Study on aldol condensation of acetic acid with formaldehyde to acrylic acid over TiO2 modified VPO
-
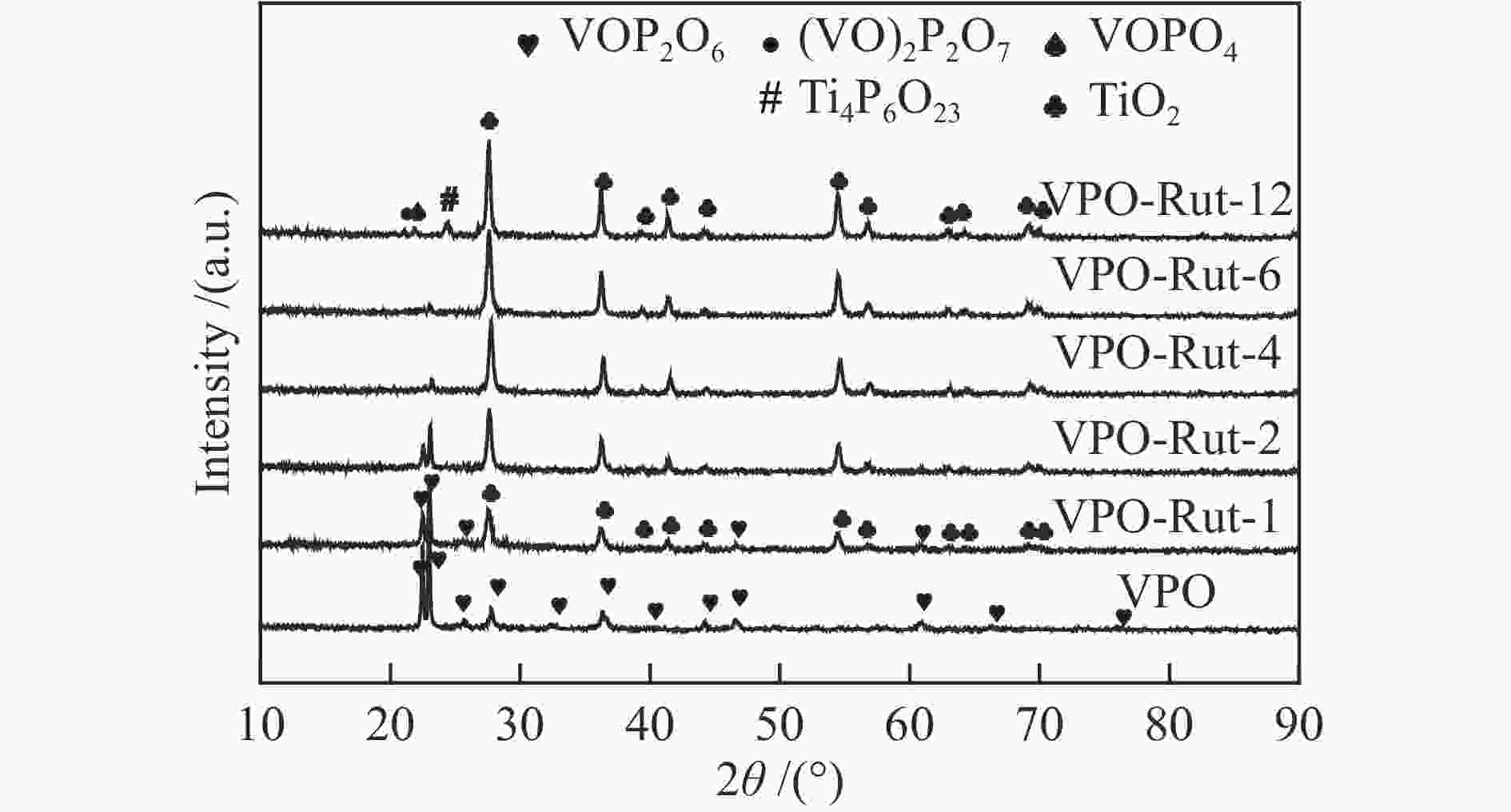
摘要: 采用有机溶剂热法制备了系列TiO2改性的钒磷氧催化剂,利用TEM、XRD、XPS、NH3-TPD及CO2-TPD对催化剂结构及表面物化性质进行了表征,以醋酸甲醛合成丙烯酸为探针反应对催化剂的反应性能进行了评价。结果表明,与未改性的钒磷氧催化剂相比,TiO2的加入显著改变钒磷氧催化剂的(
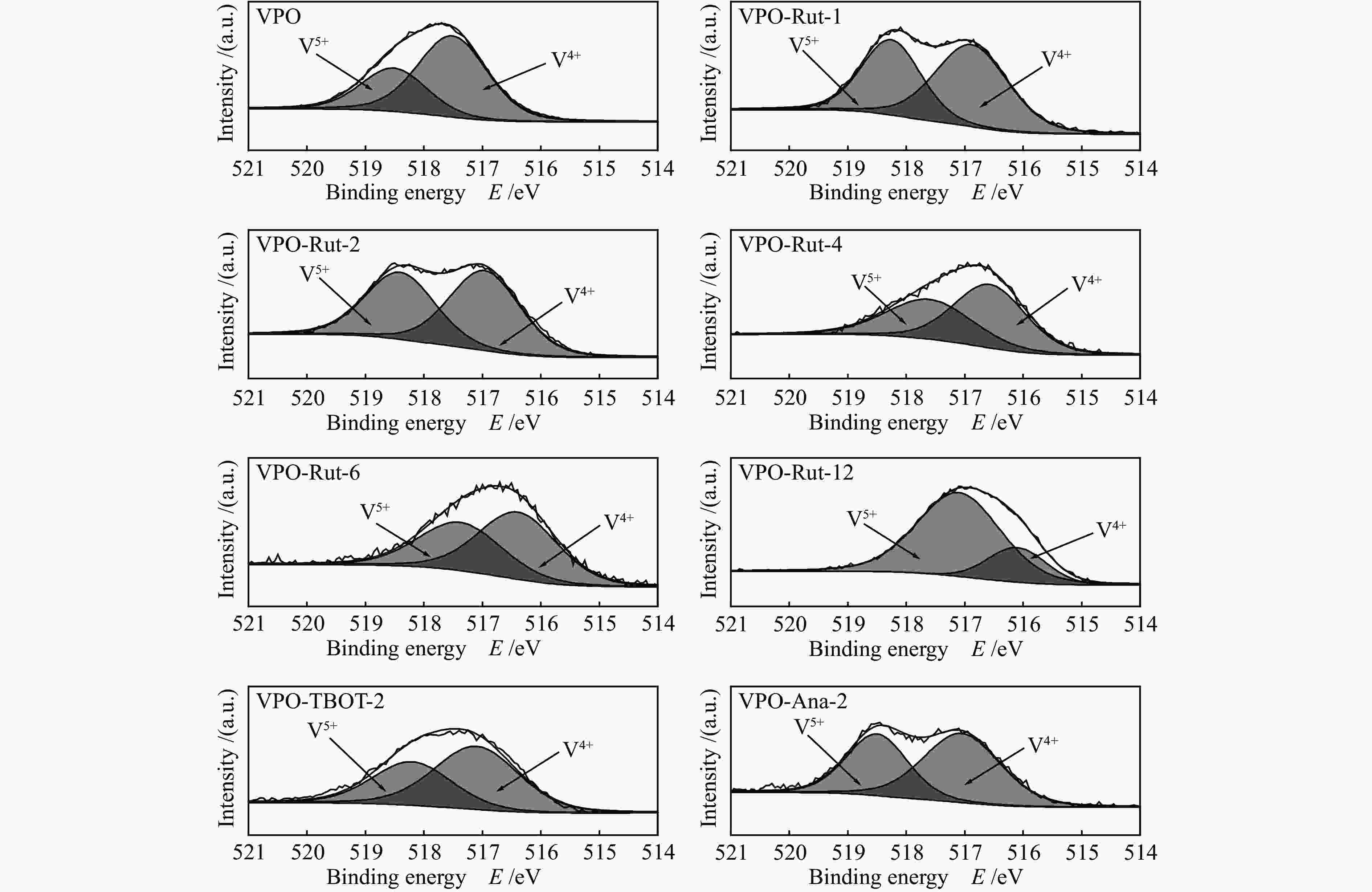
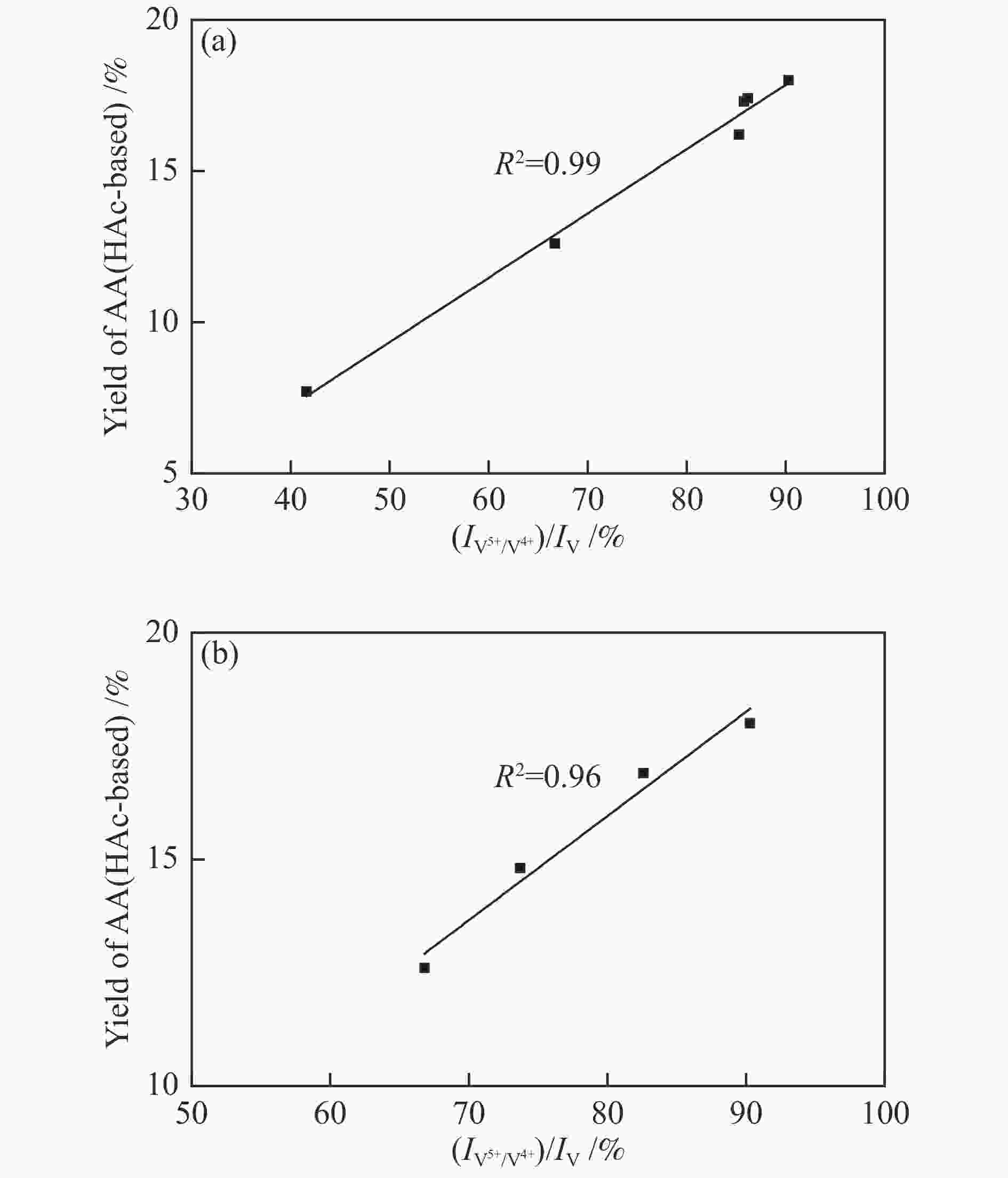
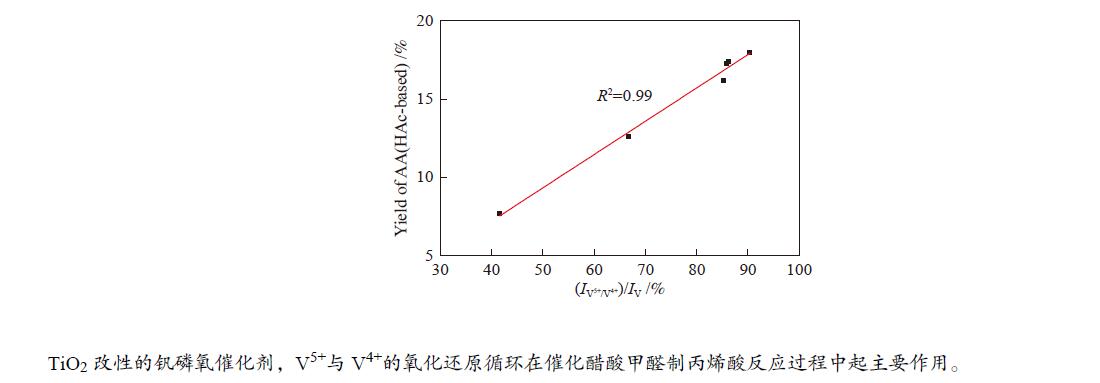
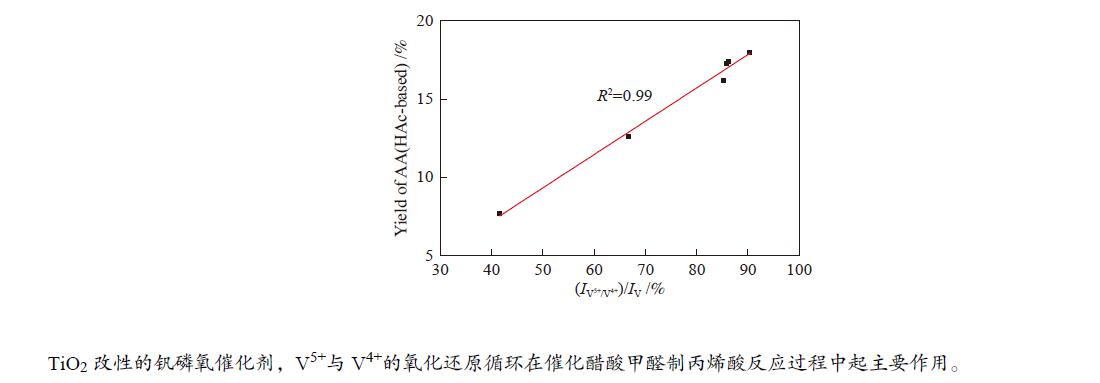
${I_{{{\rm{V}}^{{\rm{5 + }}}}/{{\rm{V}}^{{\rm{4 + }}}}}} $ )/IV比值,当TiO2的前驱体为金红石相并且Ti/V物质的量比为2.0时,(${I_{{{\rm{V}}^{{\rm{5 + }}}}/{{\rm{V}}^{{\rm{4 + }}}}}} $ )/IV比值达到最高,因此,该催化剂具有最高的丙烯酸收率(18.0%)和丙烯酸生成速率(6.61 mmol/(g·h)),表明对于TiO2改性的钒磷氧催化剂,V5+与V4+的氧化还原循环在催化醋酸甲醛制丙烯酸反应过程中起主要作用。-
关键词:
- TiO2改性钒磷氧催化剂 /
- 羟醛缩合 /
- 丙烯酸
Abstract: A class of TiO2 modified VPO catalysts were prepared by organic solvent-heating method in the present work. The catalysts were characterized by TEM, XRD, XPS, NH3-TPD and CO2-TPD techniques. The catalytic performances were evaluated in the aldol condensation of acetate acid and formaldehyde to acrylic acid in a fixed-bed reactor. The results show that the addition of TiO2 significantly changes the proportion of V content forming V5+ and V4+ ion pair to total V with the unmodified VPO catalyst. As the precursor of TiO2 is rutile phase and Ti/V molar ratio is 2.0, the proportion of V content forming V5+ and V4+ ion pair to total V reaches the maximum, resulting in the best acrylic acid yield (18.0%) and acrylic acid space-time yield (6.61 mmol/(g·h)). It indicates that for the VPO catalyst modified by TiO2, the redox cycle of V5+ and V4+ ion pair plays a major role in the catalytic reaction of formaldehyde and acetate acid to acrylic acid.-
Key words:
- TiO2 modified VPO catalysts /
- aldol condensation /
- acrylic acid
-
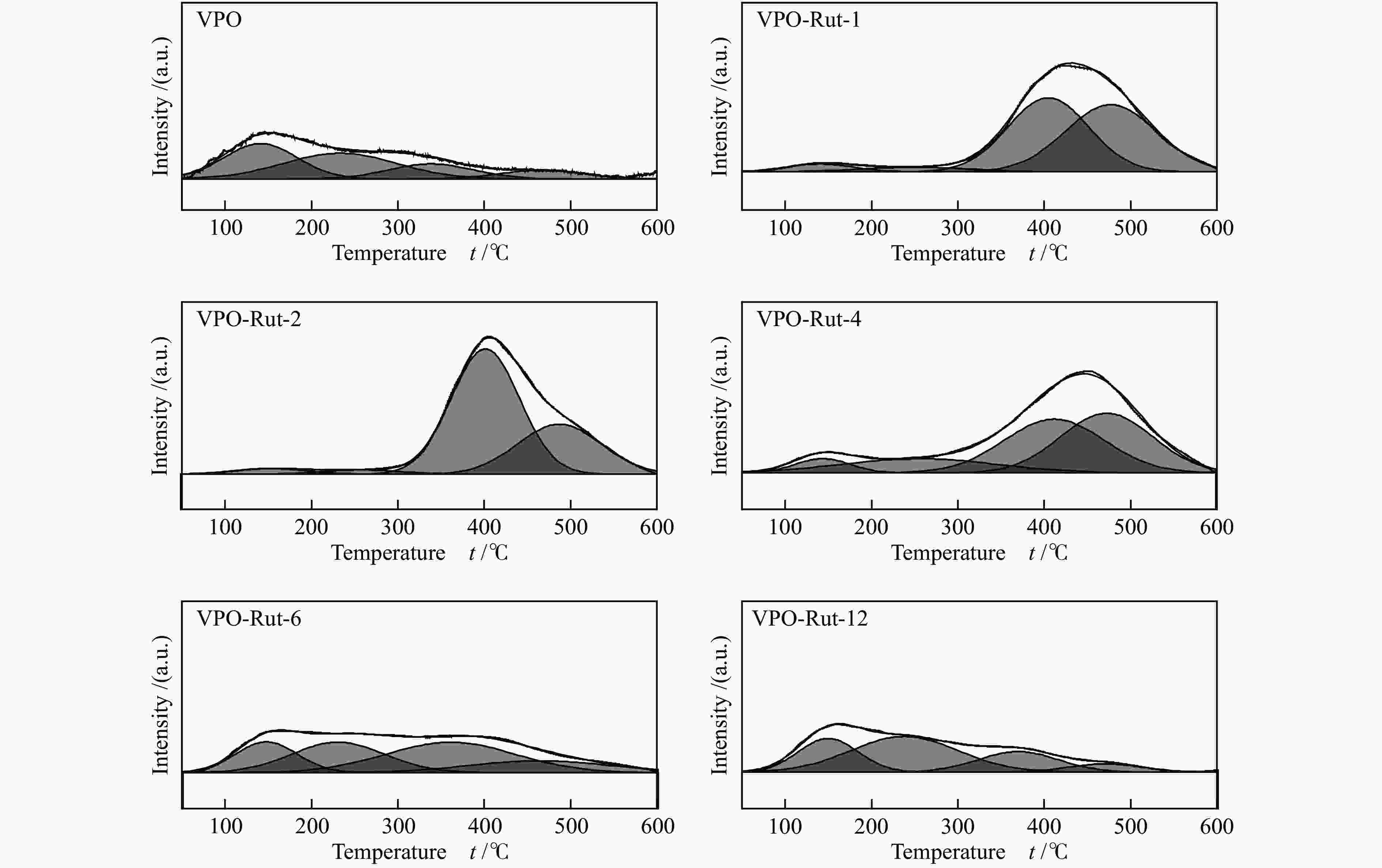
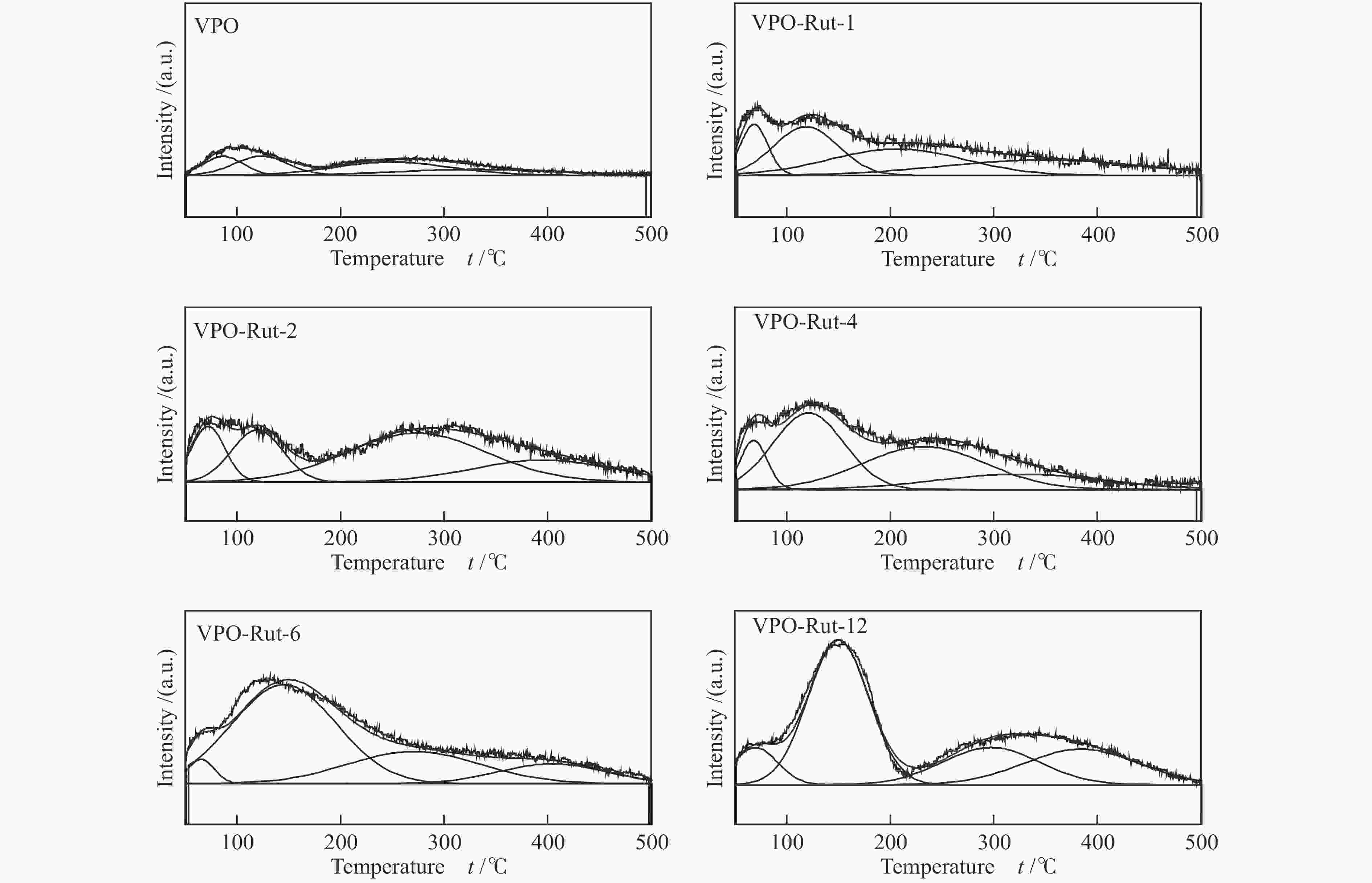
图 9 催化剂(
${I_{{{\rm{V}}^{{\rm{5 + }}}}/{{\rm{V}}^{{\rm{4 + }}}}}} $ )/IV比值与其催化性能的关系Figure 9 Relationship between proportion of V content forming V5+ and V4+ ion pair to total V on the catalyst surface and the reaction performances
(a): VPO catalysts with different TiO2 content; (b): VPO catalysts with different TiO2 precursors
表 1 催化剂的表面组成
Table 1 Surface composition of catalysts
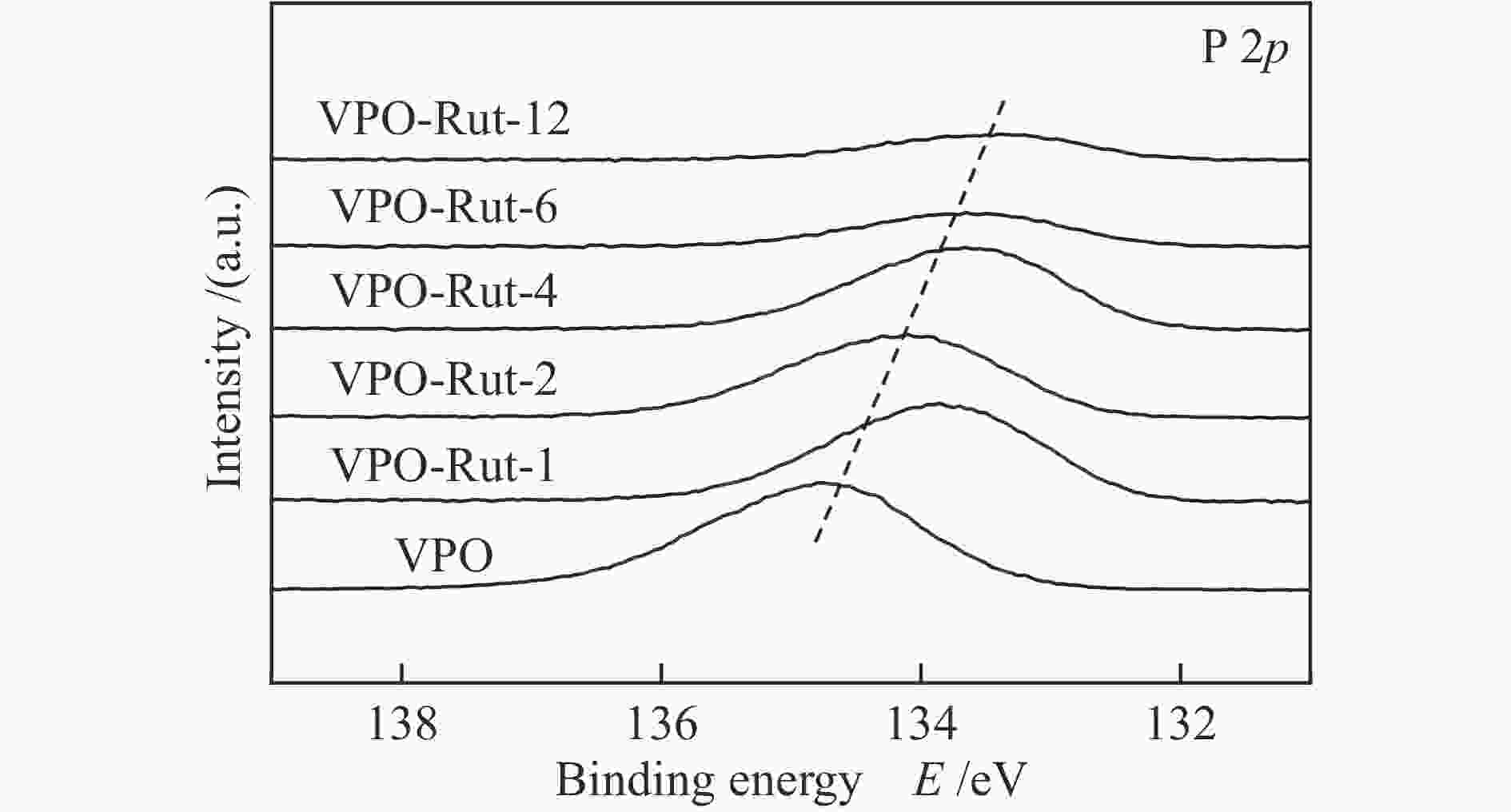
Sample Relative amount / % P/V (mole ratio) Ti/V (mole ratio) (${I_{{{\rm{V}}^{{\rm{5 + }}}}/{{\rm{V}}^{{\rm{4 + }}}}}} $)/IV Binding energy/eV P V5+ V4+ Ti O P V5+ V4+ VPO 17.4 1.7 3.4 0 77.5 3.4 0 66.8% 134.9 518.5 517.5 VPO-Rut-1 14.1 1.6 2.1 3.1 79.0 3.7 0.81 85.3% 133.9 518.3 516.9 VPO-Rut-2 10.9 1.3 1.5 4.4 82.0 3.9 1.5 90.3% 134.2 518.4 517.0 VPO-Rut-4 10.4 1.0 1.3 10.2 77.2 4.6 4.5 85.8% 133.8 517.6 516.6 VPO-Rut-6 5.2 0.8 1.0 10.0 82.9 2.7 5.3 86.2% 133.7 517.4 516.4 VPO-Rut-12 2.4 2.4 0.6 12.3 82.2 0.78 4.0 41.6% 133.5 517.1 516.1 VPO-TBOT-2 15.8 0.8 1.5 5..8 76.0 6.5 2.4 75.6% 134.3 518.4 517.1 VPO-Ana-2 15.0 0.7 1.0 6.8 76.5 8.6 3.9 82.6% 134.3 518.5 517.1 (${I_{{{\rm{V}}^{{\rm{5 + }}}}/{{\rm{V}}^{{\rm{4 + }}}}}} $)/IV: Proportion of V content forming V5+ and V4+ ion pair to total V 表 2 催化剂的表面酸碱性
Table 2 Surface acidity and basicity of catalysts
Sample NH3-TPD Total acid quantities /
(μmol·g−1)CO2-TPD Total basic
quantities /
(μmol·g−1)weak acid
quantities /
(μmol·g−1)intermediate
acid quantities /
(μmol·g−1)weak basic
quantities /
(μmol·g−1)intermediate basic
quantities /
(μmol·g−1)VPO 7.9 10.3 18.2 10.8 12.1 22.9 VPO-Rut-1 42.1 630.6 672.6 49.3 37.8 87.1 VPO-Rut-2 102.4 1910.9 2013.3 60.2 56.5 116.7 VPO-Rut-4 98.5 478.5 576.9 46.4 72.0 118.4 VPO-Rut-6 150.2 204.9 355.2 132.3 105.3 237.6 VPO-Rut-12 172.5 110.3 282.8 159.4 129.8 289.2 表 3 催化剂的反应性能
Table 3 Reaction performances of catalysts
Sample Conv. of HAc /% Selec. of AA /% Yield of AA /% STY /(mmol·g−1·min−1 ) VPO 15.1 83.0 12.5 4.62 VPO-Rut-1 19.5 83.0 16.2 5.95 VPO-Rut-2 21.1 85.4 18.0 6.61 VPO-Rut-4 21.0 82.1 17.3 6.34 VPO-Rut-6 20.6 84.6 17.4 6.39 VPO-Rut-12 11.8 65.1 7.7 2.81 VPO-TBOT-2 18.6 79.7 14.8 5.43 VPO-Ana-2 20.1 83.8 16.9 6.24 the reaction condition: amount of catalysts 6 g; HAc/HCHO = 3/1; volume of feedstock 0.25 mL/min; LHSV is 2.5 h−1 ; reaction temperature 365 ℃ (HAc, acetic acid; AA, acrylic acid ) 表 4 温度对催化剂反应性能的影响
Table 4 Effect of reaction temperature on the catalytic performance
Temp. /℃ Conv. of HAc /% Selec. of AA /% Yield of AA /% STY /
(mmol·g−1·min−1 )320 17.2 81.4 14.0 5.14 350 20.9 84.1 17.5 6.44 365 21.1 85.4 18.0 6.61 380 15.8 80.5 12.7 4.65 reaction condition: amount of catalysts 6 g; HAc/HCHO = 3∶1; volume of feedstock 0.25 mL/min; LHSV is 2.5 h−1; (HAc, acetic acid; AA, acrylic acid ) 表 5 液空对催化剂的反应性能的影响
Table 5 Effect of LHSV on the catalytic performance
LHSV /h−1 Conv. of HAc /% Selec. of AA /% Yield of AA /% STY / (mmol·g−1·min−1 ) 1.0 25.7 80.9 20.8 3.04 2.5 21.1 85.4 18.0 6.61 4.0 14.7 72.3 10.7 6.26 reaction condition: amount of catalysts 6 g; HAc/HCHO = 3∶1; reaction temperature 365 ℃ (HAc, acetic acid; AA, acrylic acid) -
[1] BAILEY O H, MONTAG R A, YOO J S. Methacrylic acid synthesis. Part 1. Condensation of propionic acid with formaldehyde over alkali metal cation on silica catalysts[J]. Appl Catal A: Gen,1992,88(2):163−177. doi: 10.1016/0926-860X(92)80213-V [2] ANASTAS P T, WARNER J C. (1998) Green Chemistry: Theory and Practice[M]. New York: Oxford University Press. [3] ZUO C C, LI Y P, LI C S, CAO S S, YAO H Y, ZHANG S J. Thermodynamics and separation process for quaternary acrylic systems[J]. AICHE J,2016,62(1):228−240. doi: 10.1002/aic.15015 [4] GUO X, YANG D, ZUO C, PENG Z, LI C, ZHANG S. Catalysts, process optimization, and kinetics for the production of methyl acrylate over vanadium phosphorus oxide catalysts[J]. Ind Eng Chem Res,2017,56(20):5860−5871. doi: 10.1021/acs.iecr.7b01212 [5] 朱俊杰, 王志遵, 李常青, 商红岩. 改性聚丙烯酸高级酯的合成及其降凝效果的考察[J]. 燃料化学学报,2002,30(4):328−331. doi: 10.3969/j.issn.0253-2409.2002.04.008ZHU Jun-jie, WANG Zhi-zun, LI Chang-qing, SHANG Hong-yan. Synthesis and evaluation of MPAE as pour point depressant for diesels[J]. J Fuel Chem Technol,2002,30(4):328−331. doi: 10.3969/j.issn.0253-2409.2002.04.008 [6] KADAR J, HEENE-WUERL N, HAHN S, NAGENGAST J, KEHRER M, TACCARDI N, COLLIAS D, DZIEZOK P, WASSERSCHEID P, ALBERT J. Acrylic acid synthesis from lactide in a continuous liquid-phase process[J]. ACS Sustainable Chem Eng,2019,7(7):7140−7147. doi: 10.1021/acssuschemeng.8b06538 [7] BOTELLA P, NIETO J M L, SOLSONA B, MIFSUD A, MARQUEZ F. The preparation, characterization, and catalytic behavior of MoVTeNbO catalysts prepared by hydrothermal synthesis[J]. J Catal,2002,209(2):445−455. doi: 10.1006/jcat.2002.3648 [8] BU Y J, SANG S K, SANG H M. Performance of WOx-added Mo-V-Te-Nb-O catalysts in the partial oxidation of propane to acrylic acid[J]. Appl Catal A: Gen,2010,378(1):76−82. doi: 10.1016/j.apcata.2010.02.002 [9] WEN F, GE Q J, YU J F, XU H Y. Catalytic selective oxidation of propane to acrylic acid in a fixed-bed reactor with an O2-distributor[J]. Ind Eng Chem Res,2011,50(4):1962−1967. doi: 10.1021/ie1018386 [10] MA Z L, MA X G, LIU H C, HE Y L, ZHU W L, GUO X W, LIU Z M. A green route to methyl acrylate and acrylic acid by an aldol condensation reaction over H-ZSM-35 zeolite catalysts[J]. Chem Commun,2017,53(65):9071−9074. doi: 10.1039/C7CC04574C [11] YANG D, WANG G, WU H, GUO X, ZHANG S, LI Z, LI C. Deactivation behavior on VPO and VPO-Zr catalysts in the aldol condensation of methyl acetate and formaldehyde[J]. Catal Today,2018,316:122−128. doi: 10.1016/j.cattod.2018.06.019 [12] ZUO C, GE T, WANG G, GUO X, LI C, ZHANG S. Enhanced catalytic activity with oxygen for methyl acrylate production via cross-aldol condensation reaction[J]. Chem Eng Technol,2018,41(7):1331−1341. doi: 10.1002/ceat.201700500 [13] LIU J, WANG P, FENG Y, XU Z, FENG X, JI W, AU C-T. Precisely phase-modulated VPO catalysts with enhanced inter-phase conjunction for acrylic acid production through the condensation of acetic acid and formaldehyde[J]. J Catal,2019,374:171−182. doi: 10.1016/j.jcat.2019.04.032 [14] LIU J, XU P, WANG P, XU Z, FENG X, JI W, AU C-T. Vanadium phosphorus oxide/siliceous mesostructured cellular foams: Efficient and selective for sustainable acrylic acid production via condensation route[J]. Sci Rep,2019,9(1):1−10. [15] LIU J, WANG P, XU P, XU Z, FENG X, JI W, ARANDIYAN H, AU C-T. How to achieve a highly selective yet simply available vanadium phosphorus oxide-based catalyst for sustainable acrylic acid production via acetic acid-formaldehyde condensation[J]. Chem Commun,2020,56(7):1022−1025. doi: 10.1039/C9CC08700A [16] AI M. Vapor-phase aldol condensation of formaldehyde with acetic acid on V2O5P2O5 catalysts[J]. J Catal,1987,107(1):201−208. doi: 10.1016/0021-9517(87)90285-5 [17] FENG X, SUN B, YAO Y, SU Q, JI W, AU C-T. Renewable production of acrylic acid and its derivative: New insights into the aldol condensation route over the vanadium phosphorus oxides[J]. J Catal,2014,314:132−141. doi: 10.1016/j.jcat.2014.04.005 [18] YANG D, SARARUK C, SUZUKI K, LI Z, LI C. Effect of calcination temperature on the catalytic activity of VPO for aldol condensation of acetic acid and formalin[J]. Chem Eng J,2016,300:160−168. doi: 10.1016/j.cej.2016.04.107 [19] HE T, QU Y, WANG J. Experimental and theoretical study for vapor phase aldol condensation of methyl acetate and formaldehyde over alkali metal oxides supported on SBA-15[J]. Ind Eng Chem Res,2018,57(8):2773−2786. doi: 10.1021/acs.iecr.7b02841 [20] ZUO C, GE T, GUO X, LI C, ZHANG S. Synthesis and catalytic performance of Cs/P modified ZSM-5 zeolite in aldol condensation of methyl acetate with different sources of formaldehyde[J]. Microporous Mesoporous Mater,2018,256:58−66. doi: 10.1016/j.micromeso.2017.07.045 [21] ZHAO H, ZUO C, YANG D, LI C, ZHANG S. Effects of support for vanadium phosphorus oxide catalysts on vapor-phase aldol condensation of methyl acetate with formaldehyde[J]. Ind Eng Chem Res,2016,55(50):12693−12702. doi: 10.1021/acs.iecr.6b03079 [22] WANG Y, WANG Z, HAO X, ZHANG W, ZHU W. Nb-Doped vanadium phosphorus oxide catalyst for the aldol condensation of acetic acid with formaldehyde to acrylic acid[J]. Ind Eng Chem Res,2018,57(36):12055−12060. doi: 10.1021/acs.iecr.8b02132 [23] YANG D, SARARUK C, WANG H, ZHANG S, LI Z, LI C. Effect of metal ion in bulk VPO in aldol condensation of formaldehyde and methyl acetate to methyl acrylate[J]. Ind Eng Chem Res,2018,57(1):93−100. doi: 10.1021/acs.iecr.7b03521 [24] WANG A, HU J, YIN H, LU Z, XUE W, SHEN L, LIU S. Aldol condensation of acetic acid with formaldehyde to acrylic acid over Cs(Ce, Nd) VPO/SiO2 catalyst[J]. Rsc Adv,2017,7(76):48475−48485. doi: 10.1039/C7RA09778F [25] VAN, VEEN, ANDRE, C. , MUELLER, SEBASTIAN, LIU, YUE, SUN, XIANYONG. Coke formation and deactivation pathways on H-ZSM-5 in the conversion of methanol to olefins[J]. J Catal,2015,325:48−59. doi: 10.1016/j.jcat.2015.02.013 -





 下载:
下载: