Study on performance of Ag-modified layered copper silicate catalyst for hydrogenation of dimethyl oxalate to methyl glycolate
-
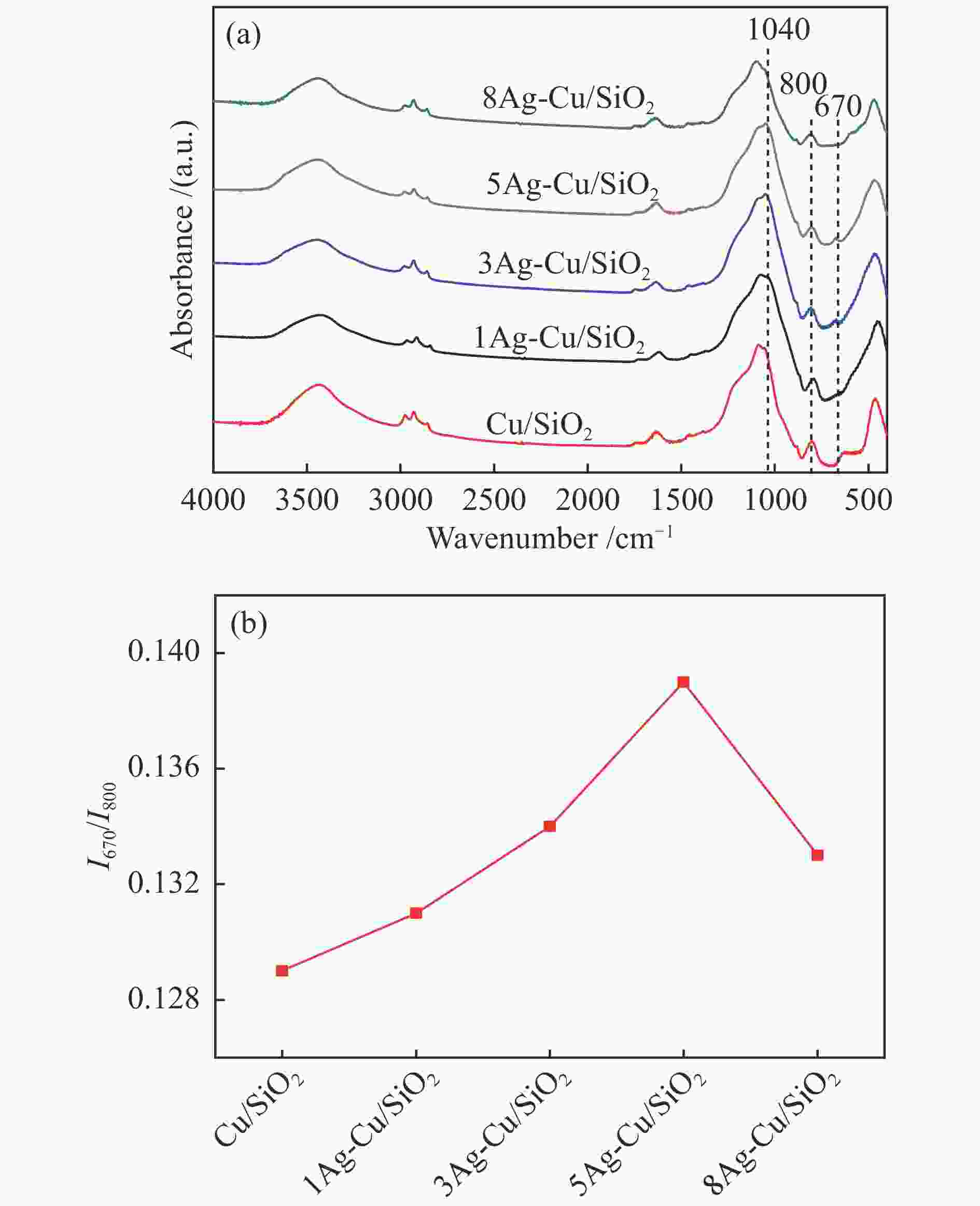
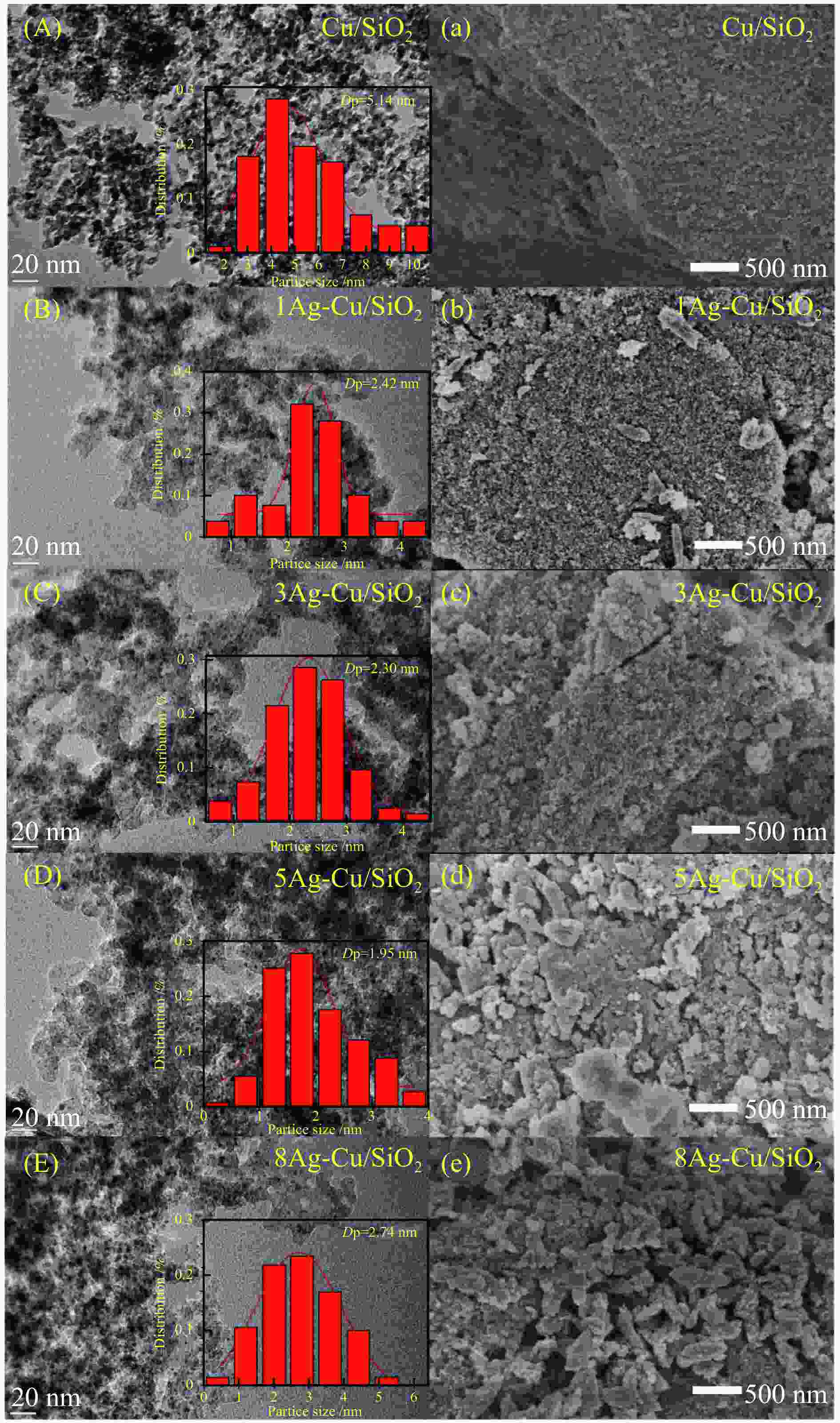
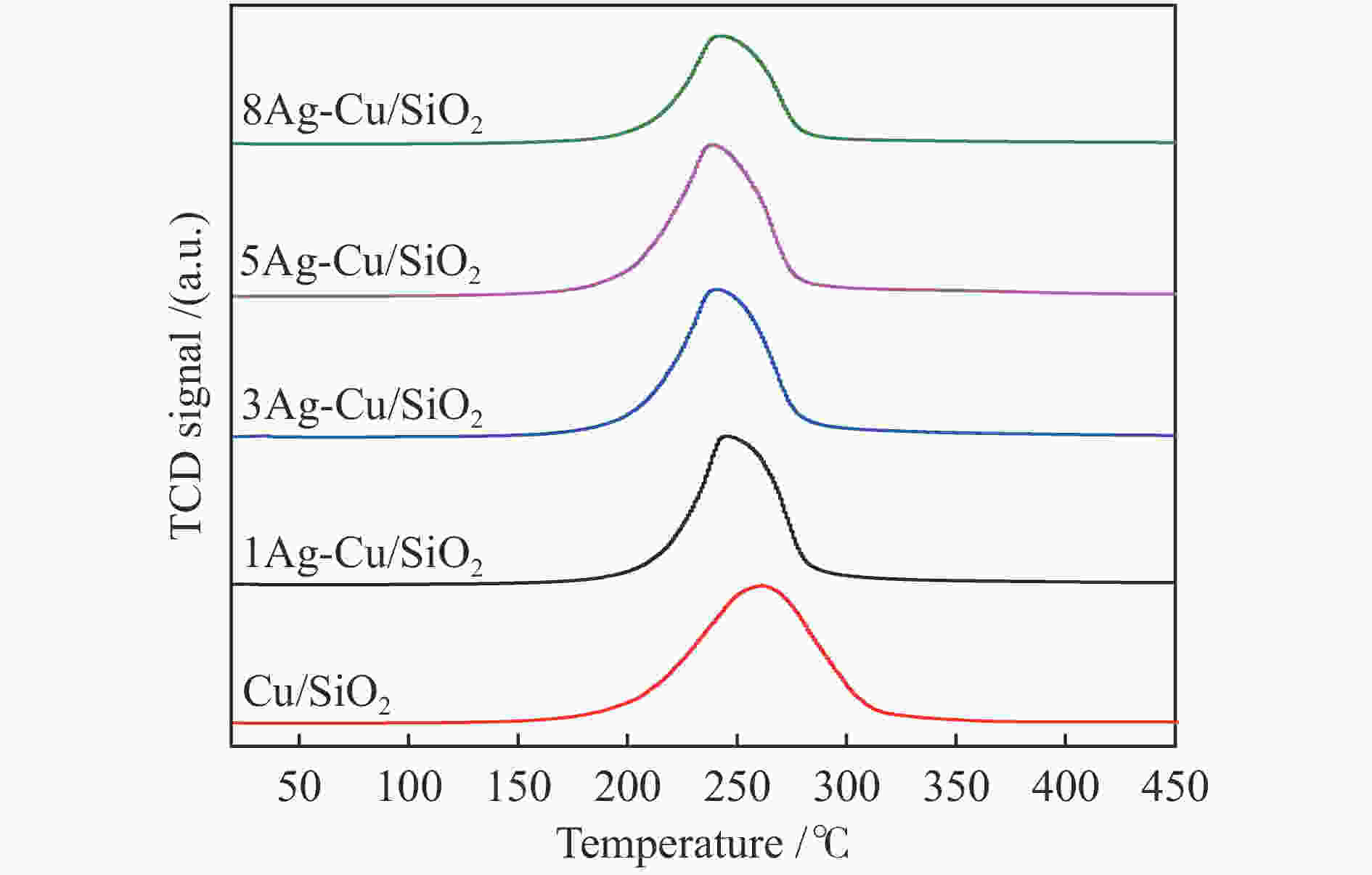
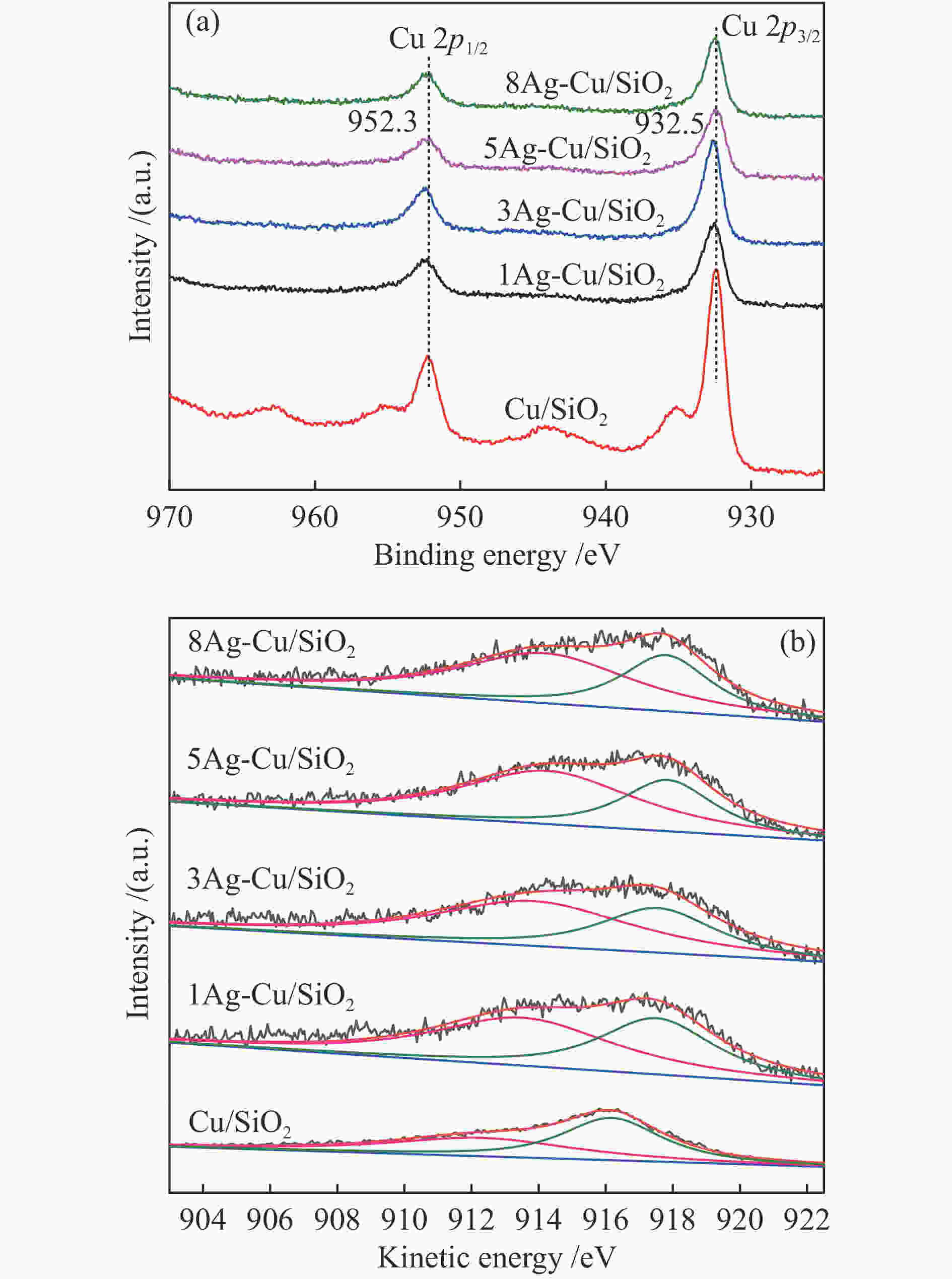
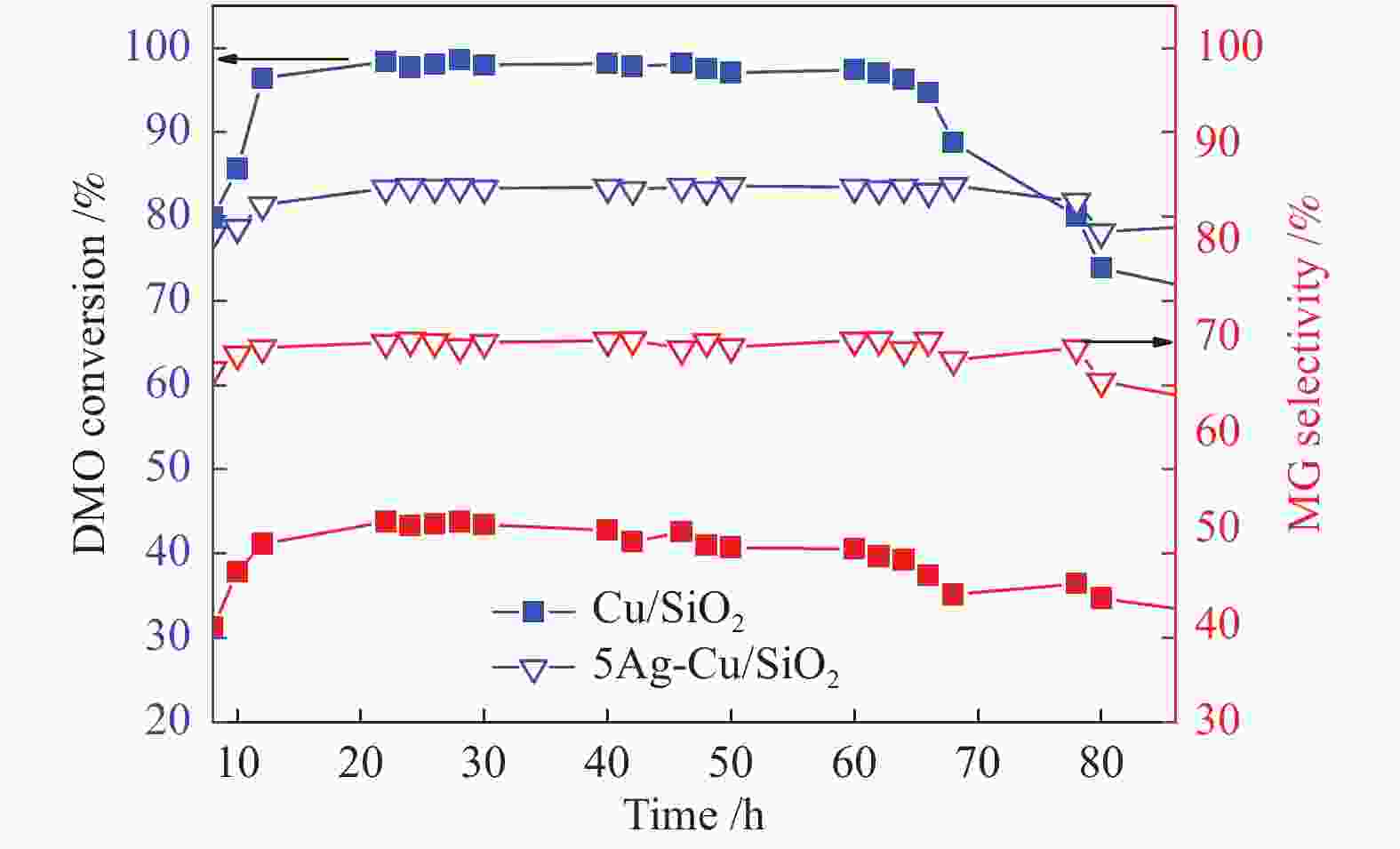
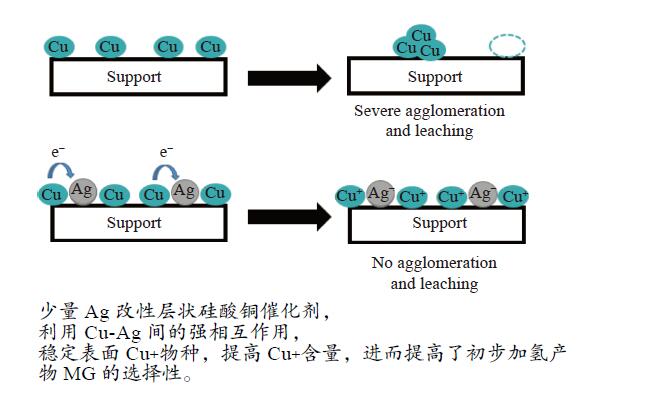
摘要: 本研究采用溶胶-凝胶法制备了一系列CuAg/SiO2催化剂用于草酸二甲酯(DMO)加氢合成MG,使用XRD、N2吸附-脱附、FT-IR、TEM、SEM、H2-TPR、XPS等方法对其结构进行表征,重点考察了Ag含量对催化剂结构及性能的影响。当Ag负载量为5%时,5Ag-Cu/SiO2催化剂活性最高,DMO的转化率和MG的选择性分别可达83.7%和72.2%。表征结果表明,适量Ag的引入可有效提高活性组分Cu的分散度,增加催化剂表面Cu+的含量,从而提高催化剂活性。此外,Ag与Cu之间的电子转移可以有效稳定Cu+,进而提高催化剂的稳定性。Abstract: Methyl glycolate (MG) is a high value-added chemical intermediate and widely used in the fields of medicine, chemical industry, fodder and dyes. A series of CuAg/SiO2 catalysts were prepared by sol-gel method for hydrogenation of dimethyl oxalate to MG. The structure of catalysts was characterized by XRD, N2-physical adsorption, FT-IR, TEM, H2-TPR, and XPS, and the influence of Ag loading on catalytic performance was investigated. The 5Ag-Cu/SiO2 catalyst with Ag loading of 5% exhibited the best catalytic performance with DMO conversion of 83.7% and MG selectivity of 72.2%. The characterization results showed that introducing appropriate amount of Ag not only improved the dispersion of copper species, but also increased the content of Cu+, thereby improving the catalytic activity of CuAg/SiO2 catalysts. In addition, the electron transfer between Ag and Cu could effectively stabilize Cu+, and eventually improved the stability of the catalyst.1) #: 共同第一作者
-
表 1 催化剂的物理化学和织构性质
Table 1 Physicochemical and textural properties of catalysts
Catalyst Cu loading
w/%aAg loading
w/%aCu dispersion/
%bCu surface/
(m2·g−1)bSBET/
(m2·g−1)cvtotal/
(cm3·g−1)dd/nme Cu/SiO2 28.6 0 14.7 99.4 313 0.58 7.14 1Ag-Cu/SiO2 28.2 0.5 25.3 171.2 235 0.79 12.4 3Ag-Cu/SiO2 27.6 1.7 25.9 175.2 240 0.76 10.73 5Ag-Cu/SiO2 27.1 2.0 27.2 184.0 222 0.71 12.6 8Ag-Cu/SiO2 26.4 2.3 23.7 160.3 226 0.73 13.11 Ag/SiO2 − 1.8 − − − − − a: Cu loading and Ag loading conducted by ICP; b: Cu dispersion determined by N2O surface oxidation; c: BET specific surface areas;
d: Total pore volume by BET; e: Average pore diameter was calculated from adsorption branch by BJH表 2 还原后不同焙烧温度催化剂表面铜物种占比
Table 2 Percentage of copper species on the surface of catalysts at different calcination temperatures after activation
Catalyst E/eV $X_{{\rm{Cu}}}^+$%a Cu+ Cu0 Cu/SiO2 912.3 916.2 44.8 1Ag-Cu/SiO2 913.5 917.5 57.2 3Ag-Cu/SiO2 913.8 917.5 63.1 5Ag-Cu/SiO2 914.2 917.8 66.3 8Ag-Cu/SiO2 914.1 917.8 64.7 a: ratio of Cu+ to (Cu++Cu0) obtained by deconvolution of Cu LMM spectra -
[1] HOLM M S, SARAVANAMURUGAN S, TAARNING E. Conversion of sugars to lactic acid derivatives using heterogeneous zeotype catalysts[J]. Science,2010,328(5978):602−605. doi: 10.1126/science.1183990 [2] BARRI S A I, CHADWICK D. Carbonylation of formaldehyde with zeolite catalysts[J]. Catal Lett,2011,141(6):749−753. doi: 10.1007/s10562-011-0616-0 [3] YADAV G D, GUPTA V R. Synthesis of glyoxalic acid from glyoxal[J]. Process Biochem,2000,36:73−78. doi: 10.1016/S0032-9592(00)00175-8 [4] ZHENG X L, LIN H Q, ZHENG J W, DUAN X P, YUAN Y Z. Lanthanum oxide-modified Cu/SiO2 as a high-performance catalyst for chemoselective hydrogenation of dimethyl oxalate to ethylene glycol[J]. ACS Catal,2013,3(12):2738−2749. doi: 10.1021/cs400574v [5] CHENG S, MAO D S, GUO X M, YU J. Synthesis of methyl glycolate from the hydrogenation of dimethyl oxalate on Ag/SiO2 catalyst: the effects of Ag contents and promoters[J]. React Kinet Mech Catal,2019,126(2):1067−1079. doi: 10.1007/s11144-018-01527-z [6] MATTEOLI U, MENCHI G, BIANCHI M, PIACENTI F, IANELLI S, NARDELLI M. Structure and catalytic activity of phosphine-substituted ruthenium carbonyl carboxylates[J]. J Organomet Chem,1995,498(2):177−186. doi: 10.1016/0022-328X(95)05518-T [7] ZHANG M J, YANG Y W, LI A T, YAO D W, GAO Y Q, FAYISA B A, WANG M Y, HUANG S Y, LV J, WANG Y, MA X B. Nanoflower-like Cu/SiO2 catalyst for hydrogenation of ethylene carbonate to methanol and ethylene glycol: Enriching H2 adsorption[J]. ChemCatChem,2020,12(14):3670−3678. doi: 10.1002/cctc.202000365 [8] YIN A Y, GUO X Y, DAI W L, FAN K G. High activity and selectivity of Ag/SiO2 catalyst for hydrogenation of dimethyl oxalate[J]. Chem Commun,2010,46(24):4348−4350. doi: 10.1039/c0cc00581a [9] GONG J L, YUE H R, ZHAO Y J, ZHAO S, ZHAO L, LV J, WANG S P, MA X B. Synthesis of ethanol via syngas on Cu/SiO2 catalysts with balanced Cu0-Cu+ sites[J]. J Am Chem Soc,2012,134(34):13922−13925. doi: 10.1021/ja3034153 [10] AN J W, WANG X H, ZHAO J X, JIANG S H, QUAN Y H, PEI Y L, WU M M, REN J. Density-functional theory study on hydrogenation of dimethyl oxalate to methyl glycolate over copper catalyst: Effect of copper valence state[J]. Mol Catal,2020,482:110667. doi: 10.1016/j.mcat.2019.110667 [11] LU X D, WANG G F, YANG Y, KONG X P, CHEN J G. A boron-doped carbon aerogel-supported Cu catalyst for the selective hydrogenation of dimethyl oxalate[J]. New J Chem,2020,44(8):3232−3240. doi: 10.1039/C9NJ05956C [12] SUN Y N, MA Q X, GE Q J, SUN J. Tunable synthesis of ethanol or methyl acetate via dimethyl oxalate hydrogenation on confined iron catalysts[J]. ACS Catal,2021,11(8):4908−4919. doi: 10.1021/acscatal.1c00339 [13] WANG J, SHI R N, HAO P P, SUN W, LIU S S, LI Z, REN J. Influence of oxygen-containing groups of activated carbon aerogels on copper/activated carbon aerogels catalyst and synthesis of dimethyl carbonate[J]. J Mater Sci,2018,53(3):1833−1850. doi: 10.1007/s10853-017-1639-8 [14] YE C L, GUO C L, ZANG J L. Highly active and stable CeO2-SiO2 supported Cu catalysts for the hydrogenation of methyl acetate to ethanol[J]. Fuel Process Technol,2016,143:219−224. doi: 10.1016/j.fuproc.2015.12.003 [15] BRANDS D S, POELS E K, BLIEK A. Ester hydrogenolysis over promoted Cu/SiO2 catalysts[J]. Appl Catal A: Gen,1999,184(2):279−289. doi: 10.1016/S0926-860X(99)00106-4 [16] KANDHASAMY N, RAMALINGAM G, MURUGADOSS G, RAJESH KUMAR M, MANIBALAN G, JOTHIRAMALINGAM R, YADAV H M. Copper and zinc oxide anchored silica microsphere: A superior pseudocapacitive positive electrode for aqueous supercapacitor applications[J]. J Alloy Compd,2021,888:161489. doi: 10.1016/j.jallcom.2021.161489 [17] YUE H R, ZHAO Y J, ZHAO L, LV J, WANG S P, GONG J L, MA X B. Hydrogenation of dimethyl oxalate to ethylene glycol on a Cu/SiO2/cordierite monolithic catalyst: Enhanced internal mass transfer and stability[J]. AIChE J,2012,58(9):2798−2809. doi: 10.1002/aic.12785 [18] ZHANG H W, TAN H R, JAENICKE S, CHUAH G K. Highly efficient and robust Cu catalyst for non-oxidative dehydrogenation of ethanol to acetaldehyde and hydrogen[J]. J Catal,2020,389:19−28. doi: 10.1016/j.jcat.2020.05.018 [19] ZHAO Y J, KONG L X, XU Y X, HUANG H J, YAO Y Q, ZHANG J W, WANG S P, MA X B. Deactivation mechanism of Cu/SiO2 catalysts in the synthesis of ethylene glycol via methyl glycolate hydrogenation[J]. Ind Eng Chem Res,2020,59(27):12381−12388. doi: 10.1021/acs.iecr.0c01619 [20] SUN J, YU J F, MA Q X, MENG F, WEI X X, SUN Y N, TSUBAKI N. Freezing copper as a noble metal–like catalyst for preliminary hydrogenation[J]. Sci Adv,2018,4(12):eaau3275. doi: 10.1126/sciadv.aau3275 [21] WANG M L, YAO D W, LI A T, YANG Y W, LV J, HUANG S Y, WANG Y, MA X B. Enhanced selectivity and stability of Cu/SiO2 catalysts for dimethyl oxalate hydrogenation to ethylene glycol by using silane coupling agents for surface modification[J]. Ind Eng Chem Res,2020,59(20):9414−9422. doi: 10.1021/acs.iecr.0c00789 [22] ZHAO Y J, LI S M, WANG Y, SHAN B, ZHANG J, WANG S P, MA X B. Efficient tuning of surface copper species of Cu/SiO2 catalyst for hydrogenation of dimethyl oxalate to ethylene glycol[J]. Chem Eng J,2017,313:759−768. doi: 10.1016/j.cej.2016.12.027 [23] SHU G Q, MA K, TANG S Y, LIU C J, YUE H R, LIANG B. Highly selective hydrogenation of diesters to ethylene glycol and ethanol on aluminum-promoted CuAl/SiO2 catalysts[J]. Catal Today,2021,368:173−180. doi: 10.1016/j.cattod.2019.12.034 [24] ZHAO Y J, SHAN B, WANG Y, ZHOU J H, WANG S P, MA X B. An effective CuZn-SiO2 bimetallic catalyst prepared by hydrolysis precipitation method for the hydrogenation of methyl acetate to ethanol[J]. Ind Eng Chem Res,2018,57(13):4526−4534. doi: 10.1021/acs.iecr.7b05391 [25] LIU H L, HUANG Z W, KANG H X, LI X M, XIA C, CHEN J, LIU H C. Efficient bimetallic NiCu-SiO2 catalysts for selective hydrogenolysis of xylitol to ethylene glycol and propylene glycol[J]. Appl Catal B: Environ,2018,220:251−263. doi: 10.1016/j.apcatb.2017.08.022 [26] LI H T, ZHANG Y, ZHANG H X, QIN X Q, XU Y L, WU R F, JIANG Z, ZHAO Y X. The nature of the deactivation of hydrothermally stable Ni/SiO2-Al2O3 catalyst in long-time aqueous phase hydrogenation of crude 1, 4-butanediol[J]. Chin J Chem Eng,2019,27(12):2960−2967. doi: 10.1016/j.cjche.2019.03.008 [27] DAI F, MUHAMMAD Y, GONG X, LI C S, LI Z X, ZHANG S J. Low-temperature and low-pressure fuel hydrodesulfurization by solid catalyst coupling with ionic liquids[J]. Fuel,2014,134:74−80. doi: 10.1016/j.fuel.2014.05.027 [28] REN Z, YOUNIS M N, WU H, LI C S, YANG X G, WANG G Y. Design and synthesis of La-modified copper phyllosilicate nanotubes for hydrogenation of methyl acetate to ethanol[J]. Catal Lett,2021,151(10):3089−3102. doi: 10.1007/s10562-021-03555-2 [29] LI H B, CUI Y Y, LIU Y X, ZHANG L, ZHANG Q, ZHANG J H, DAI W L. Highly efficient Ag-modified copper phyllosilicate nanotube: Preparation by co-ammonia evaporation hydrothermal method and application in the selective hydrogenation of carbonate[J]. J Mater Sci Technol,2020,47:29−37. doi: 10.1016/j.jmst.2020.02.020 [30] YU X B, VEST T A, GLEASON-BOURE N, KARAKALOS S G, TATE G L, BURKHOLDER M, MONNIER J R, WILLIAMS C T. Enhanced hydrogenation of dimethyl oxalate to ethylene glycol over indium promoted Cu/SiO2[J]. J Catal,2019,380:289−296. doi: 10.1016/j.jcat.2019.10.001 [31] WANG Y N, DUAN X P, ZHENG J W, LIN H Q, YUAN Y Z, ARIGA H, TAKAKUSAGI S, ASAKURA K. Remarkable enhancement of Cu catalyst activity in hydrogenation of dimethyl oxalate to ethylene glycol using gold[J]. Catal Sci Technol,2012,2(8):1637−1639. doi: 10.1039/c2cy20154b [32] ZHANG J W, KONG L X, CHEN Y, HUANG H J, ZHANG H H, YAO Y Q, XU Y X, XU Y, WANG S P, MA X B, ZHAO Y J. Enhanced synergy between Cu0 and Cu+ on nickel doped copper catalyst for gaseous acetic acid hydrogenation[J]. Front Chem Sci Eng,2021,15(3):666−678. doi: 10.1007/s11705-020-1982-1 [33] ZHANG L, MAO J B, LI S M, YIN J M, SUN X D, GUO X W, SONG C S, ZHOU J X. Hydrogenation of levulinic acid into gamma-valerolactone over in situ reduced CuAg bimetallic catalyst: Strategy and mechanism of preventing Cu leaching[J]. Appl Catal B: Environ,2018,232:1−10. doi: 10.1016/j.apcatb.2018.03.033 [34] HUANG Y, ARIGA H, ZHENG X L, DUAN X P, TAKAKUSAGI S, ASAKURA K, YUAN Y Z. Silver-modulated SiO2-supported copper catalysts for selective hydrogenation of dimethyl oxalate to ethylene glycol[J]. J Catal,2013,307:74−83. doi: 10.1016/j.jcat.2013.07.006 [35] ZHOU J X, GUO L Y, GUO X W, MAO J B, ZHANG S G. Selective hydrogenolysis of glycerol to propanediols on supported Cu-containing bimetallic catalysts[J]. Green Chem,2010,12(10):1835−1843. doi: 10.1039/c0gc00058b [36] YASEEN M, SHAKIRULLAH M, AHMAD I, RAHMAN A U, RAHMAN F U, Usman M, RAZZAQ R. Simultaneous operation of dibenzothiophene hydrodesulfurization and methanol reforming reactions over Pd promoted alumina based catalysts[J]. J Fuel Chem Technol,2012,40(6):714−720. doi: 10.1016/S1872-5813(12)60027-9 [37] LIU Y T, DING J, YANG J Y, BI J C, LIU K F, CHEN J G. Stabilization of copper catalysts for hydrogenation of dimethyl oxalate by deposition of Ag clusters on Cu nanoparticles[J]. Catal Commun,2017,98:43−46. doi: 10.1016/j.catcom.2017.05.007 [38] ZHAO Y J, GUO Z Y, ZHANG H J, PENG B, XU Y X, WANG Y, ZHANG J, XU Y, WANG S P, MA X B. Hydrogenation of diesters on copper catalyst anchored on ordered hierarchical porous silica: Pore size effect[J]. J Catal,2018,357:223−237. doi: 10.1016/j.jcat.2017.11.006 [39] MARCHI A J, FIERRO J L G, SANTAMARIA J, MONZON A. Dehydrogenation of isopropylic alcohol on a Cu/SiO2 catalyst: A study of the activity evolution and reactivation of the catalyst[J]. Appl Catal A: Gen,1996,142(2):375−386. doi: 10.1016/0926-860X(96)00087-7 [40] SCHOLTEN J J F, KONVALINKA J A, BEEKMAN F W. Reaction of nitrous oxide and oxygen with silver surfaces, and application to the determination of free-silver surface areas of catalysts[J]. J Catal,1973,28(2):209−220. doi: 10.1016/0021-9517(73)90003-1 [41] CHEN L F, GUO P J, QIAO M H, YAN S R, LI H X, SHEN W, XU H L, FAN K N. Cu/SiO2 catalysts prepared by the ammonia-evaporation method: texture, structure, and catalytic performance in hydrogenation of dimethyl oxalate to ethylene glycol[J]. J Catal,2008,257(1):172−180. doi: 10.1016/j.jcat.2008.04.021 [42] TOUPANCE T, KERMAREC M, LOUIS C. Metal particle size in silica-supported copper catalysts. Influence of the conditions of preparation and of thermal pretreatments[J]. J Phys Chem B,2000,104(5):965−972. doi: 10.1021/jp993399q [43] REN Z H, YOUNIS M N, ZHAO H, LI C S, YANG X G, WANG E, WANG G Y. Silver modified Cu/SiO2 catalyst for the hydrogenation of methyl acetate to ethanol[J]. Chin J Chem Eng,2020,28(6):1612−1622. doi: 10.1016/j.cjche.2020.02.011 [44] FELLINGER T P, HASCHE F, STRASSER P, ANTONIETTI M. Mesoporous nitrogen-doped carbon for the electrocatalytic synthesis of hydrogen peroxide[J]. J Am Chem Soc,2012,134(9):4072−4075. doi: 10.1021/ja300038p [45] 李祥祥, 朱贻安, 周静红, 周兴贵, 李伟. 银和铜催化草酸二甲酯加氢制乙醇酸甲酯反应机理的理论研[J]. 天然气化工-C1化学,2018,43(6):17−22.LI Xiang-xiang, ZHU Yi-an, ZHOU Jing-hong, ZHOU Xing-gui, LI Wei. Theoretical study on the mechanism of hydrogenation of dimethyl oxalate to methyl glycolate catalyzed by silver and copper[J]. Nat Gas Chem-C1 Chem,2018,43(6):17−22. [46] YAN W Q, ZHANG J B, ZHOU R J, CAO Y Q, ZHU Y A, ZHOU J H, SUI Z J, LI W, CHEN D, ZHOU X G. Identification of synergistic actions between Cu0 and Cu+ sites in hydrogenation of dimethyl oxalate from microkinetic analysis[J]. Ind Eng Chem Res,2020,59(52):22451−22459. doi: 10.1021/acs.iecr.0c04525 -




 下载:
下载: