Study on CO2 absorption-mineralization characteristics of mixed amine solution coupled with CaO and key influencing factors in mineralization process
-
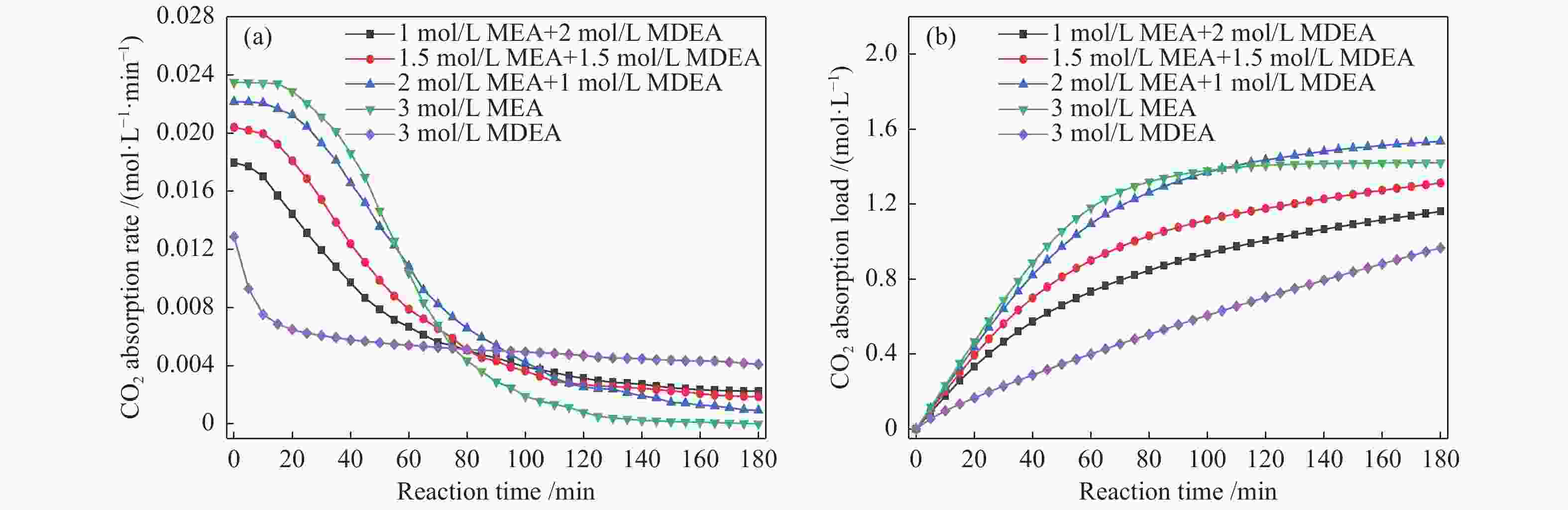
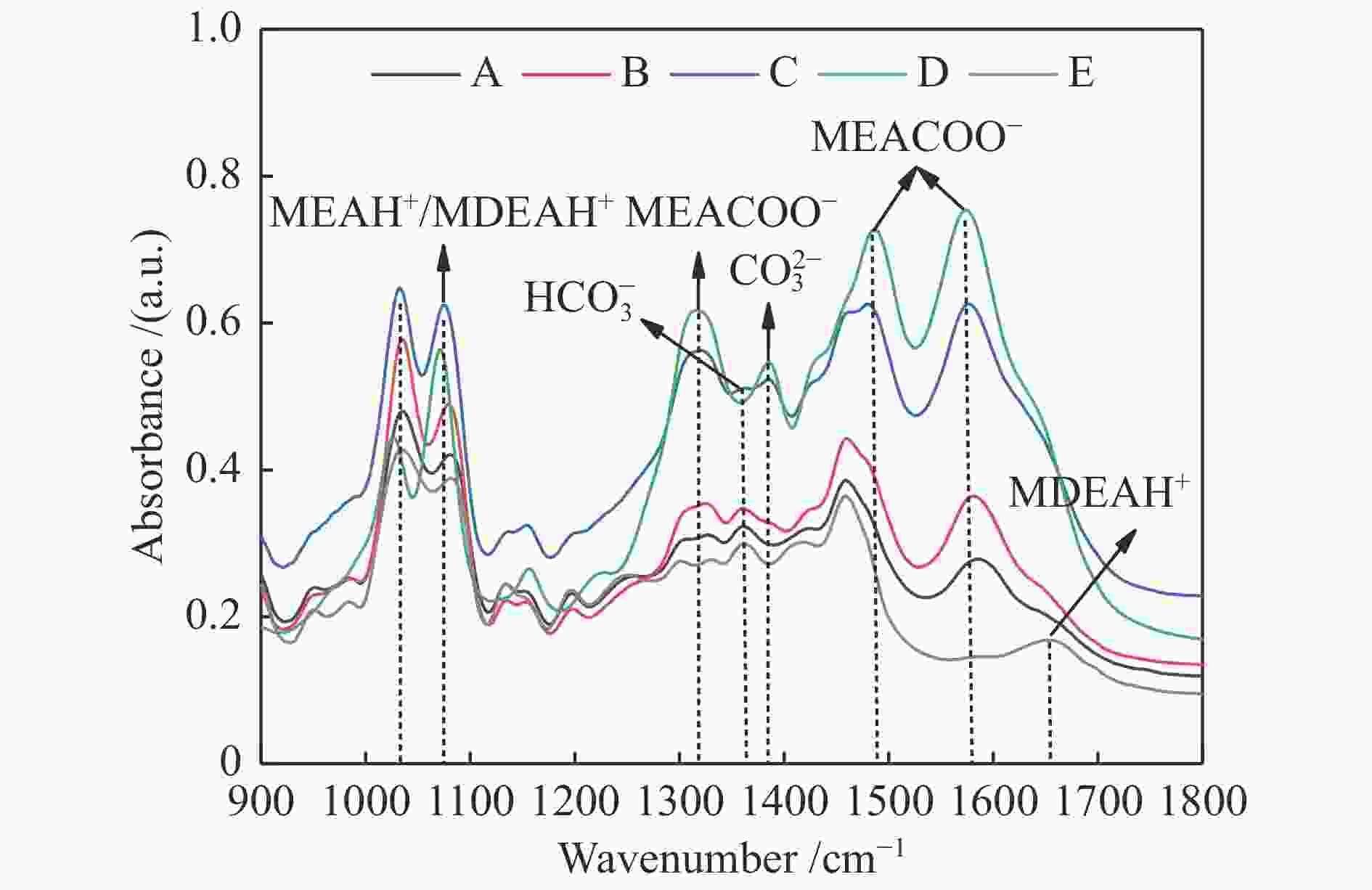
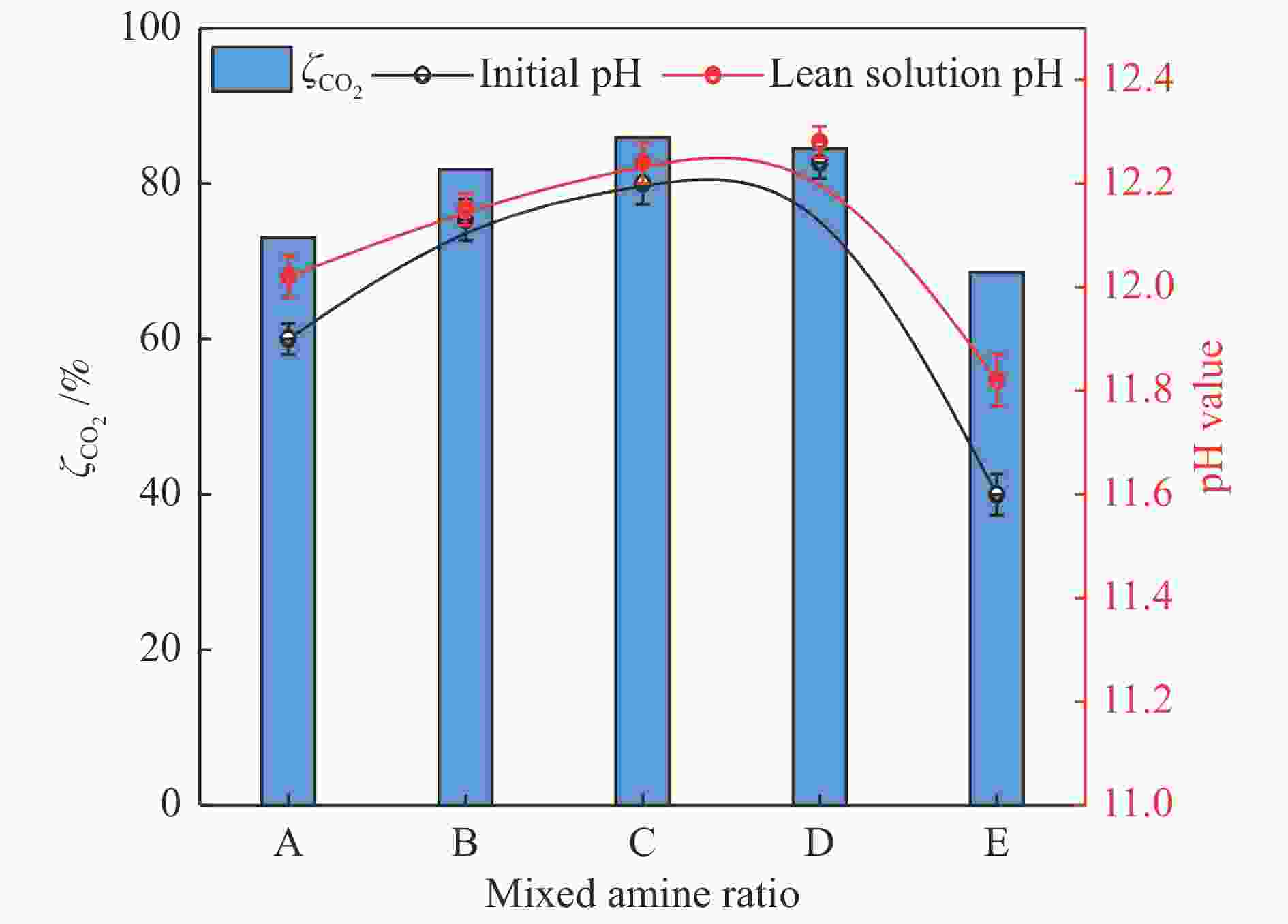
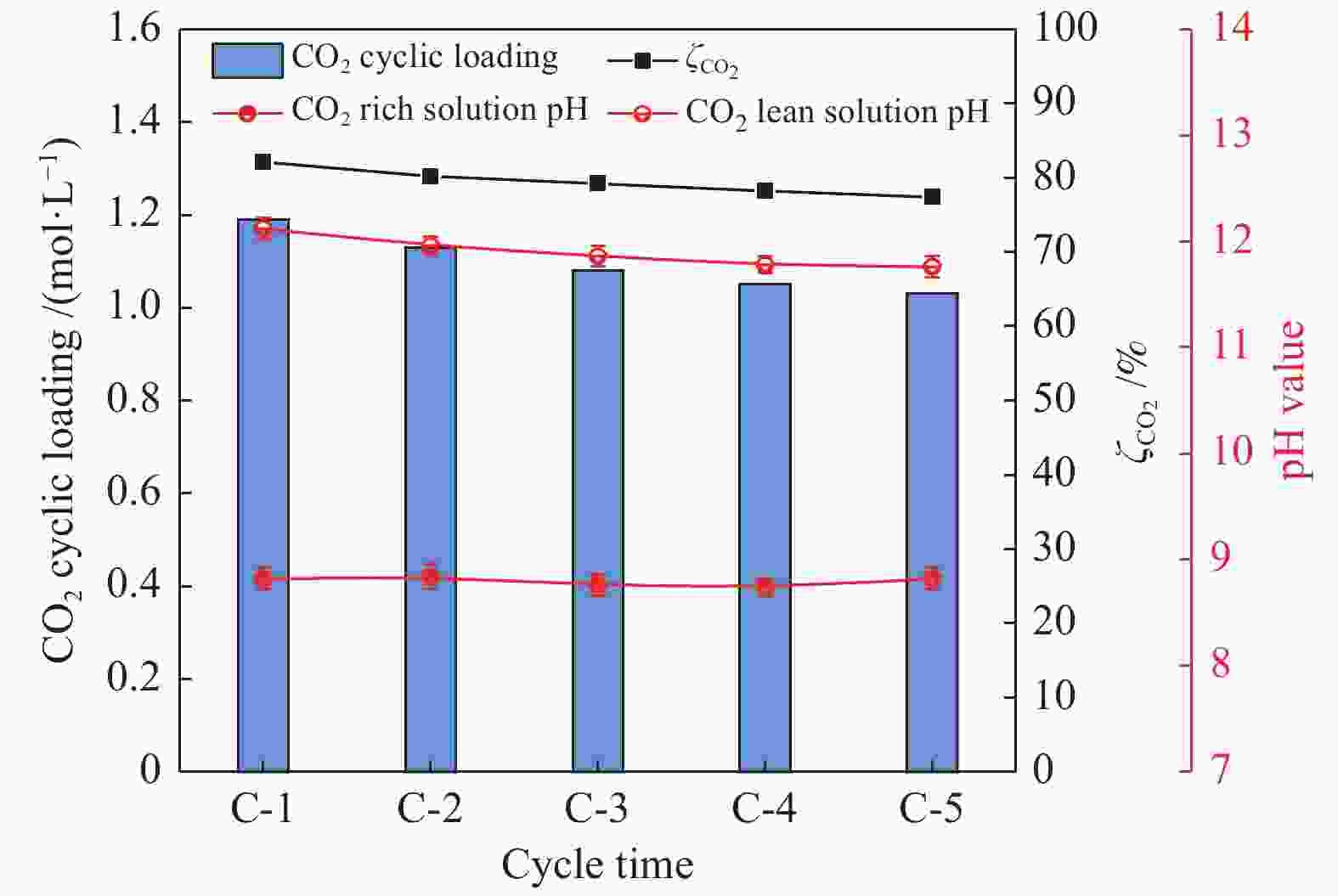
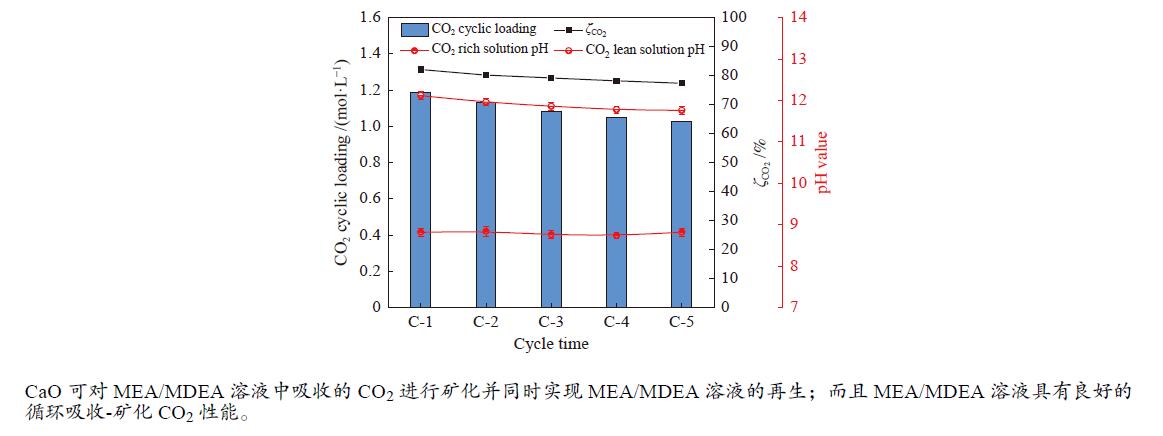
摘要: 有机胺捕集CO2是一种有效的燃烧后碳捕集(PCC)技术,同时CO2矿物碳酸化是一种安全和稳定封存CO2的方法。本研究通过将两种方式结合,以MEA/MDEA混合胺溶液作为CO2吸收剂,以CaO作为CO2矿化原料,研究了不同混合胺溶液配比、温度、反应时间和CaO添加比例等条件下混合胺溶液耦合CaO吸收-矿化CO2性能。结果表明,在常温常压下CaO可对MEA/MDEA溶液中吸收的CO2进行有效矿化并同时实现MEA/MDEA溶液的再生;并且经过五次循环吸收-矿化实验后,MEA/MDEA溶液仍保持了较高的CO2转化率(77.4%)和CO2循环负荷(1.03 mol/L)。FT-IR和XRD表征分析表明,CaO的添加向MEA/MDEA溶液中提供了大量Ca2+和OH−,可分别与溶液中的
${\rm{CO}}_3^{2-} /{\rm{HCO}}_3^- $ 、质子化胺反应生成碳酸钙沉淀和游离胺,从而同时完成对CO2的矿化和MEA/MDEA溶液的化学再生;并且得到的固体产物主要成分为碳酸钙,方解石是其主要晶型。Abstract: Amine CO2 capture is an effective post-combustion carbon capture (PCC) technology, while CO2 mineral carbonation is a safe and stable method for CO2 storage. In this paper, these two methods were combined, and the CO2 absorption-mineralization performance of mixed amine solution coupled with CaO under different ratios of mixed amine solution, temperature, reaction time and CaO addition ratio were studied by using MEA/MDEA mixed amine solution as the CO2 absorbent and using CaO as the CO2 mineralizing raw material. The results show that CaO could effectively mineralize the CO2 absorbed in MEA/MDEA solution, realizing the regeneration of MEA/MDEA solution simultaneously under normal temperature and pressure. Meanwhile, the MEA/MDEA solution can still maintain a high CO2 conversion rate (77.4%) and CO2 cycle loading (1.03 mol/L) after five cycles of absorption-mineralization experiments. The FT-IR and XRD analyses reveal that the addition of CaO makes a large amount of Ca2+ and OH- into the MEA/MDEA solution, which could react with CO$_3^{2{\rm{ - }}} $ /HCO$_3^{\rm{ - }} $ and protonated amine in the solution to form calcium carbonate precipitate and free amine respectively, thus realizing the mineralization of CO2 and the regeneration of MEA/MDEA solution. The main component of solid products obtained is calcium carbonate, and calcite is its main crystal form.-
Key words:
- CO2 /
- mixed amine /
- mineral carbonation /
- chemical regeneration /
- calcium carbonate
-
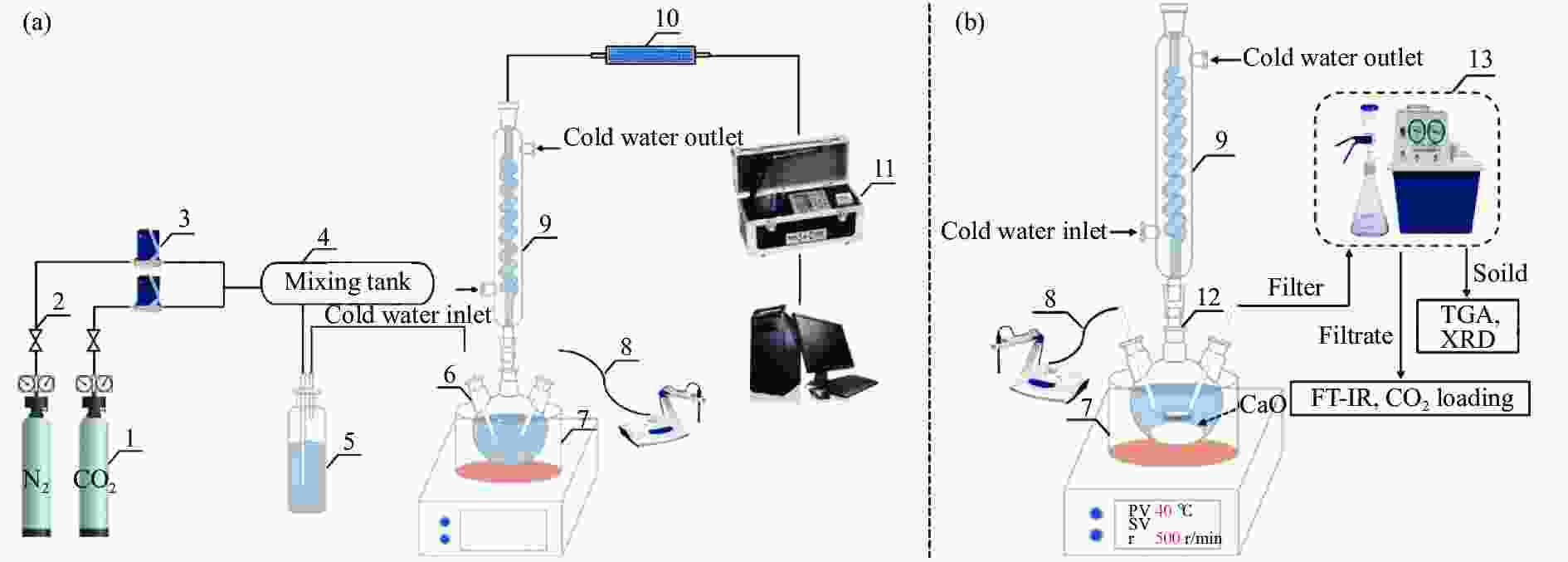
图 1 CO2吸收和矿化反应系统示意图
Figure 1 (a) System of CO2 absorption, (b) system of CO2 mineralization
1: gas cylinder; 2: pressure reducing valve; 3: mass flowmeter; 4: mixing tank; 5: buffer bottle; 6: CO2 bubbling absorption device; 7: thermostatic heating magnetic stirrer; 8: pH meter; 9: serpentine condenser; 10: drying tube; 11: flue gas analyzer; 12: slurry reaction device; 13: filter unit
表 1 MEA/MDEA混合胺溶液耦合CaO吸收-矿化CO2反应过程
Table 1 CO2 reactions in the absorption-mineralization process with MEA/MDEA mixed amine solution coupling CaO
CO2 absorption process MEA RNH2 + CO2 (aq) ↔ ${\rm{RNH} }^{\text{+} } _{2 }$COO−(Zwitterion) (1) ${\rm{RNH} }^+_{2 }$COO− + RNH2 ↔ RNHCOO− +${\rm{RNH} }^ +_{3}$ (Protonated amine) (2) RNHCOO− + H2O ↔ RNH2 + ${\rm{HCO}}_{3}^{{{ - }}} $ (3) MDEA CO2 (aq) + H2O ↔ H2CO3 ↔ H+ + ${\rm{HCO}}_{3}^{{{ - }}} $ (4) R1R2R3N + H+ → R1R2R3NH+(Protonated amine) (5) CO2 mineralization process CaO(s) + H2O → Ca2+ + 2OH− (6) Ca2+ + ${\rm{HCO}}_{3}^{{\rm{ - }}} $ → CaCO3↓+ H+ (7) ${\rm{HCO}}_{3}^{{{ - }}} $ + OH− ↔ ${\rm{CO}}_{3}^{{2}{ - }} $ + H2O (8) Ca2++$ {\rm{CO}}_{3}^{{2}{\rm{ - }}} $→CaCO3↓ (9) Mixed amine chemical
regeneration processMEA $ {\rm{RNH}}_{3}^+$ + OH− ↔ RNH2 + H2O (10) MDEA R1R2R3NH+ + OH− ↔ R1R2R3N + H2O (11) Note: RNH2 represents MEA, R1R2R3N represents MDEA, and R, R1, R2 and R3 represent different alkyl side chains -
[1] KAMRAN U, PARK S J. Chemically modified carbonaceous adsorbents for enhanced CO2 capture: A review[J]. J Clean Prod,2021,290:125776. doi: 10.1016/j.jclepro.2020.125776 [2] ZHANG Z W, BORHANI T N, OLABI A G. Status and perspective of CO2 absorption process[J]. Energy,2020,205:118057. doi: 10.1016/j.energy.2020.118057 [3] JIANG K, ASHWORTH P. The development of Carbon Capture Utilization and Storage (CCUS) research in China: A bibliometric perspective[J]. Renewable Sustainable Energy Rev,2021,138:110521. doi: 10.1016/j.rser.2020.110521 [4] YU B, YU H, LI K K, JI L, YANG Q, WANG X L, CHEN Z L, MEGHARAJ M. A diamine-based integrated absorption-mineralization process for carbon capture and sequestration: energy savings, fast kinetics, and high stability[J]. Environ Sci Technol,2018,52:13629−13637. doi: 10.1021/acs.est.8b04253 [5] HONG S J, SIM G, MOON S, PARK Y. Low-temperature regeneration of amines integrated with production of structure-controlled calcium carbonates for combined CO2 capture and utilization[J]. Energy Fuels,2020,34:3532−3539. doi: 10.1021/acs.energyfuels.9b04339 [6] LIU W, SU S, XU K, CHEN Q D, XU J, SUN Z J, WANG X L, XUE Y T, XIANG J. CO2 sequestration by direct gas-solid carbonation of fly ash with steam addition[J]. J Clean Prod,2018,178:98−107. doi: 10.1016/j.jclepro.2017.12.281 [7] 王中辉, 苏胜, 尹子骏, 安晓雪, 赵志刚, 陈逸峰, 刘涛, 汪一, 胡松, 向军. CO2矿化及吸收-矿化一体化(IAM)方法研究进展[J]. 化工进展,2021,40(4):2318−2327.WANG Zhong-hui, SU Sheng, YIN Zi-jun, AN Xiao-xue, ZHAO Zhi-gang, CHEN Yi-feng, LIU Tao, WANG Yi, HU Song, XIANG Jun. Research progress of CO2 mineralization and integrated absorption-mineralization (IAM) method[J]. Chem Eng Prog,2021,40(4):2318−2327. [8] 林海周, 杨晖, 罗海中, 裴爱国, 方梦祥. 烟气二氧化碳捕集胺类吸收剂研究进展[J]. 南方能源建设,2019,6(1):16−21.LIN Hai-zhou, YANG Hui, LUO Hai-zhong, PEI Ai-guo, FANG Meng-xiang. Research progress on amine absorbent for CO2 capture from flue gas[J]. South Energy Constr,2019,6(1):16−21. [9] HADRI N E, QUANG D V, GOETHEER E L V, ZAHRA M R M A. Aqueous amine solution characterization for post-combustion CO2 capture process[J]. Appl Energy,2017,185:1433−1449. doi: 10.1016/j.apenergy.2016.03.043 [10] XIAO M, LIU H L, IDEAM R, TONTIWACHWUTHIKUL P, LIANG Z W. A study of structure-activity relationships of commercial tertiary amines for post-combustion CO2 capture[J]. Appl Energy,2016,184:219−229. doi: 10.1016/j.apenergy.2016.10.006 [11] SEMA T, NAAMI A, FU K Y, EDALI M, LIU H L, SHI H C, LIANG Z W, IDEM R, TONTIWACHWUTHIKUL P. Comprehensive mass transfer and reaction kinetics studies of CO2 absorption into aqueous solutions of blended MDEA-MEA[J]. Chem Eng J,2012,209:501−512. doi: 10.1016/j.cej.2012.08.016 [12] RIVERA-TINOCO R, BOUALLOU C. Comparison of absorption rates and absorption capacity of ammonia solvents with MEA and MDEA aqueous blends for CO2 capture[J]. J Clean Prod,2010,18:875−880. doi: 10.1016/j.jclepro.2009.12.006 [13] PARK S, MIN J, LEE M G, JO H Y, PARK J. Characteristics of CO2 fixation by chemical conversion to carbonate salts[J]. Chem Eng J,2013,231:287−293. doi: 10.1016/j.cej.2013.07.032 [14] PARK S, LEE M G, PARK J. CO2 (carbon dioxide) fixation by applying new chemical absorption-precipitation methods[J]. Energy,2013,59:737−742. doi: 10.1016/j.energy.2013.07.057 [15] ARTI M, YOUN M H, PARK K T, KIM H J, KIM Y E, JEONG S K. Single process for CO2 capture and mineralization in various alkanolamines using calcium chloride[J]. Energy Fuels,2017,31(1):763−769. doi: 10.1021/acs.energyfuels.6b02448 [16] KANG J M, MURNANDARI A, YOUN M H, LEE W, PARK K T, KIM Y E, KIM H J, KANG S P, LEE J H, JEONG S K. Energy-efficient chemical regeneration of AMP using calcium hydroxide for operating carbon dioxide capture process[J]. Chem Eng J,2018,335:338−344. doi: 10.1016/j.cej.2017.10.136 [17] JI L, YU H, LI K K, YU B, GRIGORE M, YANG Q, WANG X L, CHEN Z L, ZENG M, ZHAO S F. Integrated absorption-mineralization for low-energy CO2 capture and sequestration[J]. Appl Energy,2018,225:356−366. doi: 10.1016/j.apenergy.2018.04.108 [18] 马伟春, 张卫风, 焦月潭, 黄珍, 李靳, 钟林新. 钙法解吸并固定乙醇胺富液中CO2[J]. 环境科学学报,2018,38(1):109−114.MA Wei-chun, ZHANG Wei-feng, JIAO Yue-tan, HUANG Zhen, LI Jin, ZHONG Lin-xin. Desorption and mineralization of CO2 in monoethanolamine-rich solution by calcium method[J]. Acta Sci Circumst,2018,38(1):109−114. [19] YAN S, FANG M X, LUO Z Y, CEN K F. Regeneration of CO2 from CO2-rich alkanolamines solution by using reduced thickness and vacuum technology: Regeneration feasibility and characteristic of thin-layer solvent[J]. Chem Eng Process,2009,48(1):515−523. doi: 10.1016/j.cep.2008.06.009 [20] LUO C, WU K J, YUE H R, LIU Y Y, ZHU Y M, JIANG W, LU H F, LIANG B. DBU-based CO2 absorption-mineralization system: Reaction process, feasibility and process intensification[J]. Chin J Chem Eng,2020,28(4):1145−1155. doi: 10.1016/j.cjche.2019.12.008 [21] ABASS A O. A review of mineral carbonation technology in sequestration of CO2[J]. J Pet Sci Eng,2013,109:364−392. doi: 10.1016/j.petrol.2013.03.013 [22] 王晓龙, 刘蓉, 纪龙, 郜时旺, 姜宁. 利用粉煤灰与可循环碳酸盐直接捕集固定电厂烟气中二氧化碳的液相矿化法[J]. 中国电机工程学报,2018,38(19):5787−5794.WANG Xiao-long, LIU Rong, JI Long, GAO Shi-wang, JIANG Ning. A new direct aqueous mineralization process using fly ash and recyclable carbonate salts to capture and storage CO2 from flue-gas[J]. Proc CSEE,2018,38(19):5787−5794. [23] GILLES R, GRAEME P. Assessing the chemical speciation during CO2 absorption by aqueous amines using in situ FTIR[J]. Ind Eng Chem Res,2012,51:14317−14324. doi: 10.1021/ie302056f [24] ROBINSON K, MCCLUSKEY A, ATTALLA M. An FTIR spectroscopic study on the effect of molecular structural variations on the CO2 absorption characteristics of heterocyclic amines[J]. ChemPhysChem,2011,12:1088−1099. doi: 10.1002/cphc.201001056 -







 下载:
下载: