062B [6] 拜冰阳, 乔琦, 李俊华, 郝吉明. 甲醛催化氧化催化剂的研究进展[J]. 催化学报,2016,37(1):102−122. doi: 10.1016/S1872-2067(15)61007-5BAI Bing-yang, QIAO Qi, LI Jun-hua, HAO Ji-ming. Progress in research on catalysts for catalytic oxidation of formaldehyde[J]. Chin J Catal,2016,37(1):102−122. doi: 10.1016/S1872-2067(15)61007-5 [7] GUO J H, LIN C X, JIANG C J, ZHANG P Y. Review on noble metal-based catalysts for formaldehyde oxidation at room temperature[J]. Appl Surf Sci,2019,475:237−255. doi: 10.1016/j.apsusc.2018.12.238 [8] HUANG H B, LEUNG D Y C. Complete elimination of indoor formaldehyde over supported Pt catalysts with extremely low Pt content at ambient temperature[J]. J Catal,2011,280:60−67. doi: 10.1016/j.jcat.2011.03.003 [9] ZHANG C B, LIU F D, ZHAI Y P, ARIGA H, YI N, LIU Y C, ASAKURA K, FLYTZANI-STEPHANOPOULOS M, HE H. Alkali-metal-promoted Pt/TiO2 opens a more efficient pathway to formaldehyde oxidation at ambient temperatures[J]. Angew Chem Int Ed,2012,51(38):9628−9632. doi: 10.1002/anie.201202034 [10] QI L F, CHENG B, YU J G, HO W K. High-surface area mesoporous Pt/TiO2 hollow chains for efficient formaldehyde decomposition at ambient temperature[J]. J Hazard Mater,2016,301:522−530. doi: 10.1016/j.jhazmat.2015.09.026 [11] CHEN H Y, RUI Z B, JI H B. Monolith-like TiO2 nanotube array supported Pt catalyst for HCHO removal under mild conditions[J]. Ind Eng Chem Res,2014,53:7629−7636. doi: 10.1021/ie5004009 [12] NIE L H, YU J G, FU J Wl. Complete decomposition of formaldehyde at room temperature over a platinum-decorated hierarchically porous electrospun titania nanofiber mat[J]. ChemCatChem,2014,6:1983−1989. doi: 10.1002/cctc.201301105 [13] NIE L H, ZHOU P, YU J G, JARONIE Mietek. Deactivation and regeneration of Pt/TiO2 nanosheet-type catalysts with exposed (001) facets for room temperature oxidation of formaldehyde[J]. J Mol Catal A: Chem,2014,390:7−13. doi: 10.1016/j.molcata.2014.02.033 [14] CUI W Y, XUE D, YUAN X L, ZHENG B, JIA M J, ZHANG W X. Acid-treated TiO2 nanobelt supported platinum nanoparticles for the catalytic oxidation of formaldehyde at ambient conditions[J]. Appl Surf Sci,2017,411:105−112. doi: 10.1016/j.apsusc.2017.03.169 [15] SU Y, JI K M, XUN J Y, ZHANG K, LIU P, ZHAO L. Catalytic oxidation of low concentration formaldehyde over Pt/TiO2 catalyst[J]. Chin J Chem Eng,2021,29:190−195. doi: 10.1016/j.cjche.2020.04.024 [16] WANG Y, LI Z, CAO Y, LI F, ZHAO W, LIN X Q, YANG J B. Fabrication of novel Ag-TiO2 nanobelts as a photoanode for enhanced photovoltage performance in dye sensitized solar cells[J]. J Alloy Compd,2016,677:294−301. doi: 10.1016/j.jallcom.2016.03.266 [17] LIN J J, SHEN J X, WANG T L, WANG R J, LIU H, CUI J J, ZHOU W J. Enhancement of photocatalytic properties of TiO2 nanobelts through surface-coarsening and surface nanoheterostructure construction[J]. Mater Sci Eng B,2011,176:921−925. doi: 10.1016/j.mseb.2011.05.018 [18] JIANG Z, YANG Y, SHANGGUAN W, JIANG Z. Influence of support and metal precursor on the state and CO catalytic oxidation activity of platinum supported on TiO2[J]. J Phys Chem C,2012,116:19396−19404. doi: 10.1021/jp3061009 [19] SARKAR D, CHATTOPADHYAY K K. Branch density-controlled synthesis of hierarchical TiO2 nanobelt and tunable three-step electron transfer for enhanced photocatalytic property[J]. ACS Appl Mater Interfaces,2014,6:10044−10059. doi: 10.1021/am502379q [20] XU F Y, LE Y, CHENG B, JIANG C X. Effect of calcination temperature on formaldehyde oxidation performance of Pt/TiO2 nanofiber composite at room temperature[J]. Appl Surf Sci,2017,426:333−341. doi: 10.1016/j.apsusc.2017.07.096 [21] QI L F, HO W, WANG J L, ZHANG P Y, YU J G. Enhanced catalytic activity of hierarchically macro-/mesoporous Pt/TiO2 toward room-temperature decomposition of formaldehyde[J]. Catal Sci Technol,2015,5:2366−2377. doi: 10.1039/C4CY01712A [22] YAN Z X, XU Z H,YU J G, JARONIEC M. Highly active mesoporous ferrihydrite supported Pt catalyst for formaldehyde removal at room temperature[J]. Environ Sci Technol,2015,49:6637−6644. [23] ZENG L, SONG W L, LI M H, ZENG D W, XIE C S. Catalytic oxidation of formaldehyde on surface of H-TiO2/H-C-TiO2 without light illumination at room temperature[J]. Appl Catal B: Environ,2014,147:490−498. [24] WANG G M, WANG H Y, LING Y C, TANG Y C, YANG X Y, FITZMORRIS R C, WANG C C, ZHANG J Z, LI Y. Hydrogen-treated TiO2 nanowire arrays for photoelectrochemical water splitting[J]. Nano Lett,2011,11:3026−3033. doi: 10.1021/nl201766h [25] NIE L H, YU J G, LI X Y, CHENG B, LIU G, JARONIEC M. Enhanced performance of NaOH-modified Pt/TiO2 toward room temperature selective oxidation of formaldehyde[J]. Environ Sci Technol,2013,47:2777−2783. doi: 10.1021/es3045949 [26] LIU L Q, ZHOU F, WANG L G, QI X J, SHI F, DENG Y Q. Low-temperature CO oxidation over supported Pt, Pd catalysts: Particular role of FeOx support for oxygen supply during reactions[J]. J Catal,2010,274:1−10. doi: 10.1016/j.jcat.2010.05.022 [27] PENG J X, WANG S D. Correlation between microstructure and performance of Pt/TiO2 catalysts for formaldehyde catalytic oxidation at ambient temperature: effects of hydrogen pretreatment[J]. J Phys Chem C,2007,111:9897−9904. [28] HE M, JI J, LIU B Y, HUANG H B. Reduced TiO2 with tunable oxygen vacancies for catalytic oxidation of formaldehyde at room temperature[J]. Appl Surf Sci,2019,473:934−942. [29] AHMAD W, PARK E, LEE H, KIM J Y, KIM B C, JURNG J, OH Y. Defective domain control of TiO2 support in Pt/TiO2 for room temperature formaldehyde (HCHO) remediation[J]. Appl Surf Sci,2021,538:147504. doi: 10.1016/j.apsusc.2020.147504 [30] LI Y B, WANG C Y, ZHANG C B, HE H. Formaldehyde oxidation on Pd/TiO2 catalysts at room temperature: The effects of surface oxygen vacancies[J]. Top Catal,2020,63:1−7. doi: 10.1007/s11244-020-01254-7 [31] ZHANG C B, HE H, TANAKA K. Catalytic performance and mechanism of a Pt/TiO2 catalyst for the oxidation of formaldehyde at room temperature[J]. Appl Catal B: Environ,2006,65:37−43. doi: 10.1016/j.apcatb.2005.12.010 [32] PENG J X, WANG S D. Performance and characterization of supported metal catalysts for complete oxidation of formaldehyde at low temperatures[J]. Appl Catal B: Environ,2007,73:282−291. doi: 10.1016/j.apcatb.2006.12.012 -




 下载:
下载: