Manipulation of hydroisomerization performance on Pt/ZSM-23 by introducing Al2O3
-
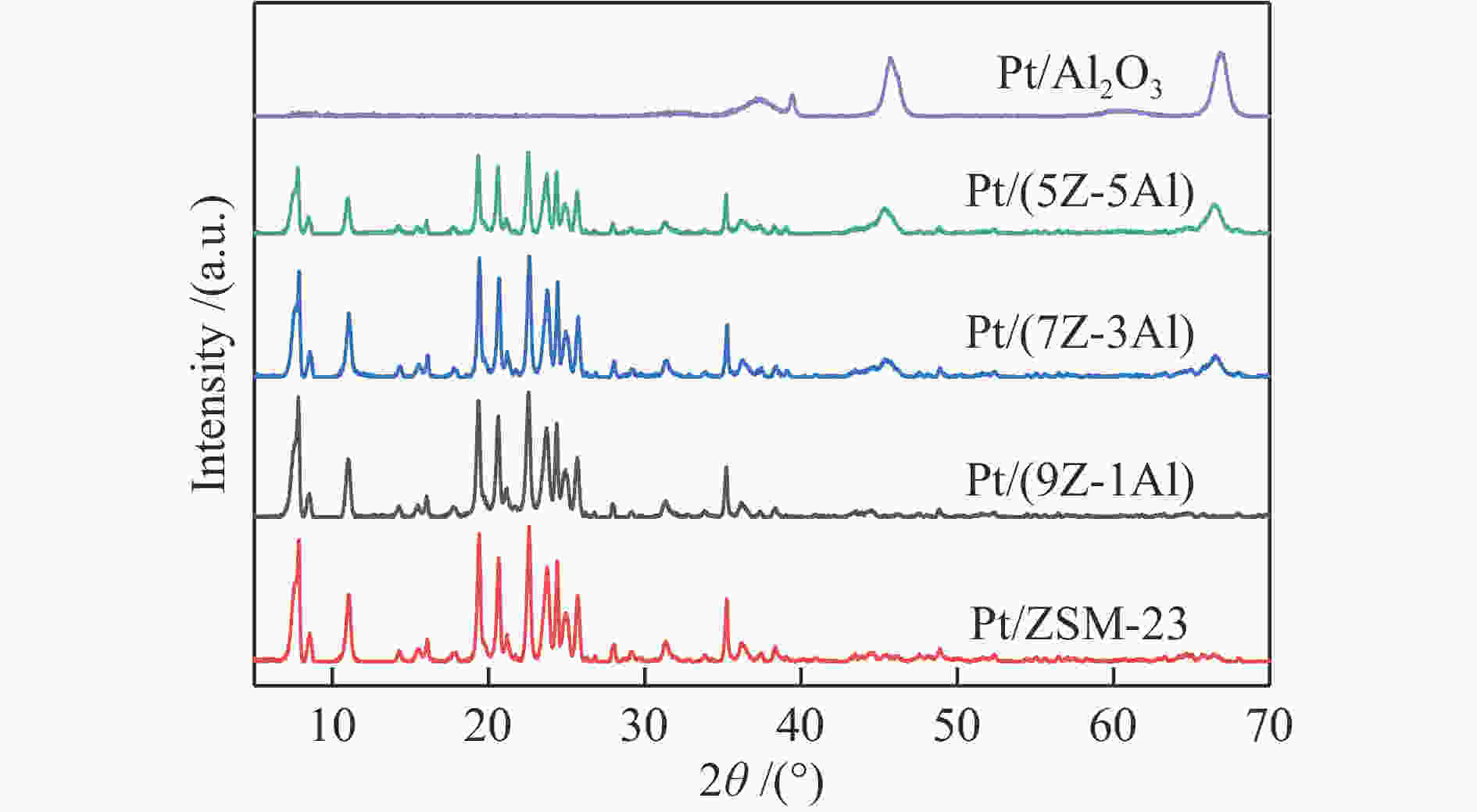
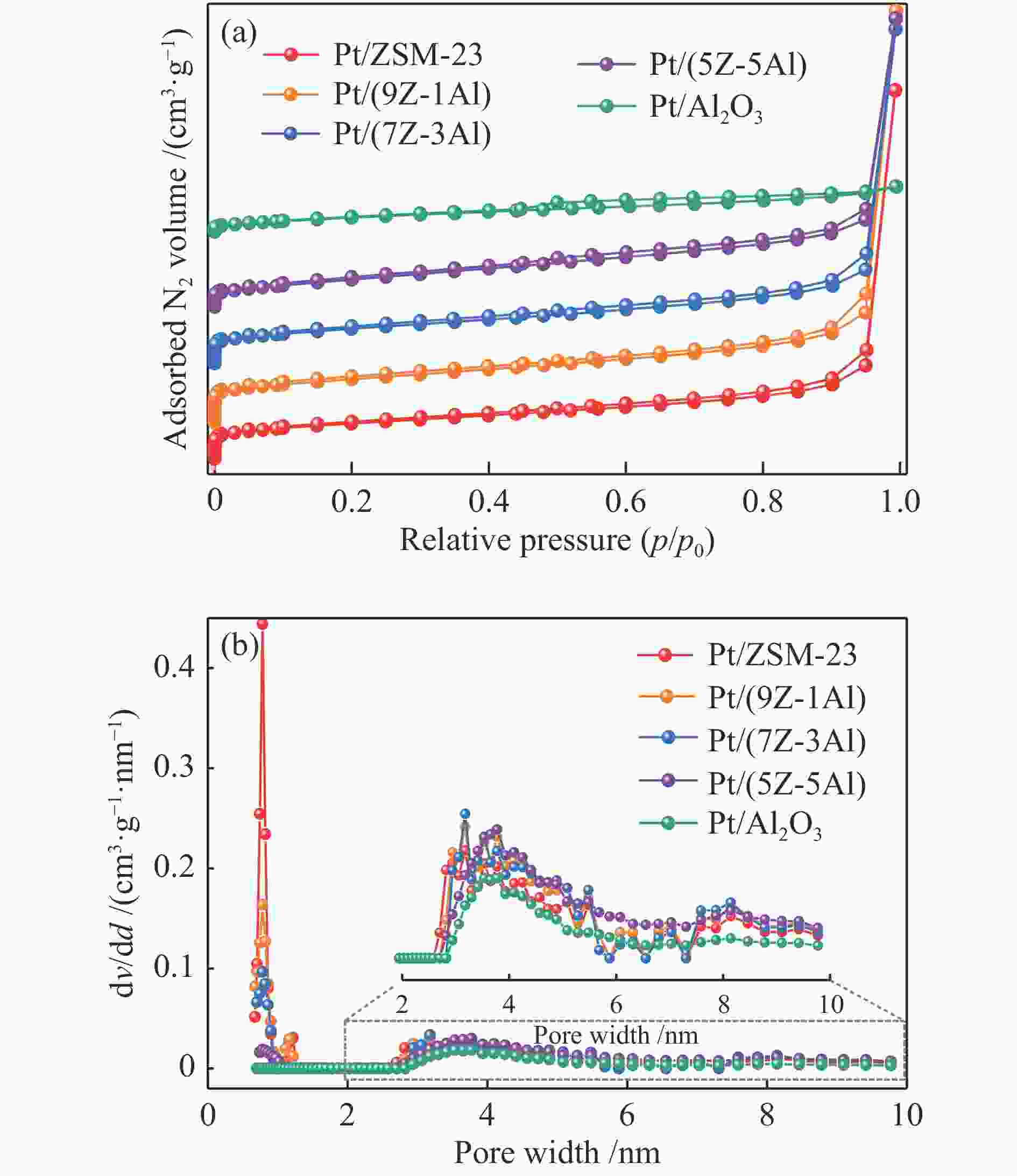
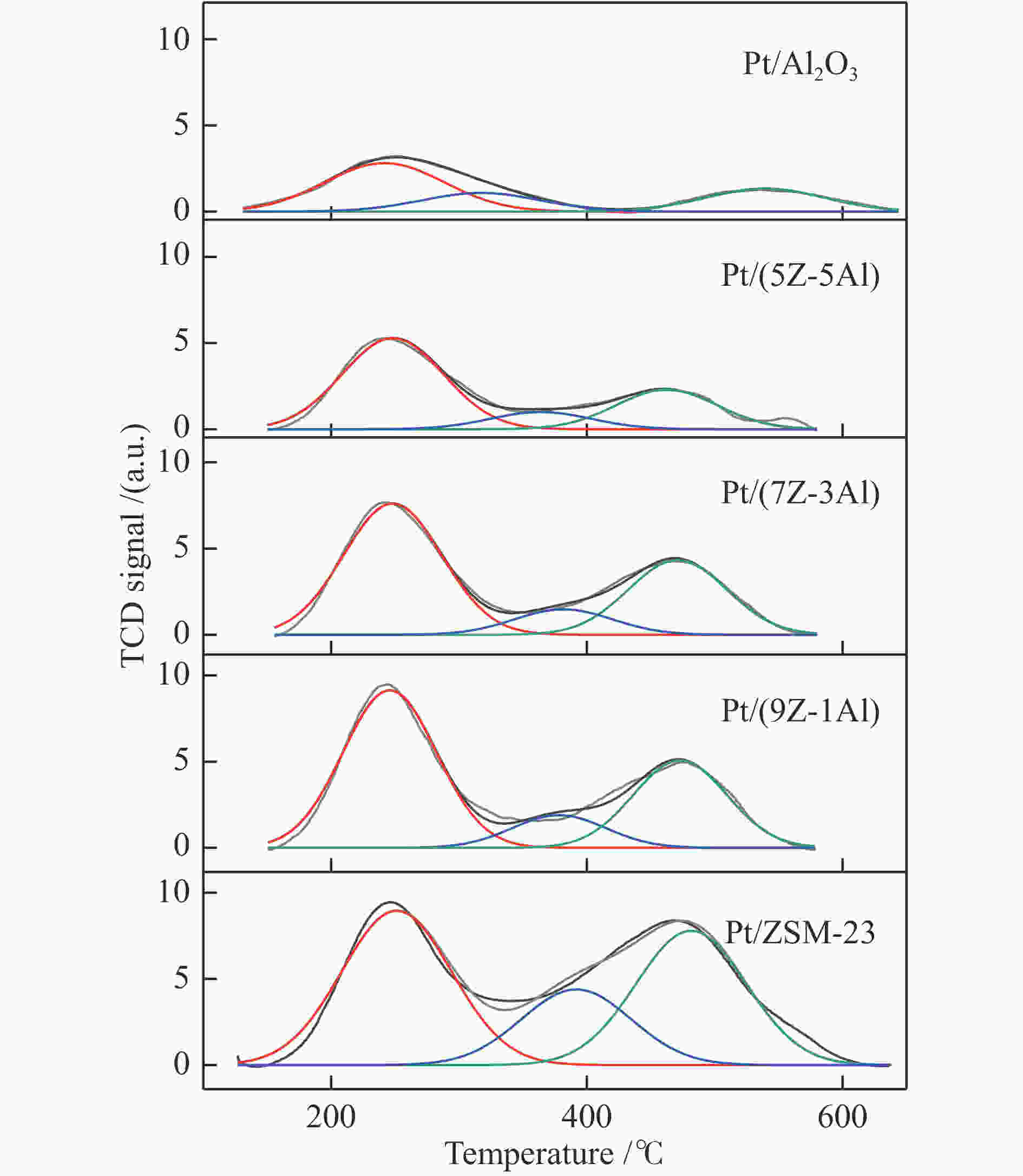
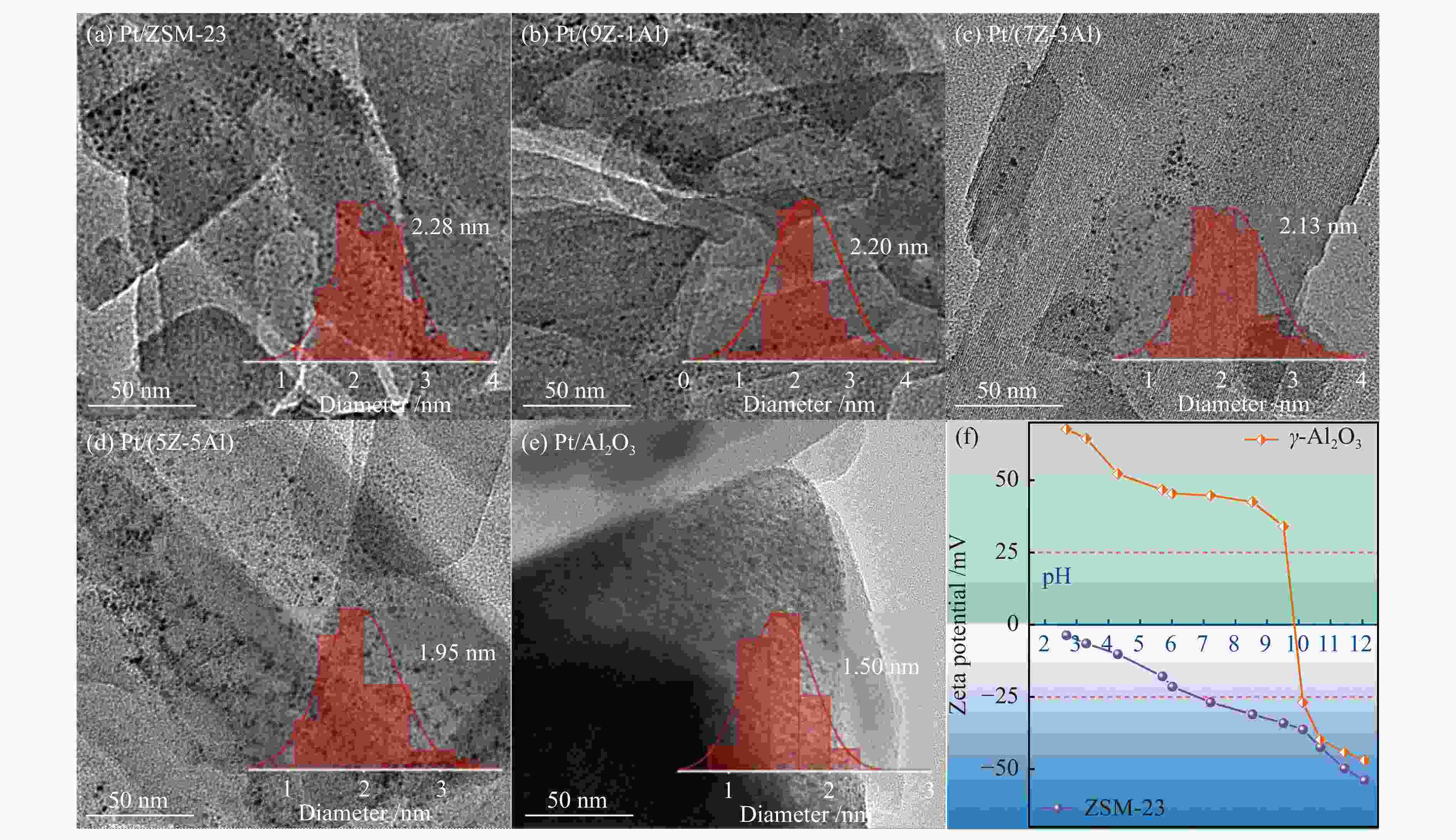
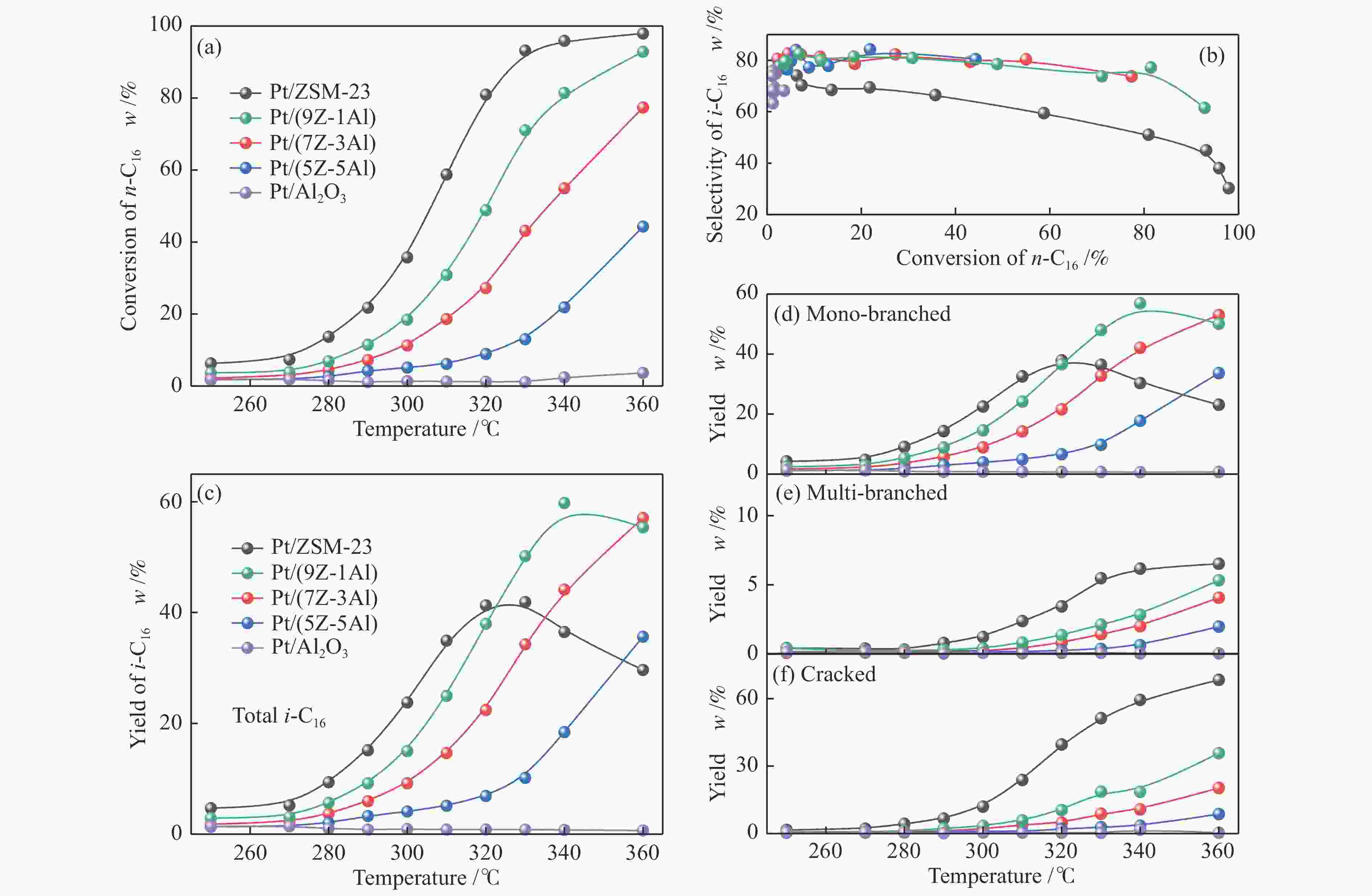
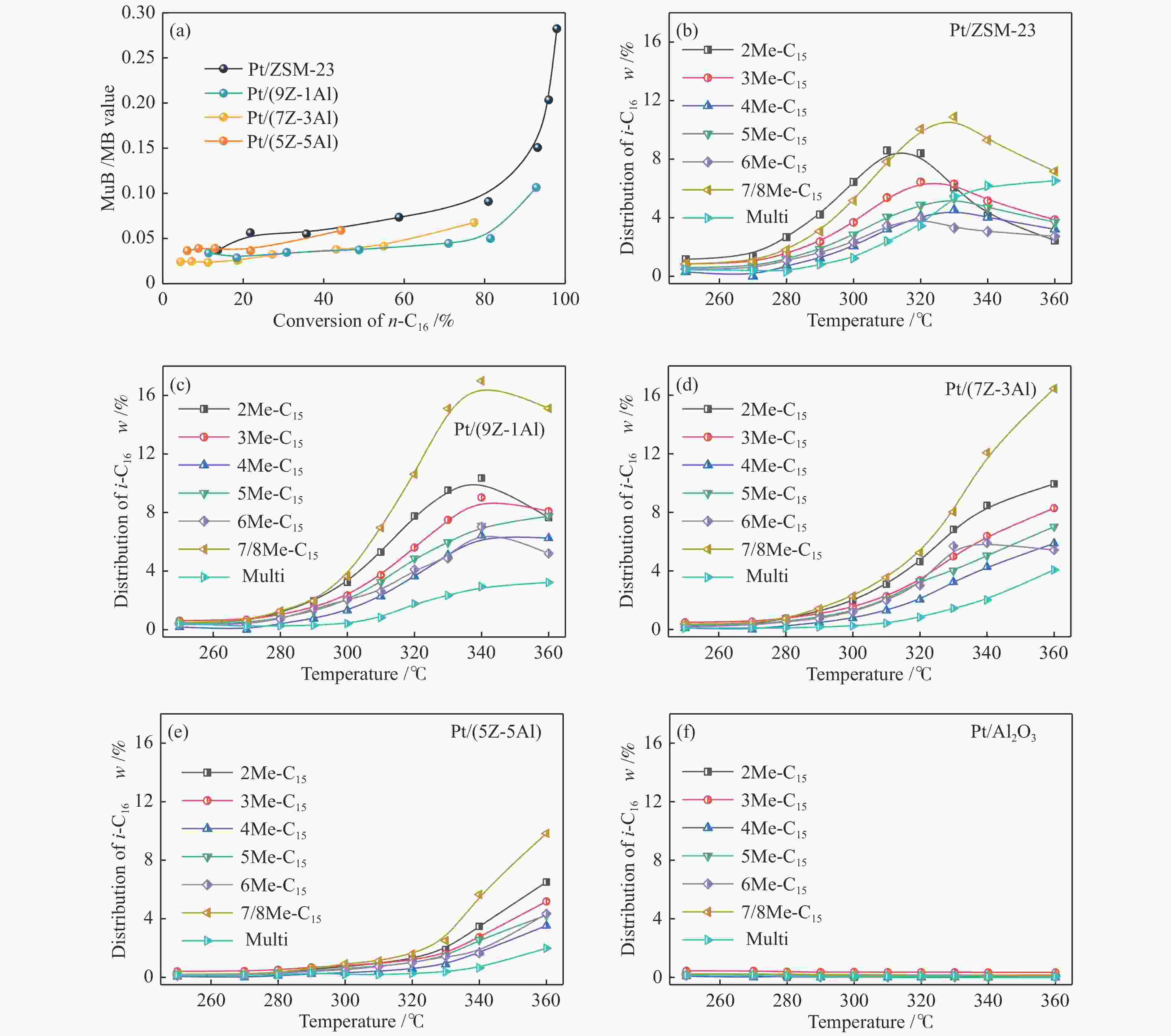
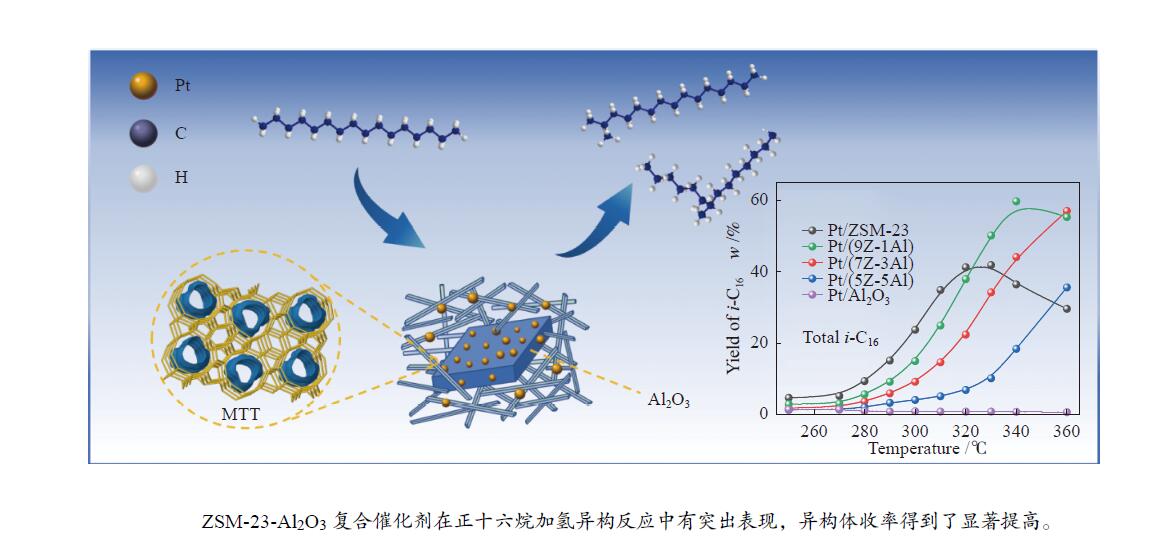
摘要: 采用双模板剂合成了ZSM-23分子筛,并制备ZSM-23-Al2O3复合载体,通过引入Al2O3调控Pt/ZSM-23催化剂的正十六烷(n-C16)加氢异构性能。采用XRD、SEM、TEM、N2物理吸附-脱附及NH3-TPD等方法探讨Al2O3对催化剂结构性质和化学性质的影响。结果表明,引入Al2O3可以提高Pt在复合载体上的分散度,降低催化剂的酸性位点浓度,有效调控了催化剂的金属-酸性浓度平衡(CPt/CA)。适宜的金属-酸性浓度平衡有利于提高对异构产物的选择性并抑制裂化副反应。Al2O3同时对ZSM-23晶粒起到了分散作用,改善了其分子筛纳米晶粒团聚现象,增加了ZSM-23中孔口的暴露数量,促进反应物和中间体的扩散。复合载体催化剂呈现了出色的异构选择性,其中,Pt/(9Z-1Al)催化剂由于具有适宜的金属-酸性浓度平衡呈现最高的异构产物收率,可达60%,明显高于Pt/ZSM-23催化剂的异构体收率(42%)。反应温度低于310 ℃时,Pt/ZSM-23催化剂的吸附模型符合孔口吸附,而高温时则为锁钥吸附。引入Al2O3后ZSM-23有更多相邻孔口暴露,吸附模型以锁钥择形为主,导致了7/8Me-C15异构产物的大量生成。Abstract: ZSM-23 zeolite was successfully synthesized in a dual-template system, and ZSM-23-Al2O3 composites with different ratios were also prepared. The hydroisomerization performance of Pt/ZSM-23 catalyst was manipulated by introducing Al2O3, and the influence of Al2O3 on physicochemical properties was investigated by XRD, SEM, TEM, N2 physical adsorption-desorption and NH3-TPD characterizations. The results showed that Al2O3 improved the dispersion of Pt, significantly reduced the acid sites concentration of the catalyst, and regulated the metal-acid balance in quantitative. The suitable metal-acid balance concentration could improve the selectivity of isomers and suppress the cracking reactions. Meanwhile, Al2O3 dispersed the ZSM-23 grains, which improved the dispersion and increased the number of exposed pores in ZSM-23. Thus the diffusion efficiency of reactants and intermediates could be promoted and the isomer products selectivity could be improved. All composite catalysts showed high selectivity of isomer products, among which, Pt/(9Z-1Al) had the highest yield of isomer products due to its suitable metal-acid concentration balance, reached 60% at 340 ℃, which was a significant improvement compared with Pt/ZSM-23 (42%). When the reaction temperature was lower than 310 ℃, the pore mouth mechanism dominated in Pt/ZSM-23, while the key-lock mechanism was significantly strengthened at higher reaction temperature. After the introduction of Al2O3, more adjacent pores in ZSM-23 were exposed and the key-lock mechanism became the domination, which led to a large number of 7/8Me-C15 isomers.
-
Key words:
- ZSM-23 /
- Al2O3 /
- hydroisomerization /
- n-hexadecane
-
表 1 Pt/ZSM-23、Pt/Al2O3及其复合载体催化剂的结构性质
Table 1 Textural properties of Pt/ZSM-23, Pt/Al2O3, and Pt catalysts supported ZSM-23-Al2O3
Sample SBETa /
(m2·g−1)Smicrob /
(m2·g−1)Sextc /
(m2·g−1)vtotald /
(cm3·g−1)Pt/ZSM-23 221 119 102 0.78 Pt/(9Z-1Al) 219 95 124 0.79 Pt/(7Z-3Al) 189 68 121 0.70 Pt/(5Z-5Al) 137 21 116 0.60 Pt/Al2O3 60 0 60 0.09 a BET method, b t-plot method, c Sext = SBET−Smicro,
d vtotal determined at p/p0 = 0.99表 2 由NH3-TPD确定的催化剂酸性位浓度
Table 2 Concentration of acid sites of catalysts determined by NH3-TPD
Sample (Si/Al2)a Acidic sites/ (NH3 mmol·g−1)b weak medium strong total Pt/ZSM-23 79 0.124 0.061 0.108 0.293 Pt/(9Z-1Al) 52 0.110 0.033 0.069 0.213 Pt/(7Z-3Al) 29 0.096 0.029 0.062 0.187 Pt/(5Z-5Al) 21 0.076 0.020 0.050 0.146 Pt/Al2O3 − 0.041 0.006 0.019 0.066 a determined by ICP-AES, b determined by NH3-TPD and calculated by the deconvolution method 表 3 催化剂的金属担载特性及酸性质
Table 3 Metal loading and acid properties of catalysts
Sample Pt wa/% dPtb /nm Dc /% CPtd /(μmol·g−1) CA /(μmol·g−1) CPt/CA Pt/ZSM-23 0.60 2.28 50 15 169 0.09 Pt/(9Z-1Al) 0.64 2.20 51 17 102 0.17 Pt/(7Z-3Al) 0.69 2.13 53 19 91 0.21 Pt/(5Z-5Al) 0.73 1.95 58 22 70 0.31 Pt/Al2O3 0.82 1.50 75 32 25 1.27 a determined by ICP-AES, b average size with standard deviations analyzed by more than 200 particles from TEM images, tolerance scope was ± 0.49 nm, which was obtained via normally distributed data statistics, c D = 1.13/dPt size[27], d CPt denotes density of surface Pt atoms, calculated by Pt loading and dispersion -
[1] ZHANG S, ZHANG Y, TIERNEY J W, WENDER I. Anion-modified zirconia: Effect of metal promotion and hydrogen reduction on hydroisomerization of n-hexadecane and Fischer-Tropsch waxes[J]. Fuel Process Technol,2001,69(1):59−71. doi: 10.1016/S0378-3820(00)00133-8 [2] CALEMMA V, PERATELLO S, PEREGO C. Hydroisomerization and hydrocracking of long chain n-alkanes on Pt/amorphous SiO2-Al2O3 catalyst[J]. Appl Catal A: Gen,2000,190(1/2):207−218. doi: 10.1016/S0926-860X(99)00292-6 [3] REGALI F, LIOTTA L F, VENEZIA A M, MONTES V, BOUTONNET M, JRS S. Effect of metal loading on activity, selectivity and deactivation behavior of Pd/silica-alumina catalysts in the hydroconversion of n-hexadecane[J]. Catal Today,2014,223(3):87−96. [4] MARTENS J A, VANBUTSELE, JACOBS P A, DENAYER, OCAKOGLU. Evidences for pore mouth and key-lock catalysis in hydroisomerization of long n-alkanes over 10-ring tubular pore bifunctional zeolites[J]. Catal Today,2001,65(2):111−116. [5] MARTENS J A, JACOBS P A, WEITKAMP J. Attempts to rationalize the distribution of hydrocracked products. I qualitative description of the primary hydrocracking modes of long chain paraffins in open zeolites[J]. Appl Catal,1986,20(1/2):239−281. doi: 10.1016/0166-9834(86)80020-3 [6] ZHANG M, LIU Q, LONG H, SUN L, MURAYAMA T, QI C. Insights into Au nanoparticle size and chemical state of Au/ZSM-5 catalyst for catalytic cracking of n-octane to increase propylene production[J]. J Phys Chem C,2021,125(29):16013−16023. doi: 10.1021/acs.jpcc.1c04608 [7] 侯冉冉, 胡志海, 郭庆洲, 毕云飞, 高杰, 李洪辉. 加氢异构催化剂性质对长链烷烃异构行为的影响[J]. 石油学报(石油加工),2022,38(1):20−28.HOU Ran-ran, HU Zhi-hai, GUO Qing-zhou, BI Yun-fei, GAO Jie, LI Hong-hui. Effects of catalysts on catalytic performance in long chain n-alkanes isomerization[J]. Acta Pet Sin(Pet Process Sect),2022,38(1):20−28. [8] BURTON A W. A priori phase prediction of zeolites: Case study of the structure-directing effects in the synthesis of MTT-type zeolites[J]. J Am Chem Soc,2007,129(24):7627−7637. doi: 10.1021/ja070303u [9] JR A, LAPIERRE R B, SCHLENKER J L, WOOD J D, VALYOCSIK E W, RUBIN M K, HIGGINS J B, ROHRBAUGH W J. The framework topology of ZSM-23: A high silica zeolite[J]. Zeolites,1985,5(6):352−354. doi: 10.1016/0144-2449(85)90123-X [10] HOLZINGER J, NIELSEN M, BEATO P, BROGAARD R Y, BUONO C, DYBALLA M. Identification of distinct framework aluminum sites in zeolite ZSM-23: A combined computational and experimental 27Al NMR study[J]. J Phys Chem C,2019,123(13):7831−7844. doi: 10.1021/acs.jpcc.8b06891 [11] DEGNAN T F. Applications of zeolites in petroleum refining[J]. Top Catal,2000,13(4):349−356. doi: 10.1023/A:1009054905137 [12] HENGSAWAD T, SRIMINGKWANCHAI C, BUTNARK S, RESASCO D E, JONGPATIWUT S. Effect of metal-acid balance on hydroprocessed renewable jet fuel synthesis from hydrocracking and hydroisomerization of biohydrogenated diesel over Pt-supported catalysts[J]. Ind Eng Chem Res,2018,57(5):1429−1440. doi: 10.1021/acs.iecr.7b04711 [13] CHEN Y, LI C, WANG L, ZHANG M, LIANG C. Seed-assisted synthesis of ZSM-23 zeolites in the absence of alkali metal ions[J]. Microporous Mesoporous Mater,2017,252:146−153. doi: 10.1016/j.micromeso.2017.06.013 [14] HUYBRECHTS W, THYBAUT J W, DE WAELE B R, VANBUTSELE G, HOUTHOOFD K J, BERTINCHAMPS F, DENAYER J F M, GAIGNEAUX E M, MARIN G B, BARON G V, JACOBS P A, MARTENS J A. Bifunctional catalytic isomerization of decane over MTT-type aluminosilicate zeolite crystals with siliceous rim[J]. J Catal,2006,239(2):451−459. doi: 10.1016/j.jcat.2006.02.020 [15] SMIRNOVA M Y, PIRYUTKO L V, BRESTER Y S, PARFENOV M V, NOSKOV A S. Effect of the ZSM-23 synthesis method on the properties of Pt/ZSM-23/Al2O3 catalysts in n-decane conversion[J]. Pet Chem,2020,60(2):212−218. doi: 10.1134/S0965544120020085 [16] HUYBRECHTS W, VANBUTSELE G, HOUTHOOFD K J, BERTINCHAMPS F, NARASIMHAN C, GAIGNEAUX E M. Skeletal isomerization of octadecane on bifunctional ZSM-23 zeolite catalyst[J]. Catal Lett,2005,100(3/4):235−242. doi: 10.1007/s10562-004-3461-6 [17] CHEN Y, LI C, CHEN X, LIU Y, LIANG C. Synthesis of ZSM-23 zeolite with dual structure directing agents for hydroisomerization of n-hexadecane[J]. Microporous Mesoporous Mater,2018,268:216−224. doi: 10.1016/j.micromeso.2018.04.033 [18] ALVAREZ F, RIBEIRO F R, PEROT G, THOMAZEAU C, GUISNET M. Hydroisomerization and hydrocracking of alkanes: 7. Influence of the balance between acid and hydrogenating functions on the transformation of n-decane on PtHY catalysts[J]. J Catal,1996,162(2):179−189. doi: 10.1006/jcat.1996.0275 [19] ZHANG Y, LIU D, LOU B, YU R, MEN Z. Hydroisomerization of n-decane over micro/mesoporous Pt-containing bifunctional catalysts: Effects of the MCM-41 incorporation with Y zeolite[J]. Fuel,2018,226(15):204−212. [20] BATALHA N, PINARD L, BOUCHY C, GUILLON E, GUISNET M. n-Hexadecane hydroisomerization over Pt-HBEA catalysts. Quantification and effect of the intimacy between metal and protonic sites[J]. J Catal,2013,307:122−131. doi: 10.1016/j.jcat.2013.07.014 [21] 向江南, 刘伟, 刘成连, 王琰, 陈树伟, 毕士楠, 范彬彬, 李瑞丰. 低硅铝比ZSM-48分子筛合成及其正构十二烷临氢异构催化性能研究[J]. 燃料化学学报,2020,48(1):83−90. doi: 10.3969/j.issn.0253-2409.2020.01.010XIANG Jiang-nan, LIU Wei, LIU Cheng-lian, WANG Yan, CHEN Shu-wei, BI Shi-nan, FAN Bin-bin, LI Rui-feng. Synthesis and hydroisomerization performance of n-C12 over ZSM-48 molecular sieve with low silicon-aluminum ratio[J]. J Fuel Chem Technol,2020,48(1):83−90. doi: 10.3969/j.issn.0253-2409.2020.01.010 [22] MÖLLER K, BEIN T. Crystallization and porosity of ZSM-23[J]. Microporous Mesoporous Mater,2011,143(2):253−262. [23] 戴清, 杨玉旺, 裴仁彦, 吴同旭, 蔡奇, 郭秋双. ZSM-23/活性氧化铝复合催化剂在甲醇脱水反应中的应用研究[J]. 无机盐工业,2018,50(2):70−74.DAI Qing, YANG Yu-wang, PEI Ren-yan, WU Tong-xu, CAI Qi, GUO Qiu-shuang. Study on application of ZSM-23/active Al2O3 composite catalysts in methanol dehydration reaction[J]. Inorg Chem Ind,2018,50(2):70−74. [24] LV G, WANG C, CHI K, LIU H, WANG P, MA H, QU W, TIAN Z. Effects of Pt site distributions on the catalytic performance of Pt/SAPO-11 for n-dodecane hydroisomerization[J]. Catal Today,2018,316:43−50. doi: 10.1016/j.cattod.2018.04.072 [25] ZECEVIC J, VANBUTSELE G, DE JONG K P, MARTENS J A. Nanoscale intimacy in bifunctional catalysts for selective conversion of hydrocarbons[J]. Nature,2015,528(7581):245−248. doi: 10.1038/nature16173 [26] BEN MOUSSA O, TINAT L, JIN X, BAAZIZ W, DURUPTHY O, SAYAG C, BLANCHARD J. Heteroaggregation and selective deposition for the fine design of nanoarchitectured bifunctional catalysts: Application to hydroisomerization[J]. ACS Catal,2018,8(7):6071−6078. doi: 10.1021/acscatal.8b01461 [27] CHENG K, WAL L I, YOSHIDA H, OENEMA J, HARMEL J. Impact of the spatial organization of bifunctional metal-zeolite catalysts on the hydroisomerization of light alkanes[J]. Angew Chem Int Ed,2020,132(9):3620−3628. doi: 10.1002/ange.201915080 [28] BATALHA N, PINARD L, POUILLOUX Y, GUISNET M. Bifunctional hydrogenating/acid catalysis: Quantification of the intimacy criterion[J]. Catal Lett,2013,143(6):587−591. doi: 10.1007/s10562-013-1003-9 [29] TIAN S, CHEN J. Hydroisomerization of n-dodecane on a new kind of bifunctional catalyst: Nickel phosphide supported on SAPO-11 molecular sieve[J]. Fuel Process Technol,2014,122:120−128. doi: 10.1016/j.fuproc.2014.01.031 [30] KINGER G, VINEK H. n-Nonane hydroconversion on Ni and Pt containing HMFI, HMOR and HBEA[J]. Appl Catal A: Gen,2001,218(1):139−149. [31] MENG J, CUI T, BAI D, LI C, CHEN X, LIANG C. Excellent catalytic performance over hierarchical ZSM-48 zeolite: Cooperative effects of enhanced mesoporosity and highly-accessible acidity[J]. Fuel,2022,324:124589. [32] 宁强, 刘粟侥, 张怀科, 陈志强, 任杰. 金属前躯体溶剂对Pt/ZSM-22催化剂临氢异构性能的影响[J]. 燃料化学学报,2018,46(12):1454−1461. doi: 10.3969/j.issn.0253-2409.2018.12.006NING Qiang, LIU Su-yao, ZHANG Huai-ke, CHEN Zhi-qiang, REN Jie. Effect of metal precursor solvent on n-dodecane isomerization of Pt/ZSM-22[J]. J Fuel Chem Technol,2018,46(12):1454−1461. doi: 10.3969/j.issn.0253-2409.2018.12.006 [33] 宋成业, 孟记朋, 李闯, 帕哈尔·泽耀东, 梁长海. ZSM-22/ZSM-23共晶分子筛的合成及其正十六烷加氢异构催化性能(英文)[J]. 燃料化学学报,2021,49(5):712−726. doi: 10.1016/S1872-5813(21)60061-0SONG Cheng-ye, MENG Ji-peng, LI Chuang, ZEYAODONG P, LIANG Chang-hai. Synthesis of ZSM-22/ZSM-23 intergrowth zeolite as the catalyst support for hydroisomerization of n-hexadecane[J]. J Fuel Chem Technol,2021,49(5):712−726. doi: 10.1016/S1872-5813(21)60061-0 [34] WEI C, ZHANG G, ZHAO L, GAO J, XU C. Effect of metal-acid balance and textual modifications on hydroisomerization catalysts for n-alkanes with different chain length: A mini-review[J]. Fuel,2021,315:122809. -





 下载:
下载: