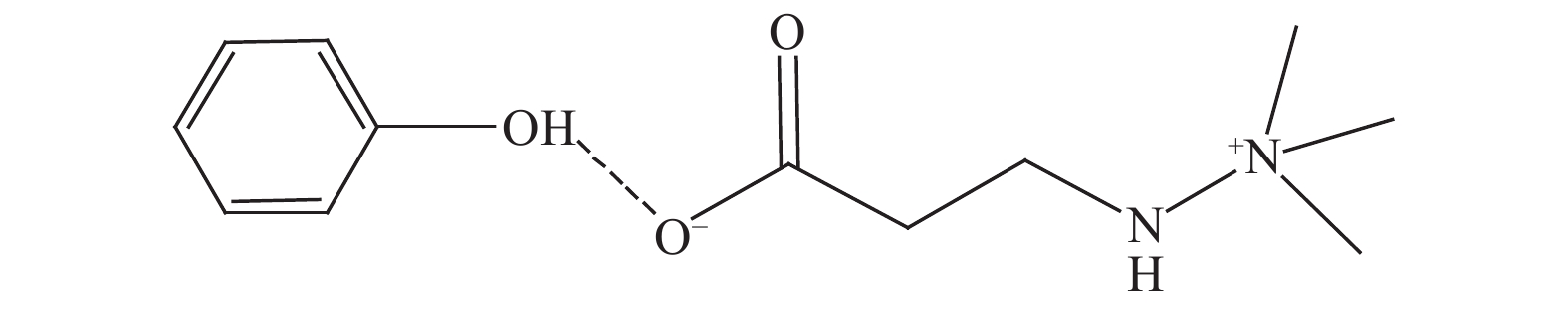
Efficient separation of phenols from phenols-containing oil mixtures via forming hydrogen bonds
-
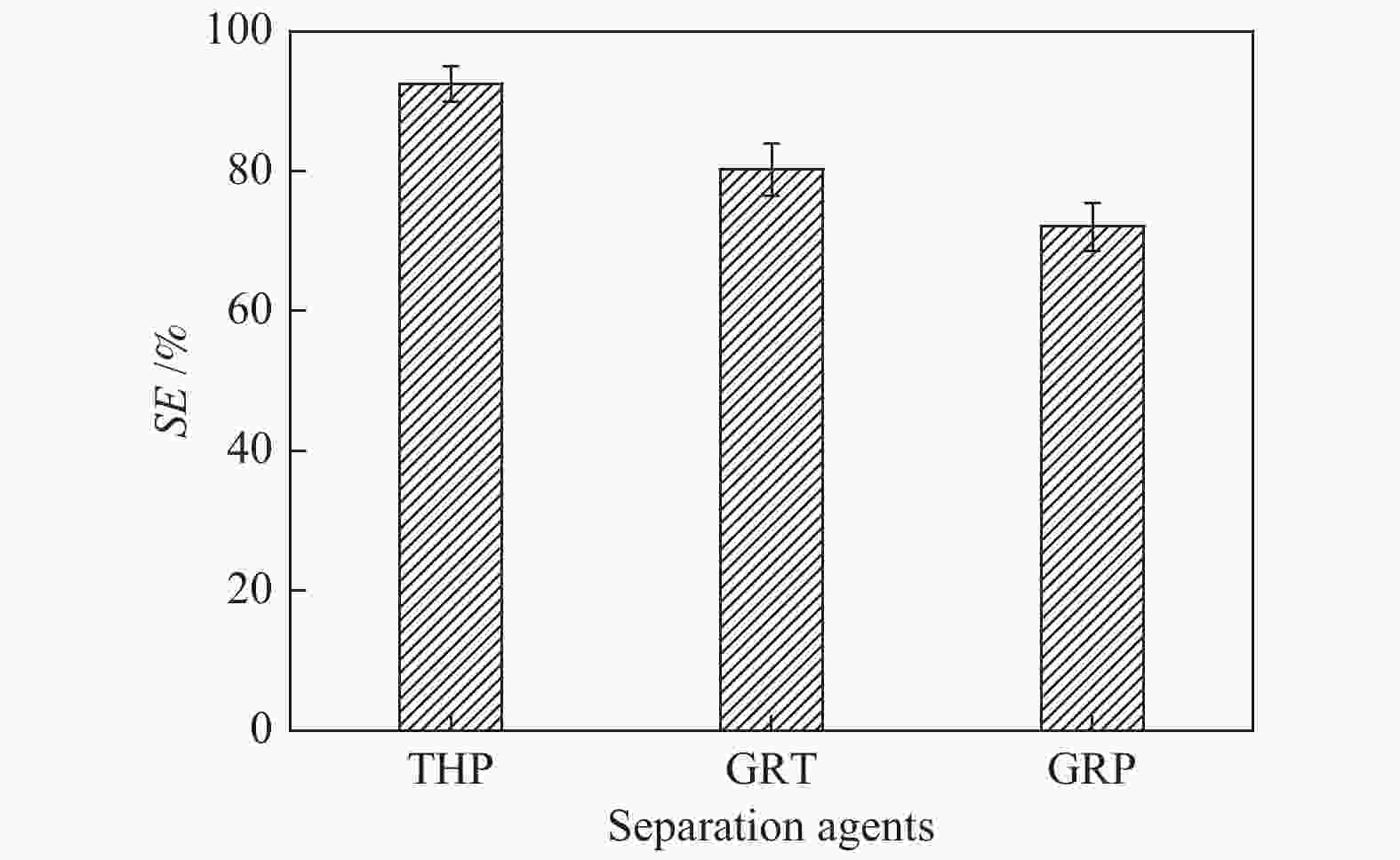
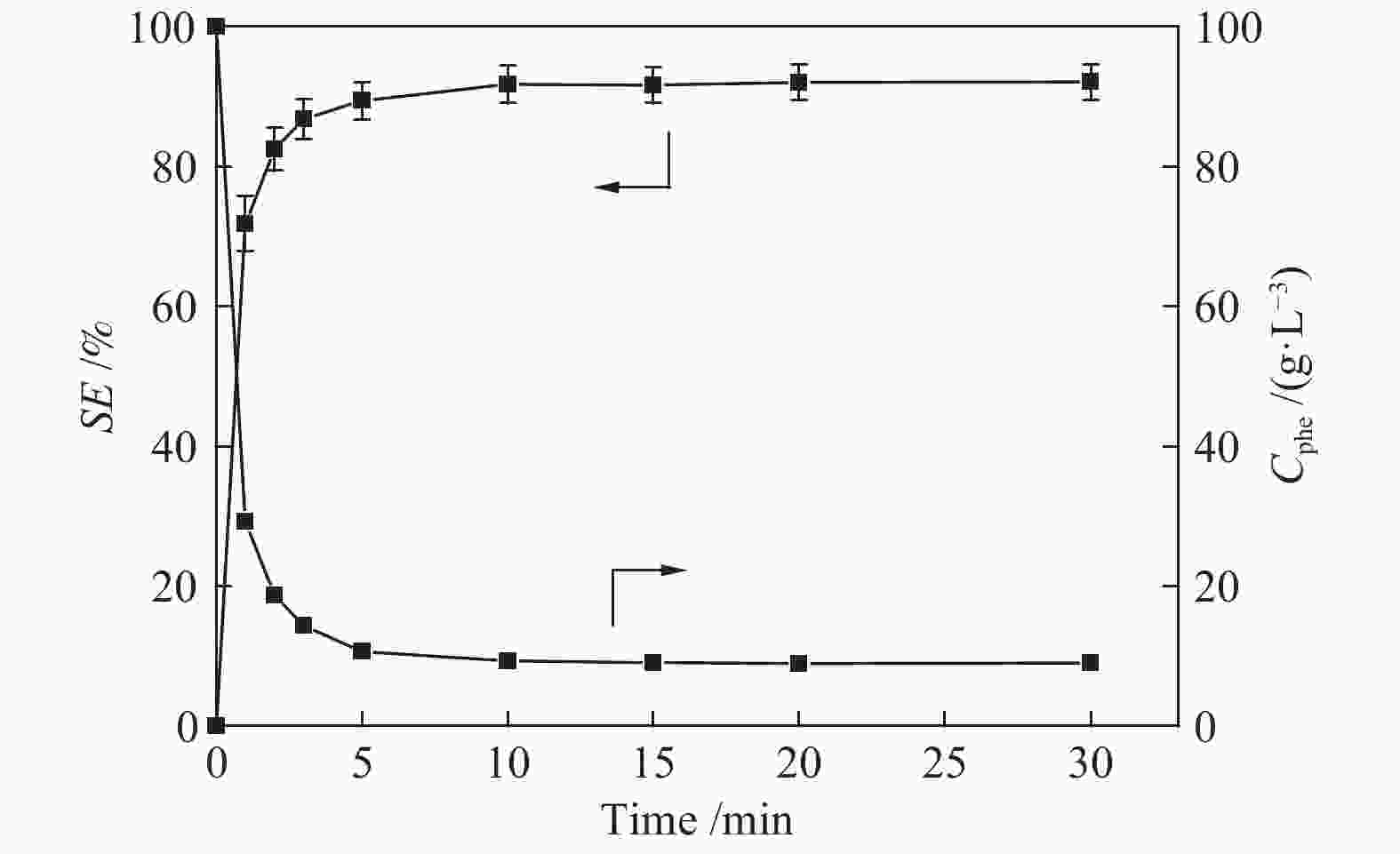
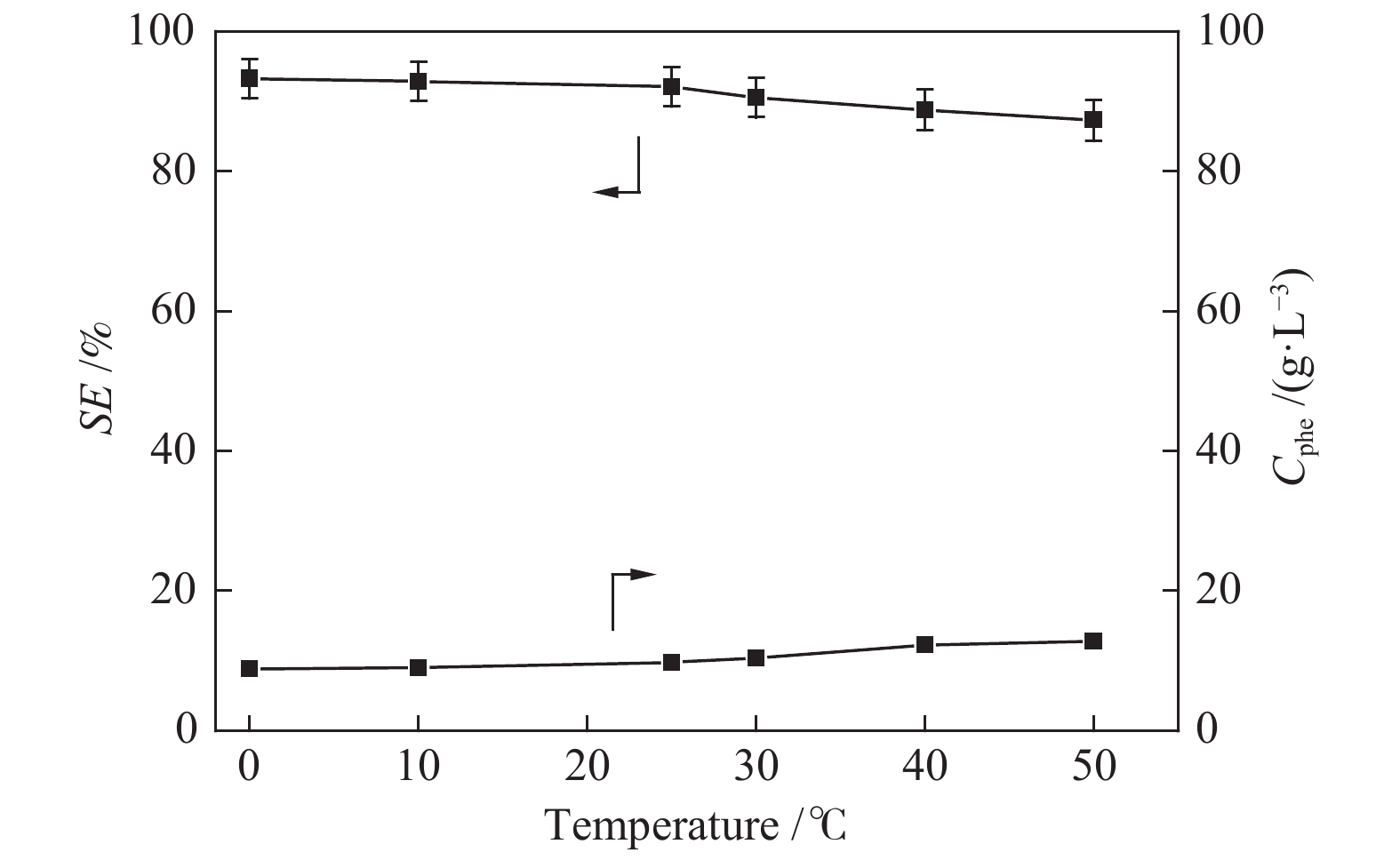
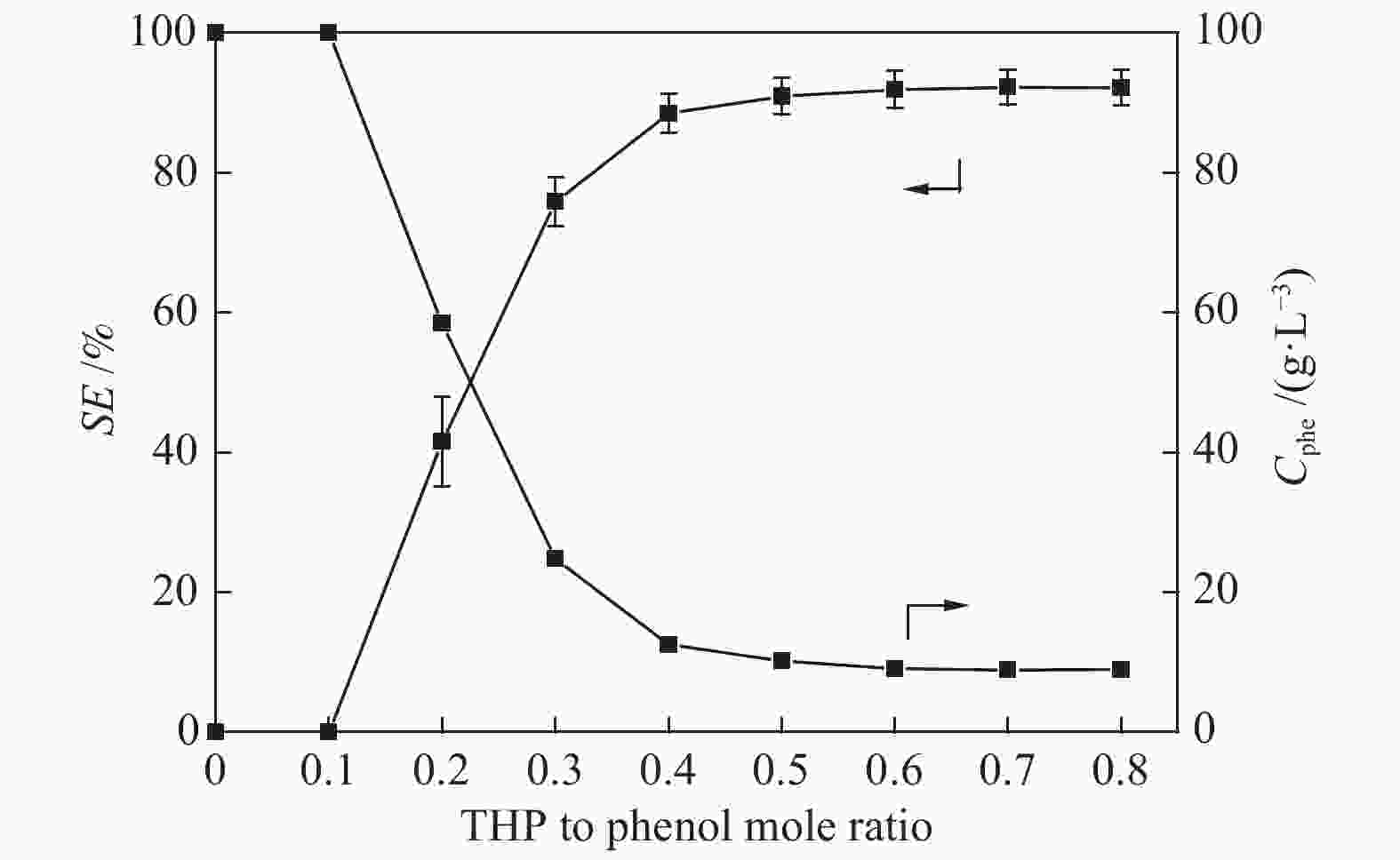
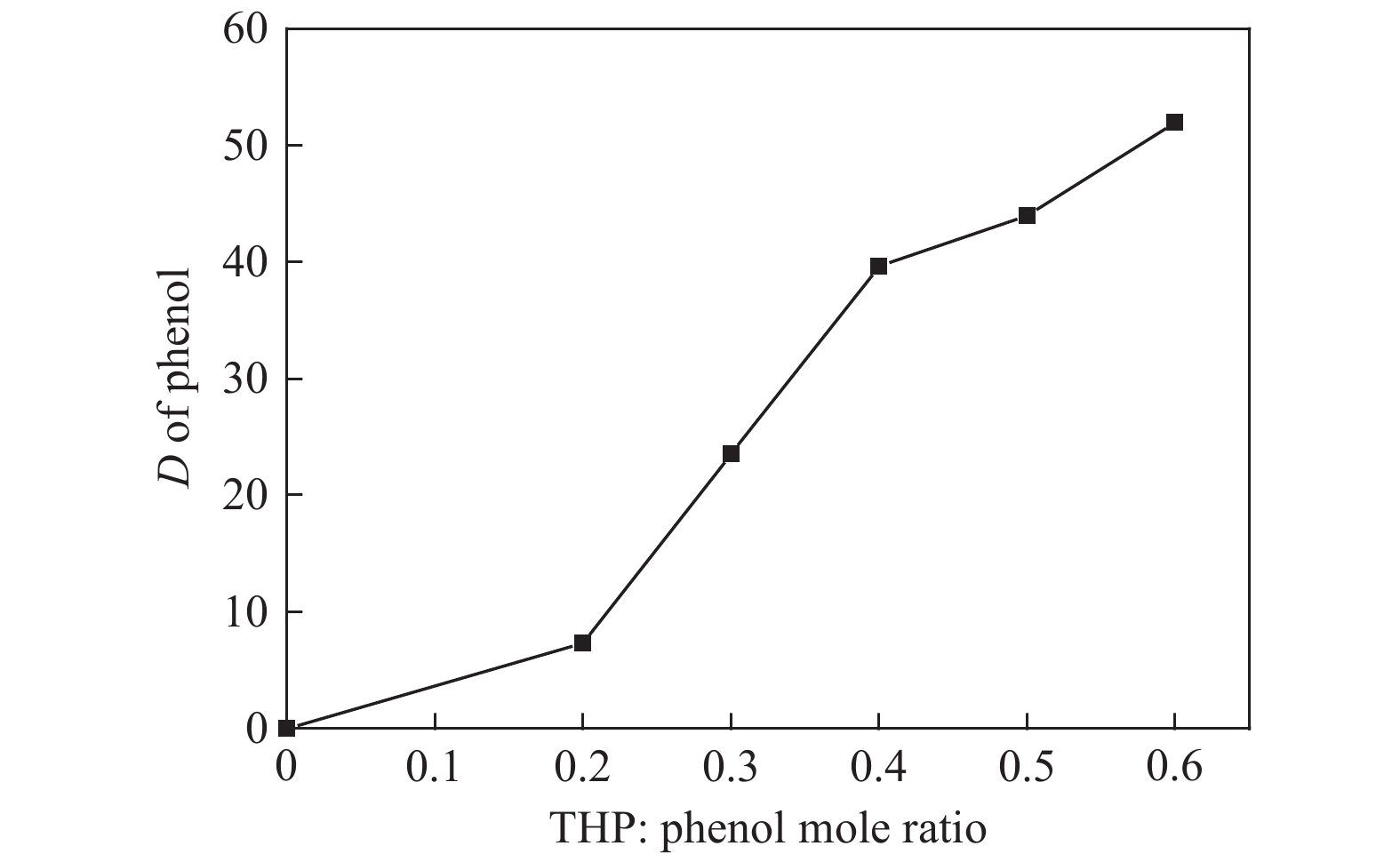
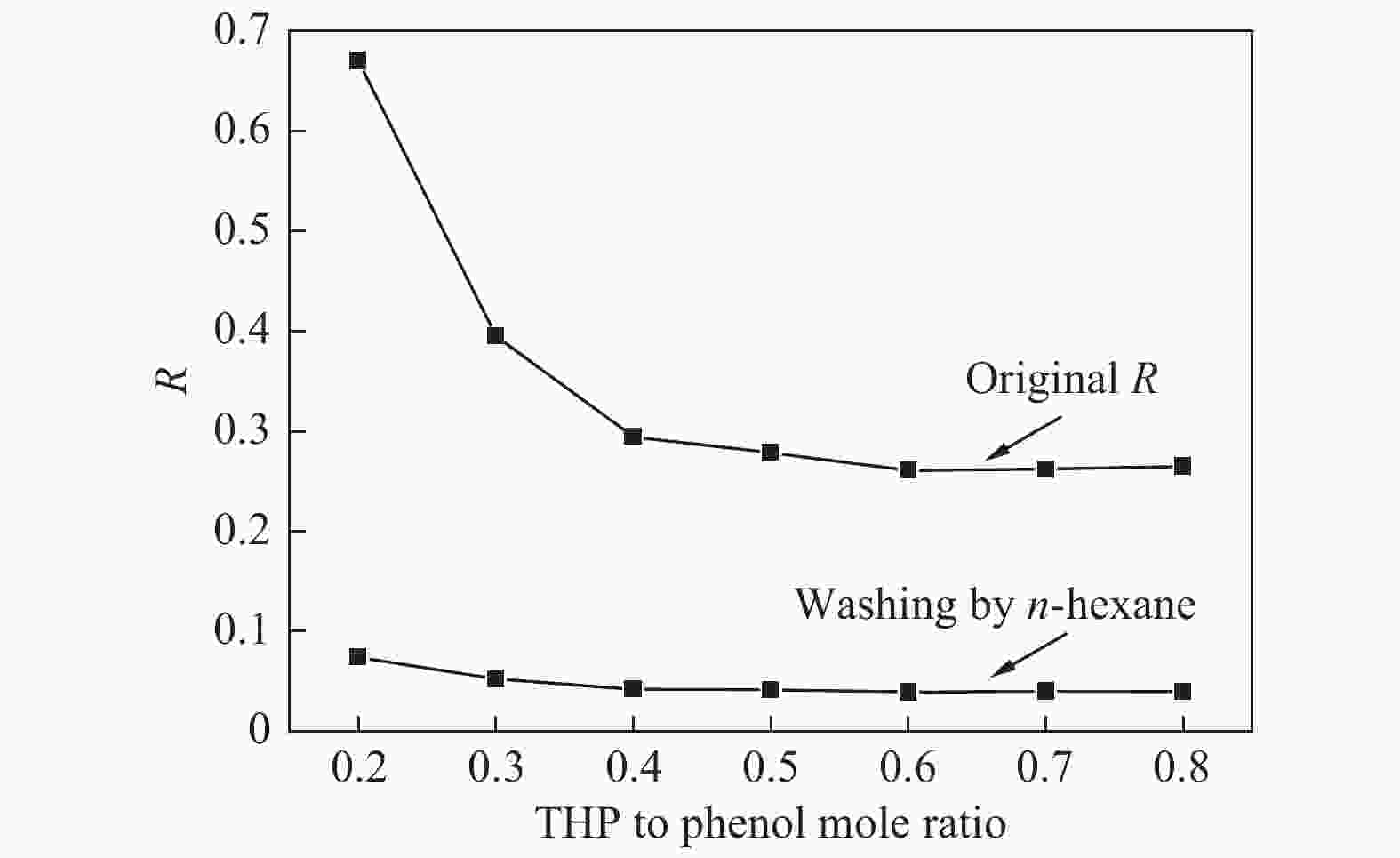
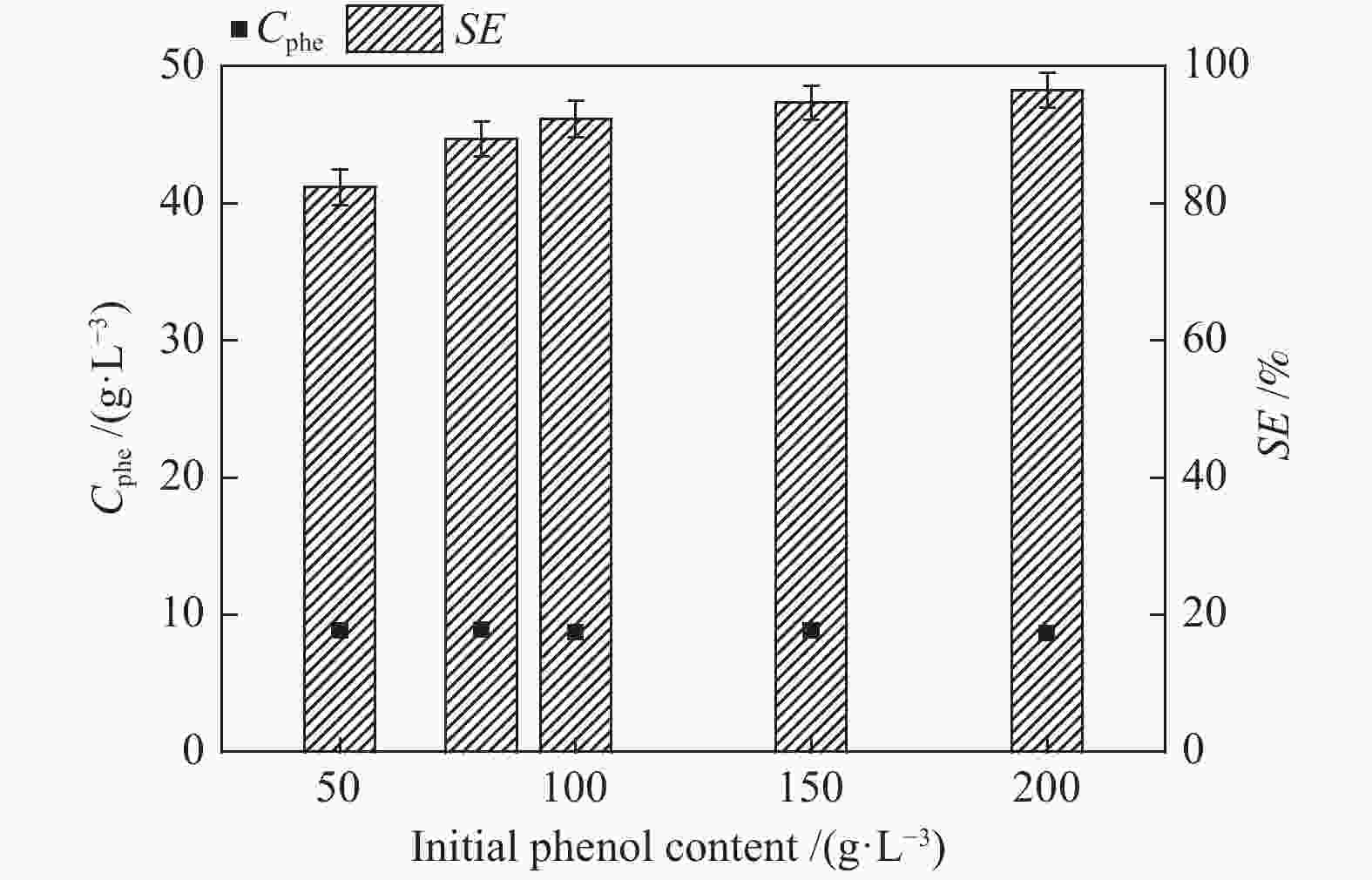
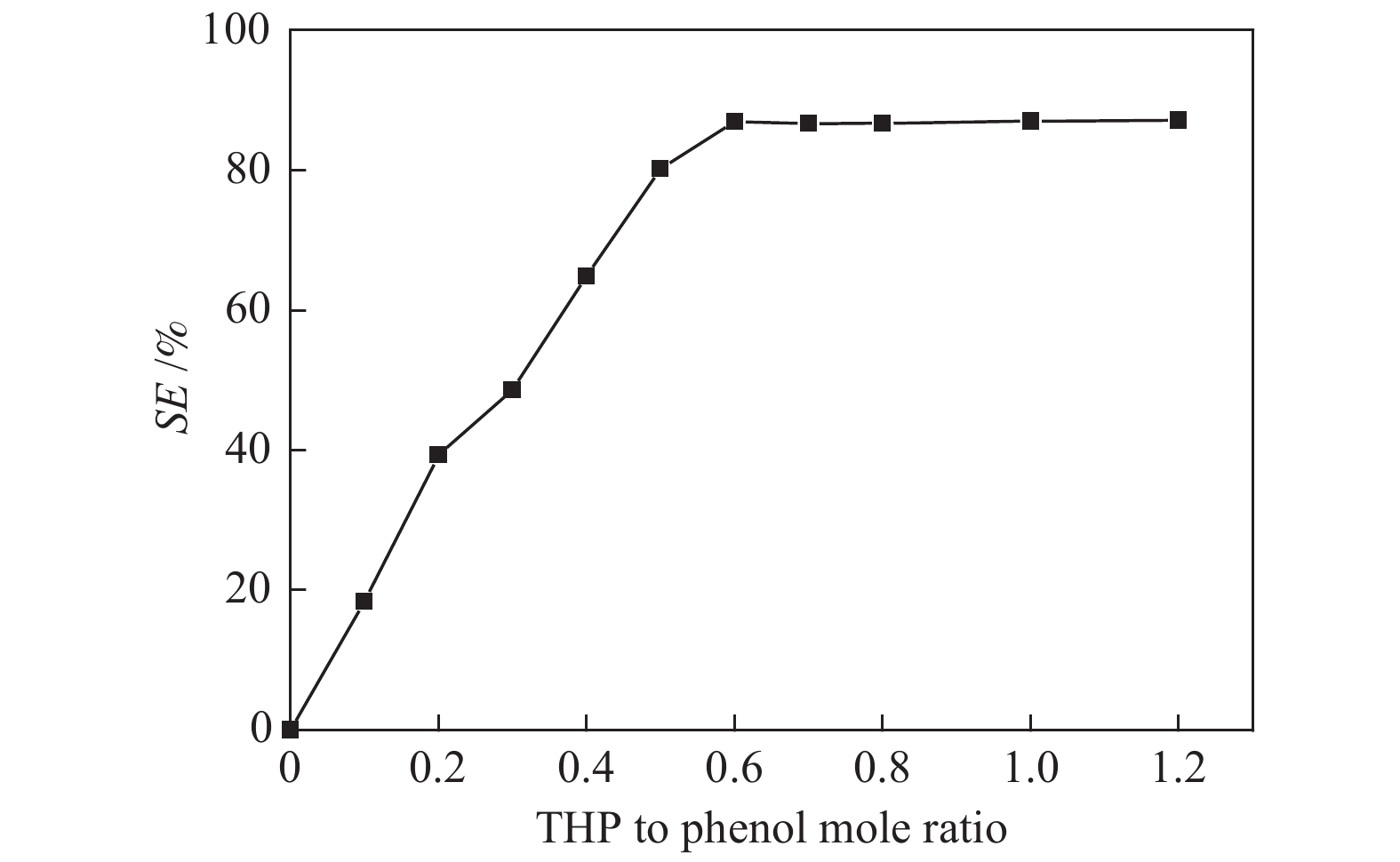
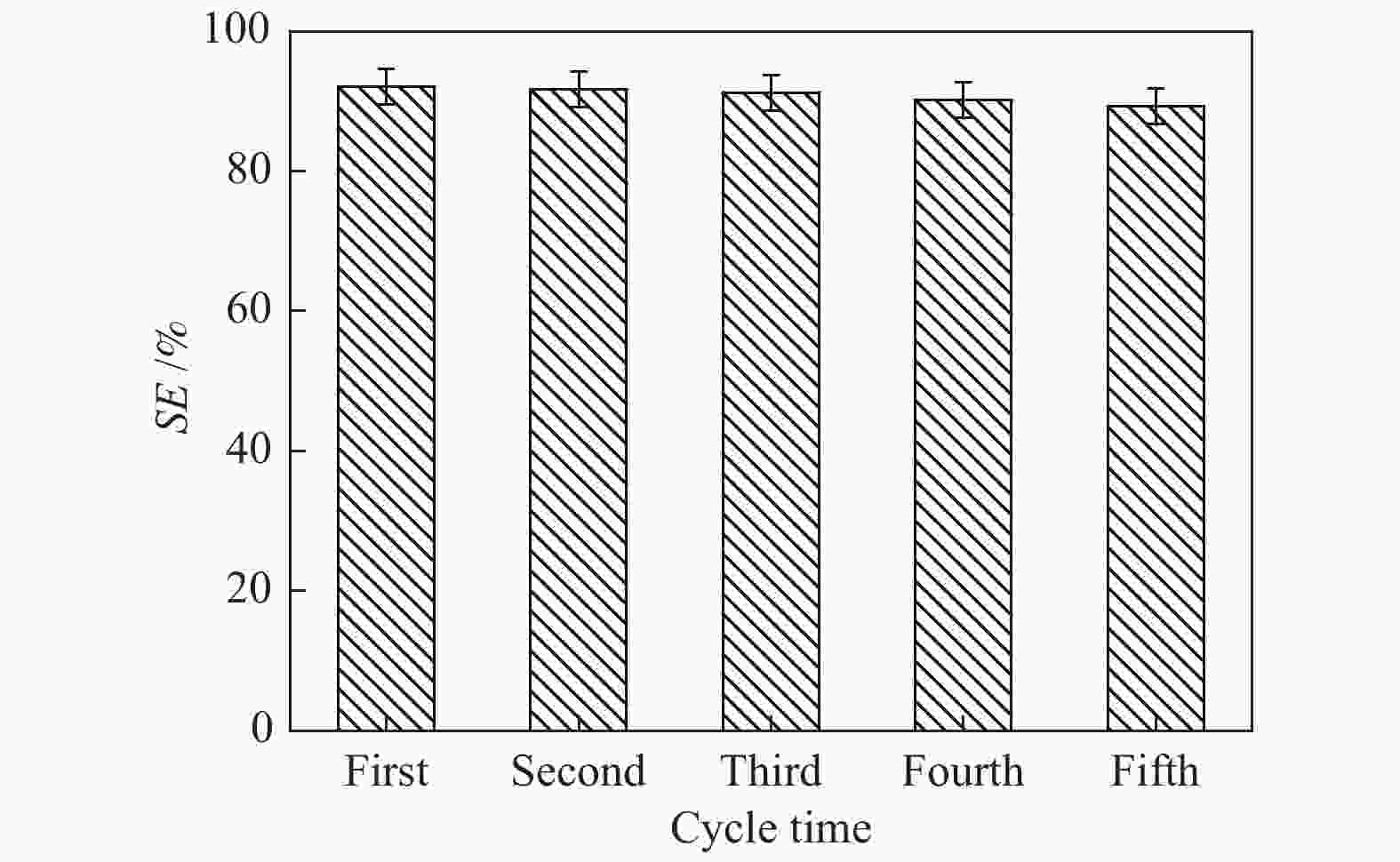
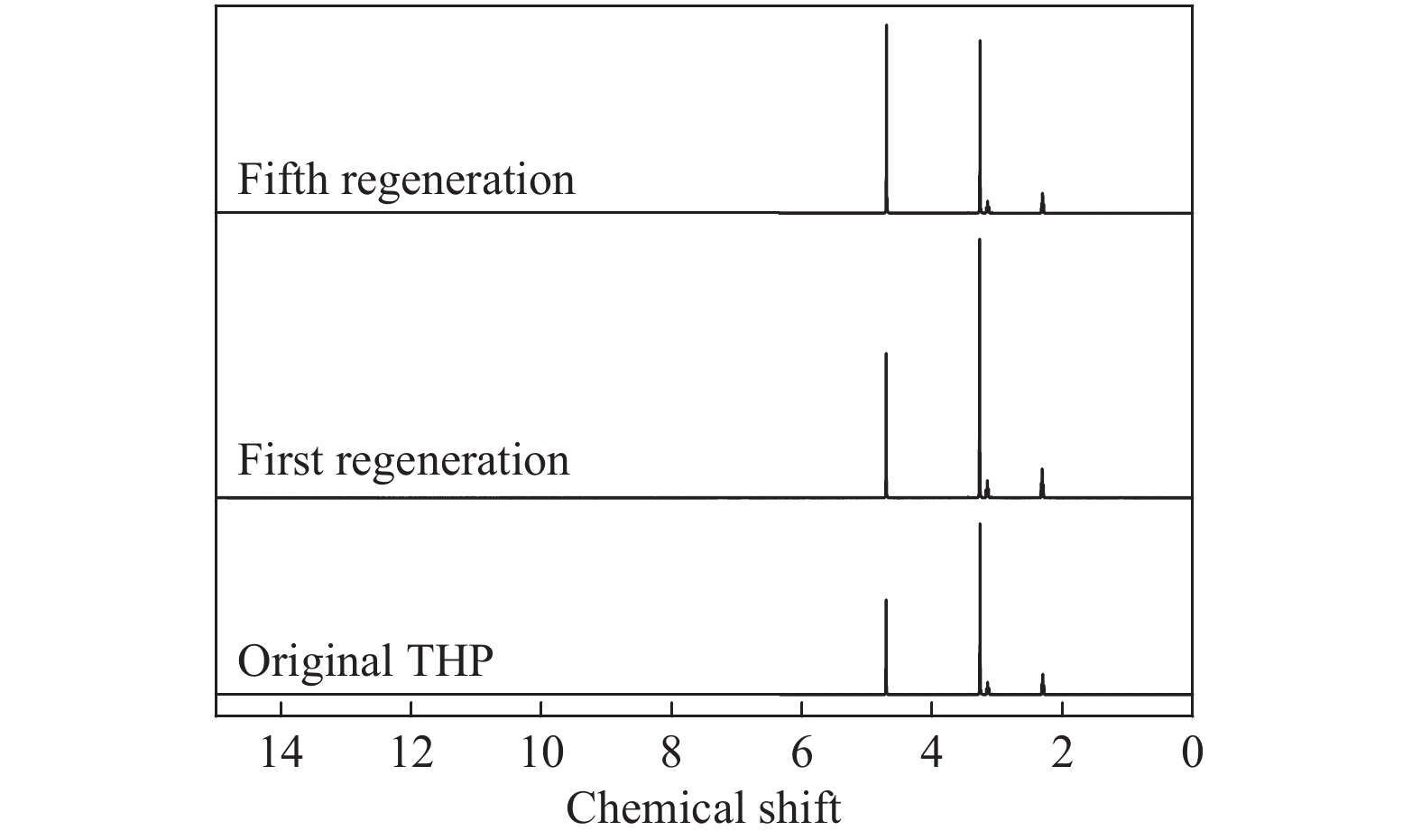
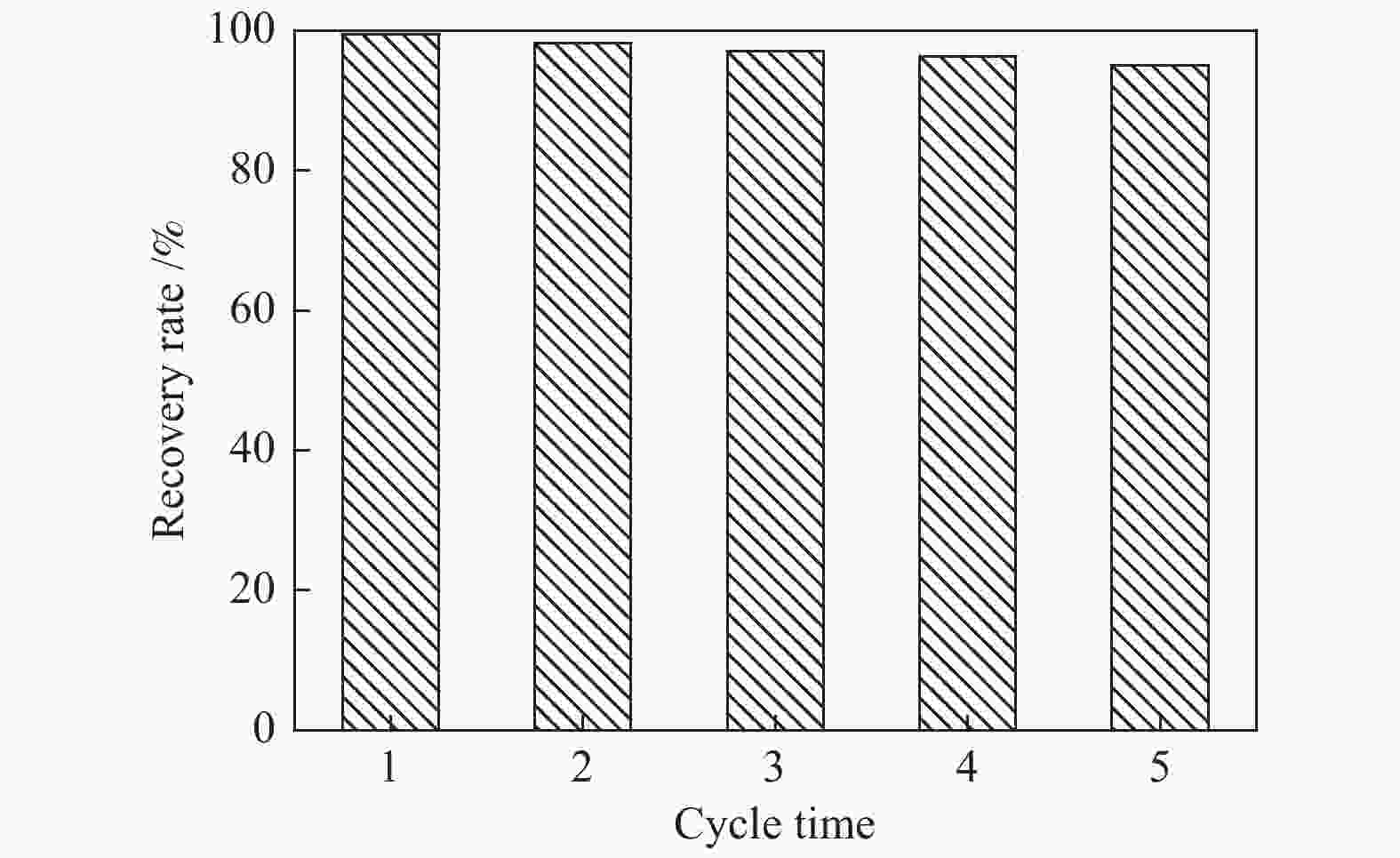
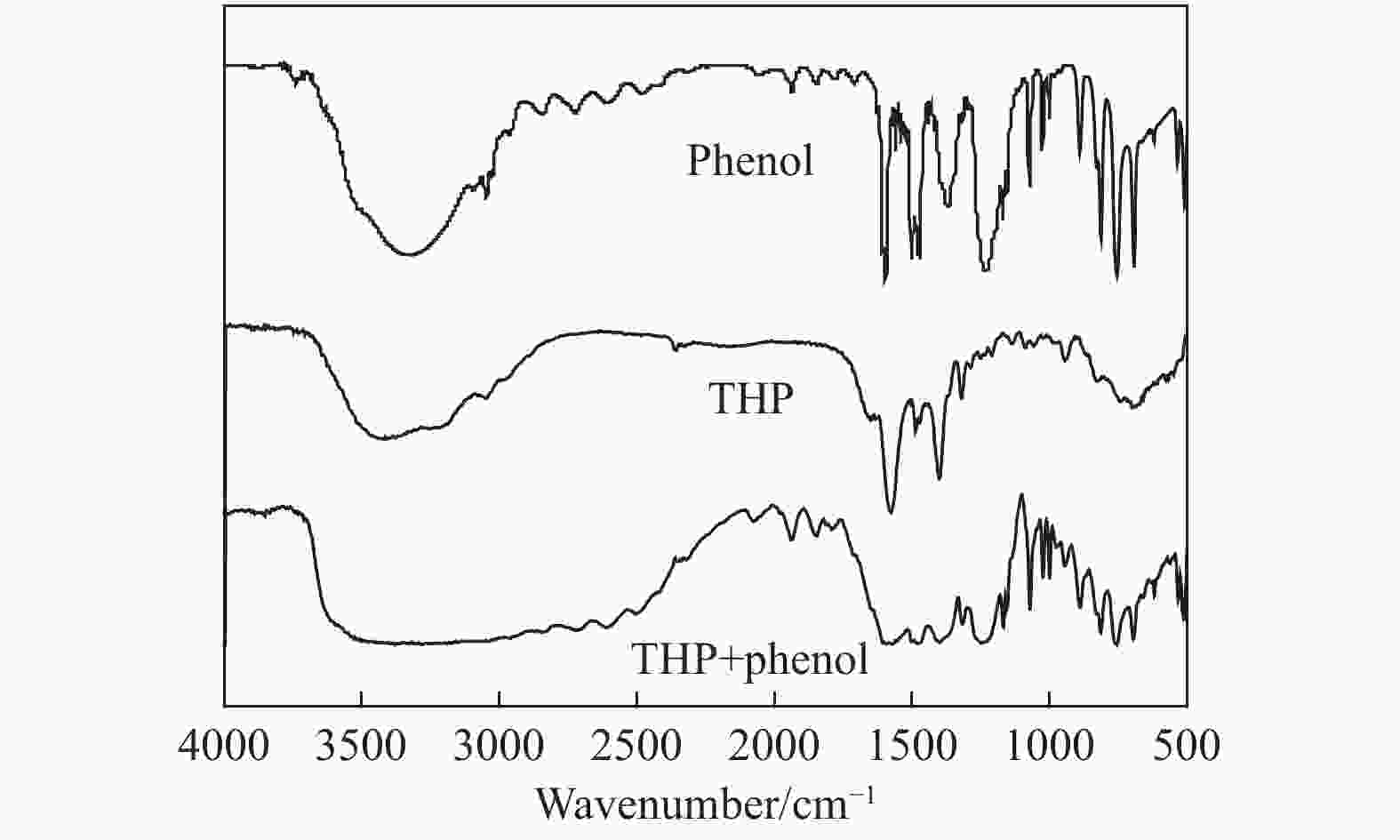
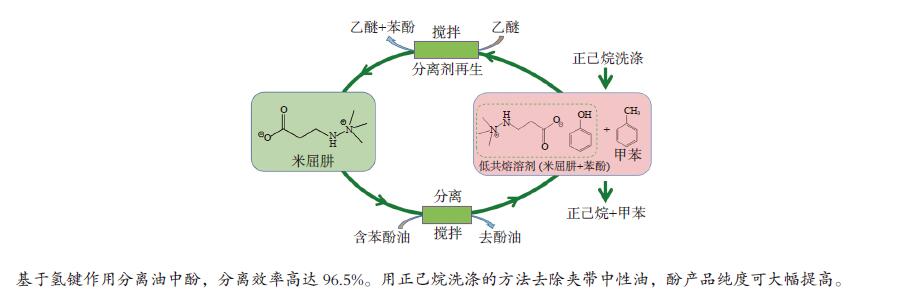
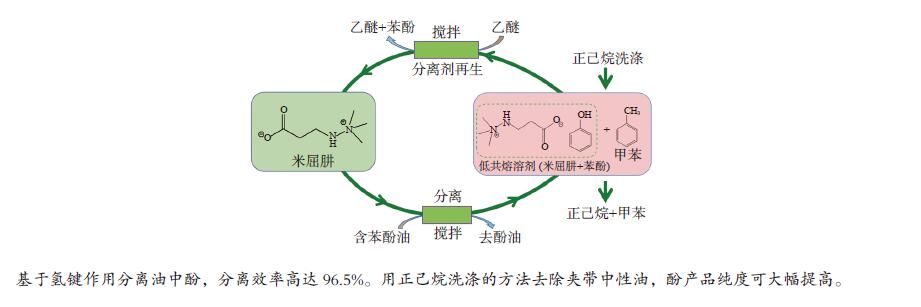
摘要: 本文采用米屈肼(THP)、吉拉尔特试剂T、吉拉德试剂P基于氢键作用分离油中的苯酚。结果发现,三种分离剂均可与苯酚形成低共熔溶剂(DES)而分离苯酚,其中米屈肼对苯酚的分离效率最高,可达96.5%;油中残余苯酚浓度低至1.3 g/L;10 min即可完成分离;苯酚的初始浓度不影响分离后油中最低残余苯酚浓度(约8.8 g/L)。采用正己烷洗涤的方法去除DES中夹带的中性油。结果发现,DES中的中性油与苯酚质量比降至0.04,说明酚产品纯度将大幅提高。米屈肼可再生并重复使用五次,性质不变。FT-IR表征发现米屈肼与苯酚间存在氢键作用。Abstract: In this work, the separation of phenol from oil mixtures was proposed by using mildronate (THP), Girard's Reagent T, and Girard's Reagent P. The results showed that the mentioned separation agents could form deep eutectic solvent (DES) with phenol and then separate phenol. The highest separation efficiency of phenol was 96.5% by THP, and the residual phenol content in oil was as low as 1.3 g/L; the separation could be finished in 10 min; the initial phenol content had no effect on the minimum residual phenol content (about 8.8 g/L) in oil after separation. An n-hexane washing method was used to remove the neutral oil entrained in DES. The results showed that the mass ratio of neutral oil to phenol in DES was reduced to 0.04, indicated that the purity of phenol product was greatly improved. THP was renewable and its properties remained unchanged after be reused for 5 times. Finally, FT-IR spectra showed that hydrogen bonds were formed between THP and phenol.
-
Key words:
- phenolic compounds /
- separation /
- deep eutectic solvent /
- neutral oil
-
表 1 实验试剂一览表
Table 1 Chemicals used in this work
Chemical CAS number Specification Supplier Phenol 108-95-2 98% Aladdin Chemical Co., Ltd., Shanghai, China Toluene 108-88-3 99% Beijing Tongguang Fine Chemicals Co., Ltd., Beijing, China n-hexane 110-54-3 97% Beijing Tongguang Fine Chemicals Co., Ltd., Beijing, China Ether 60-29-7 98% Beijing Tongguang Fine Chemicals Co., Ltd., Beijing, China Girard's reagent T 123-46-6 98% Aladdin Chemical Co., Ltd., Shanghai, China Girard's reagent P 1126-58-5 98% Aladdin Chemical Co., Ltd., Shanghai, China THP 76144-81-5 98% Aladdin Chemical Co., Ltd., Shanghai, China D2O 7789-20-0 99.9% Saen Chemical Technology Co., Ltd., Shanghai, China -
[1] WILLIAM M, MARKS S C, ALAN C. Evaluation of phenolic compounds in commercial fruit juices and fruit drinks[J]. J Agr Food Chem,2007,55(8):3148−3157. doi: 10.1021/jf062970x [2] SCHOBERT H H, SONG C. Chemicals and materials from coal in the 21st century[J]. Fuel,2002,81(1):15−32. doi: 10.1016/S0016-2361(00)00203-9 [3] 张可琢, 余雅黔, 唐瑞, 郑宇琦, 高家俊, 姜兴茂. 聚乙烯吡咯烷酮对油中酚类物质的吸附性能[J]. 燃料化学学报,2019,47(11):1305−1312. doi: 10.3969/j.issn.0253-2409.2019.11.004ZHANG Ke-zhuo, YU Ya-qian, TANG Rui, ZHENG Yu-qi, GAO Jia-jun, JIANG Xing-mao. Adsorption performance of polyvinylpyrrolidone for phenols in oil[J]. J Fuel Chem Technol,2019,47(11):1305−1312. doi: 10.3969/j.issn.0253-2409.2019.11.004 [4] 邓晶晶, 罗泽军, 王储, 朱锡锋. 疏水性离子液体对生物油水相馏分中酚类物质的萃取研究[J]. 燃料化学学报, doi: 10.1016/S1872-5813(21)60108-1.DENG Jing-jing, LUO Ze-jun, WANG Chu, ZHU Xi-feng. Study on the extraction of phenols from bio-oil aqueous fraction by hydrophobic ionic liquids[J]. J Fuel Chem Technol, doi: 10.1016/S1872-5813(21)60108-1. [5] 薛怡凡, 李梦瑶, 冯摇杰, 樊文俊, 李文英. 煤直接液化油中混合酚的分离研究[J]. 燃料化学学报,2019,47(11):1298−1304. doi: 10.3969/j.issn.0253-2409.2019.11.003XUE Yi-fan, LI Meng-yao, FENG Jie, FAN Wen-jun, LI Wen-ying. Separation of mixed phenolic compounds from direct coal liquefaction[J]. J Fuel Chem Technol,2019,47(11):1298−1304. doi: 10.3969/j.issn.0253-2409.2019.11.003 [6] 焦甜甜. 煤低温热解油高效分离及系统集成[D]. 北京: 中国科学院大学, 2016.JIAO Tian-tian. Efficient separation of low temperature coal pyrolysis oil and system integration[D]. Beijing: University of Chinese Academy of Sciences, 2016. [7] 任洪凯, 邓文安, 李传, 崔文龙. 中/低温煤焦油酚类化合物的组成研究[J]. 煤炭转化,2013,36(2):67−70. doi: 10.3969/j.issn.1004-4248.2013.02.017REN Hong-kai, DENG Wen-an, LI Chuan, CUI Wen-long. Study on the composition of phenolic compounds in middle /low temperature coal tar[J]. Coal Convers,2013,36(2):67−70. doi: 10.3969/j.issn.1004-4248.2013.02.017 [8] ZHANG G H, SUN H J. Production and market analysis of phenol in domestic[J]. Chem Ind,2014,32(10):33−35. [9] 王汝成, 孙鸣, 刘巧霞, 马燕星, 冯光, 徐龙, 马晓迅. 陕北中低温煤焦油中酚类化合物的提取与GC/MS分析[J]. 煤炭学报,2011,36(4):664−669.WANG Ru-cheng, SUN Ming, LIU Qiao-xia, MA Yan-xing, FENG Guang, XU Long, MA Xiao-xun. Extraction and GC/MS analysis of phenolic compounds in low temperature coal tar from Northern Shaanxi[J]. J China Coal Soc,2011,36(4):664−669. [10] APARICIO S, ATILHAN M, KARADAS F. Thermophysical properties of pure ionic liquids: Review of present situation[J]. Ind Eng Chem Res,2016,49(20):9580−9595. [11] ZHANG Q H, ZHANG S Q, DENG Y Q. Recent advances in ionic liquid catalysis[J]. Green Chem,2011,13:2619−2637. doi: 10.1039/c1gc15334j [12] GUO L P, WANG C M, LUO X Y, CUI G K, LI H R. Probing catalytic activity of halide salts by electrical conductivity in the coupling reaction of CO2 and propylene oxide[J]. Chem Commun,2010,46(32):5960−5962. doi: 10.1039/c0cc00584c [13] HOU Y C, REN Y H, PENG W, REN S H, WU W Z. Separation of phenols from oil using imidazolium-based ionic liquids[J]. Ind Eng Chem Res,2013,52(50):18071−18075. doi: 10.1021/ie403849g [14] 侯玉翠, 彭威, 杨春梅, 李省云, 吴卫泽. 咪唑基离子液体萃取分离模拟油酚混合物[J]. 化工学报,2013,64(S1):118−123.HOU Yu-cui, PENG Wei, YANG Chun-mei, LI Sheng-yun, WU Wei-ze. Extraction of phenolic compounds from simulated oil with imidazolium based ionic liquids[J]. J Chem Ind Eng (China),2013,64(S1):118−123. [15] 熊佳丽, 孟洪, 陆颖舟, 李春喜. 对二氯苄-聚乙烯咪唑型聚合离子液体的制备及对煤焦油中苯酚的吸附性能[J]. 高等学校化学学报,2014,35(9):2031−2036. doi: 10.7503/cjcu20140214XIONG Jia-li, MENG Hong, LU Ying-zhou, LI Chun-xi. Synthesis of 1,4-bis(chloromethyl)benzene crosslinked poly(vinylimidazolium) and its adsorptivity for phenol from coal tar[J]. Chem J Chin Univ,2014,35(9):2031−2036. doi: 10.7503/cjcu20140214 [16] JI Y A, HOU Y C, REN S H, YAO C F, WU W Z. Highly efficient separation of phenolic compounds from oil mixtures by imidazolium-based dicationic ionic liquids via forming deep eutectic solvents[J]. Energy Fuels,2017,31(9):10274−10282. doi: 10.1021/acs.energyfuels.7b01793 [17] JI Y A, HOU Y C, REN S H, YAO C F, WU W Z. Highly efficient extraction of phenolic compounds from oil mixtures by trimethylamine-based dicationic ionic liquids via forming deep eutectic solvents[J]. Fuel Process Technol,2018,171:183−191. doi: 10.1016/j.fuproc.2017.11.015 [18] 韦露, 樊友军. 低共熔溶剂及其应用研究进展[J]. 化学通报,2011,74(4):333−339.WEI Lu, FAN You-jun. Progress of deep eutectic solvents and their applications[J]. Chem Bull,2011,74(4):333−339. [19] ABBOTT A P, CAPPER G, DAVIES D L, RASHEED R K, TAMBYRAJAH V. Novel solvent properties of choline chloride/urea mixtures[J]. Chem Commun,2003,1(1):70−71. [20] ABBOTT A P, CAPPER G, MCKENZIE K J, RYDER K S. Electrodeposition of zinc–tin alloys from deep eutectic solvents based on choline chloride[J]. J Electroanal Chem,2007,599(2):4269−4277. [21] ANDREW P A, KHALID E T, GERO F, KATY J M, RYDER K S. Electrodeposition of copper composites from deep eutectic solvents based on choline chloride[J]. Phys Chem Chem Phys,2009,11(21):4269−4277. doi: 10.1039/b817881j [22] 李佳慧, 胡嘉, 赵荣祥, 乔海燕, 李秀萍. 氯化胆碱/草酸型低共熔溶剂氧化脱除模拟油硫化物[J]. 燃料化学学报,2014,42(7):870−876.LI Jia-hui, HU Jia, ZHAO Rong-xiang, QIAO Hai-yan, LI Xiu-ping. Oxidative desulfurization of model oil with choline chloride/oxalic acid as a eutectic solvent[J]. J Fuel Chem Technol,2014,42(7):870−876. [23] GUO W J, HOU Y C, REN S H, TIAN S D, WU W Z. Formation of deep eutectic solvents by phenols and choline chloride and their physical properties[J]. J Chem Eng Data,2013,58(4):866−872. doi: 10.1021/je300997v [24] GUO W J, HOU Y C, WU W Z, REN S H, TIAN S D, MARSH K N. Separation of phenol from model oils with quaternary ammonium salts via forming deep eutectic solvents[J]. Green Chem,2012,15(1):226−229. [25] PANG K, HOU Y C, WU W Z, GUO W J, PENG W, MARSH K N. Efficient separation of phenols from oils via forming deep eutectic solvents[J]. Green Chem,2012,14(9):2398−2401. doi: 10.1039/c2gc35400d [26] ZHANG Y, LI Z Y, WANG H Y, XUAN X P, WANG J J. Efficient separation of phenolic compounds from model oil by the formation of choline derivative-based deep eutectic solvents[J]. Sep Purif Technol,2016,163:310−318. doi: 10.1016/j.seppur.2016.03.014 [27] JIAO T T, ZHUANG X L, HE H Y, LI C S, CHEN H N, ZHANG S J. Separation of phenolic compounds from coal tar via liquid–liquid extraction using amide compounds[J]. Ind Eng Chem Res,2015,54(9):2573−2579. doi: 10.1021/ie504892g [28] JIAO T T, LI C S, ZHUANG X L, CAO S S, CHEN H N, ZHANG S J. The new liquid–liquid extraction method for separation of phenolic compounds from coal tar[J]. Chem Eng J,2015,266:148−155. doi: 10.1016/j.cej.2014.12.071 [29] YAO C F, HOU Y C, REN S H, WU W Z, ZHANG K, JI Y A, LIU H. Efficient separation of phenol from model oils using environmentally benign quaternary ammonium-based zwitterions via forming deep eutectic solvents[J]. Chem Eng J,2017,620−626. [30] 王震, 侯玉翠, 任树行, 孔洁, 吴卫泽. 低共熔法萃取分离油酚混合物过程中油的夹带[J]. 化工学报,2015,66:247−252.WANG Zhen, HOU Yu-cui, REN Shu-hang, KONG Jie, WU Wei-ze. Oil entrainment during separating phenols from model oils via forming deep eutectic solvents[J]. CIESC J,2015,66:247−252. [31] JI Y A, HOU Y C, REN S H, YAO C F, WU W Z. Separation of phenolic compounds from oil mixtures using environmentally benign biological reagents based on brønsted acid-lewis base interaction[J]. Fuel,2019,239:926−934. doi: 10.1016/j.fuel.2018.11.007 [32] ASPRION N, HASSE H, MAURER G. FT-IR spectroscopic investigations of hydrogen bonding in alcohol–hydrocarbon solutions[J]. Fluid Phase Equilibr,2001,186(1/2):1−25. doi: 10.1016/S0378-3812(01)00363-6 [33] MAES G, SMETS J. Hydrogen bond cooperativity: A quantitative study using matrix-isolation FT-IR spectroscopy[J]. J Phys Chem,1993,97(9):1818−1825. doi: 10.1021/j100111a017 -




 下载:
下载: