Study on wet air oxidation of phenol catalyzed by LaFeO3 perovskite
-
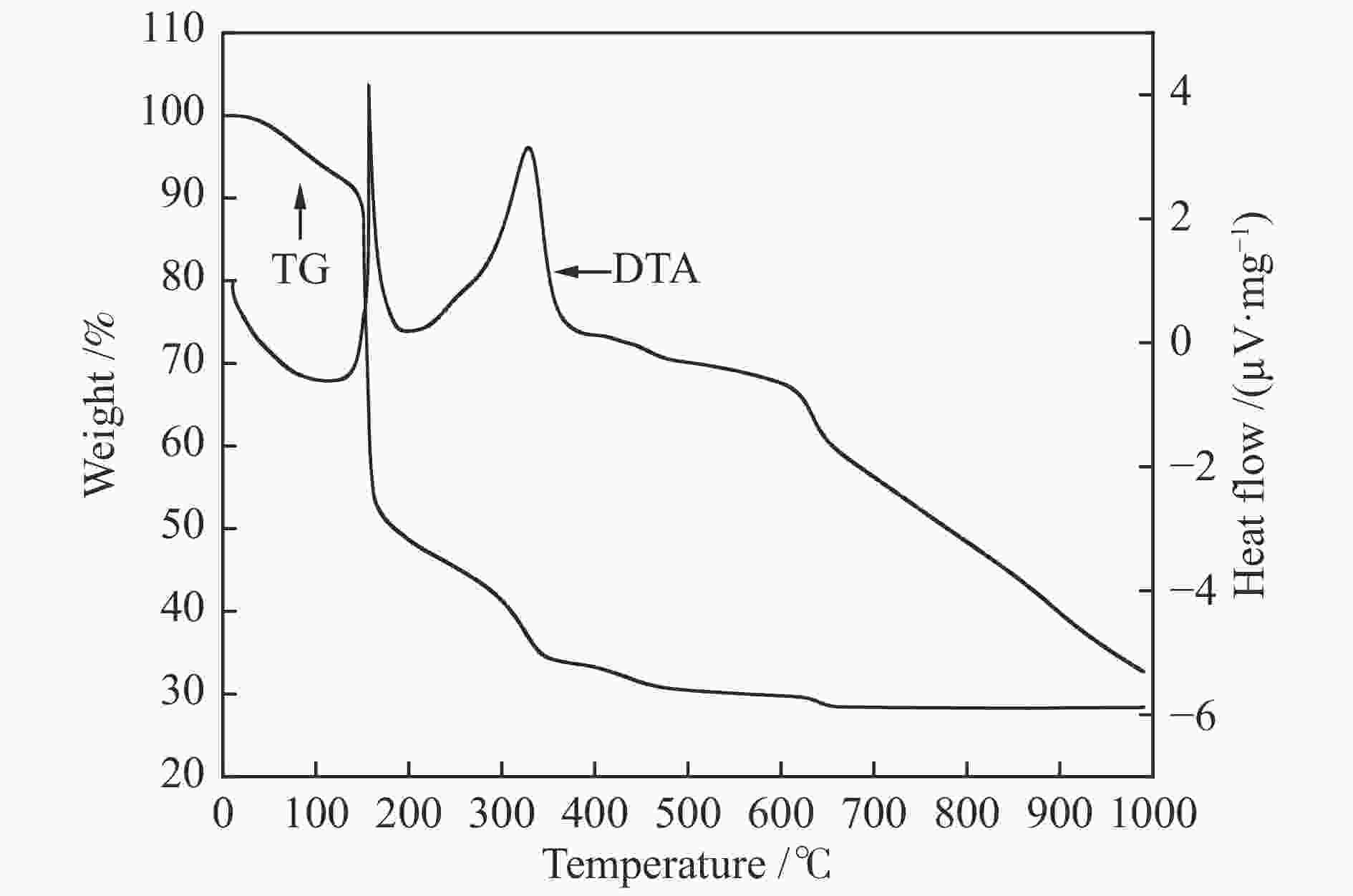
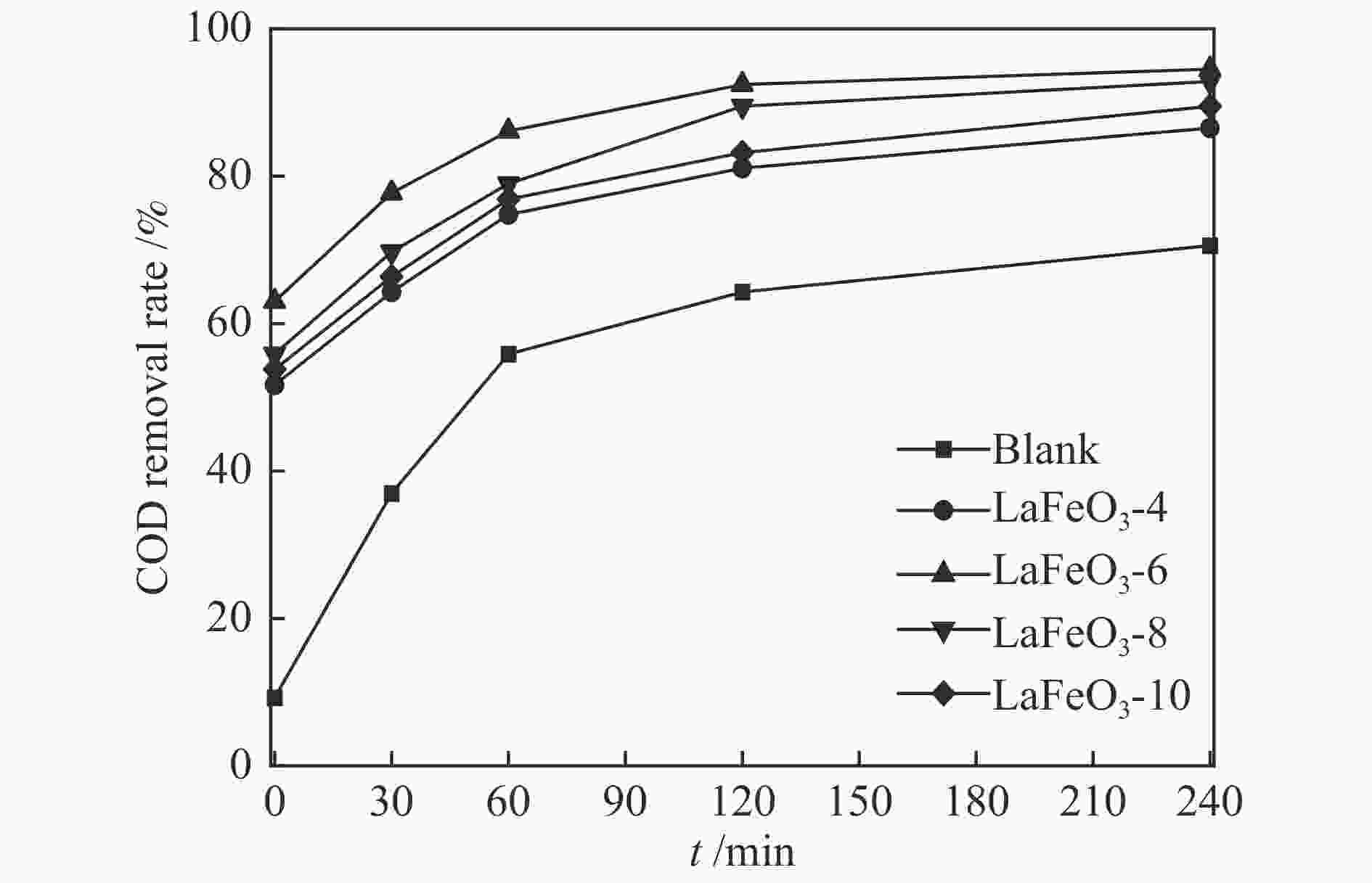
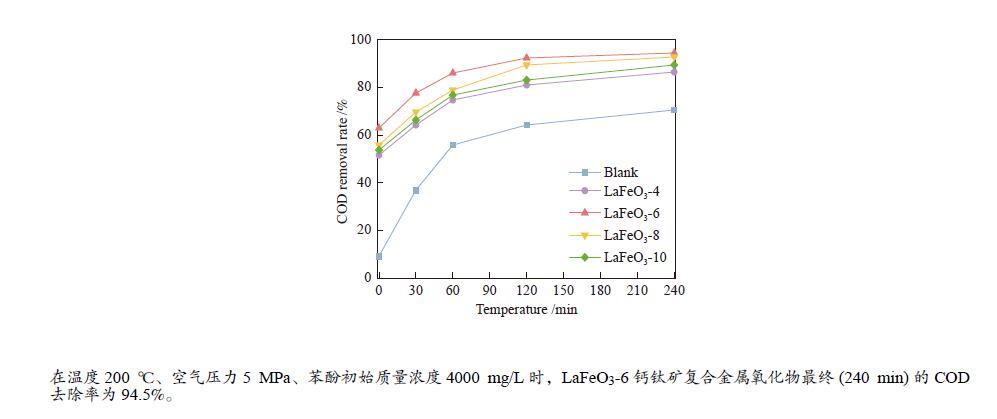
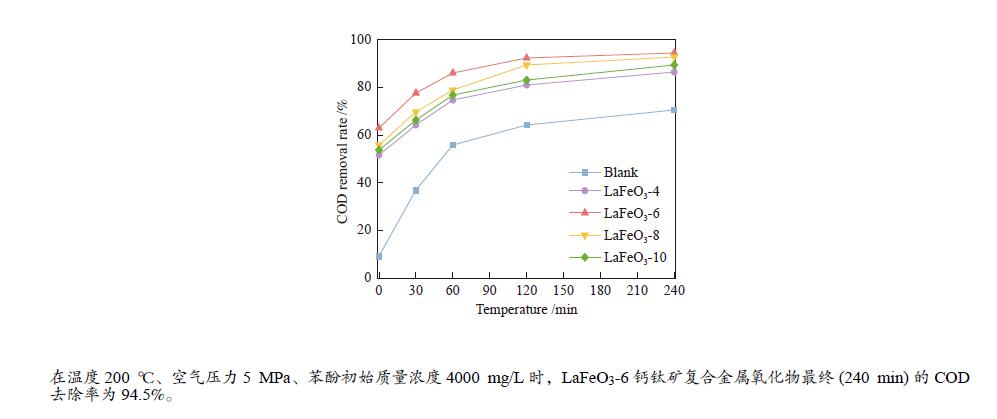
摘要: 本研究采用固相球磨法制备了四种不同球磨时间的LaFeO3钙钛矿催化材料,并将其用于催化湿式氧化苯酚反应,探讨了球磨时间对LaFeO3钙钛矿催化剂催化湿式氧化苯酚水溶液性能的影响。采用XRD、H2-TPR、TG-DTA、FT-IR、N2物理吸附、XPS等手段对LaFeO3钙钛矿催化材料进行了表征。结果表明,在反应温度为200 ℃、空气压力5 MPa、苯酚初始浓度4000 mg/L时,经过240 min的催化氧化反应后LaFeO3-6催化材料最终的COD去除率为94.5%。Abstract: In this paper, four kinds of LaFeO3 perovskite catalytic materials with different ball milling time were prepared by solid phase milling method and used to catalyze the wet air oxidation of phenol. The effect of milling time on the performance of LaFeO3 perovskite catalyst for wet air oxidation of phenol aqueous solution was investigated. LaFeO3 perovskite catalytic materials were characterized by XRD, H2-TPR, TG-DTA, FT-IR, N2 physical adsorption and XPS. The results show that when the reaction temperature is 200 ℃, the air pressure is 5 MPa and the initial concentration of phenol is 4000 mg/L, the final COD removal rate of LaFeO3-6 catalytic material is 94.5% after catalytic oxidation of 240 min.
-
Key words:
- phenol /
- LaFeO3 perovskite /
- catalytic wet air oxidation /
- COD removal rate
-
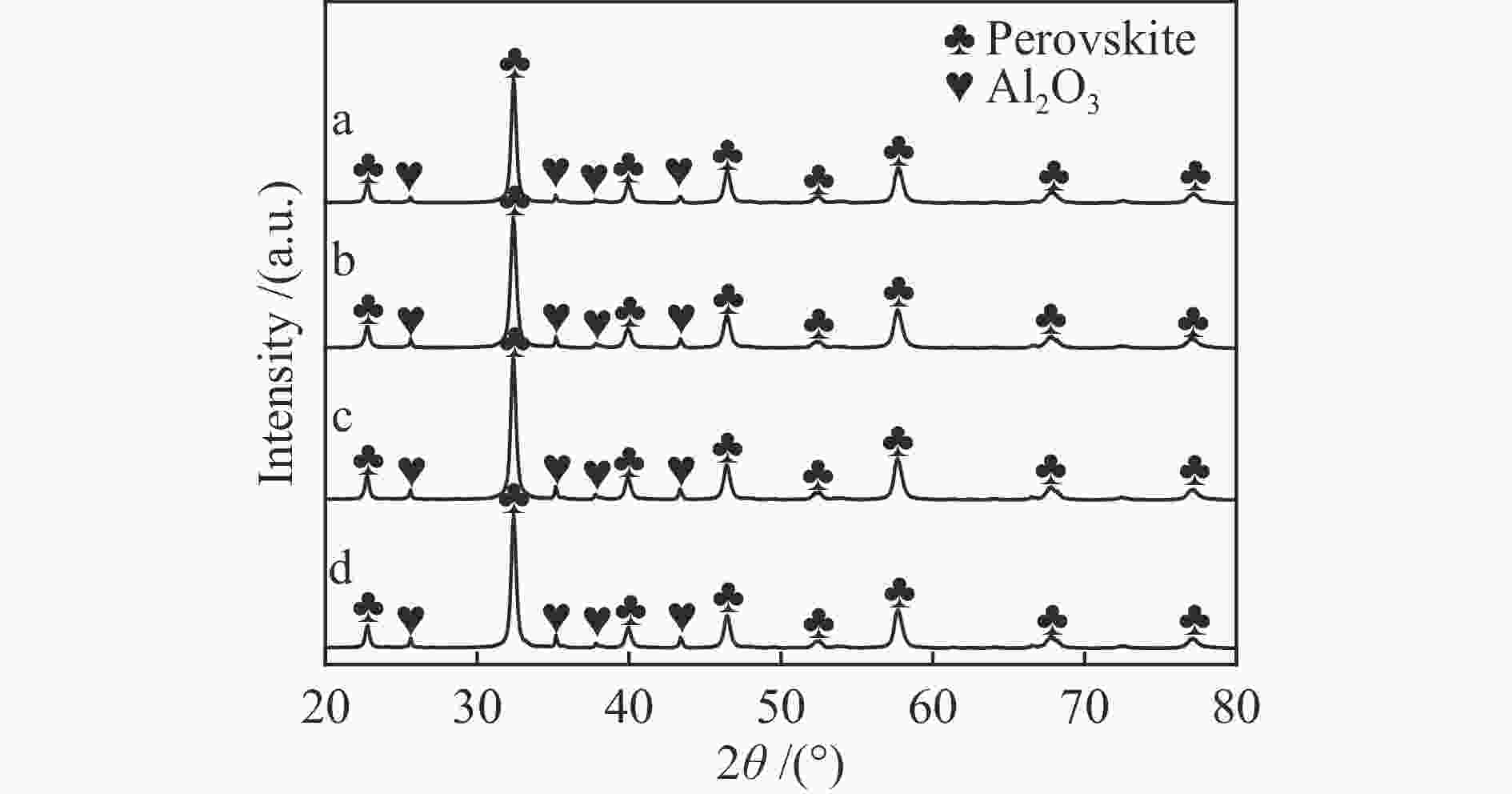
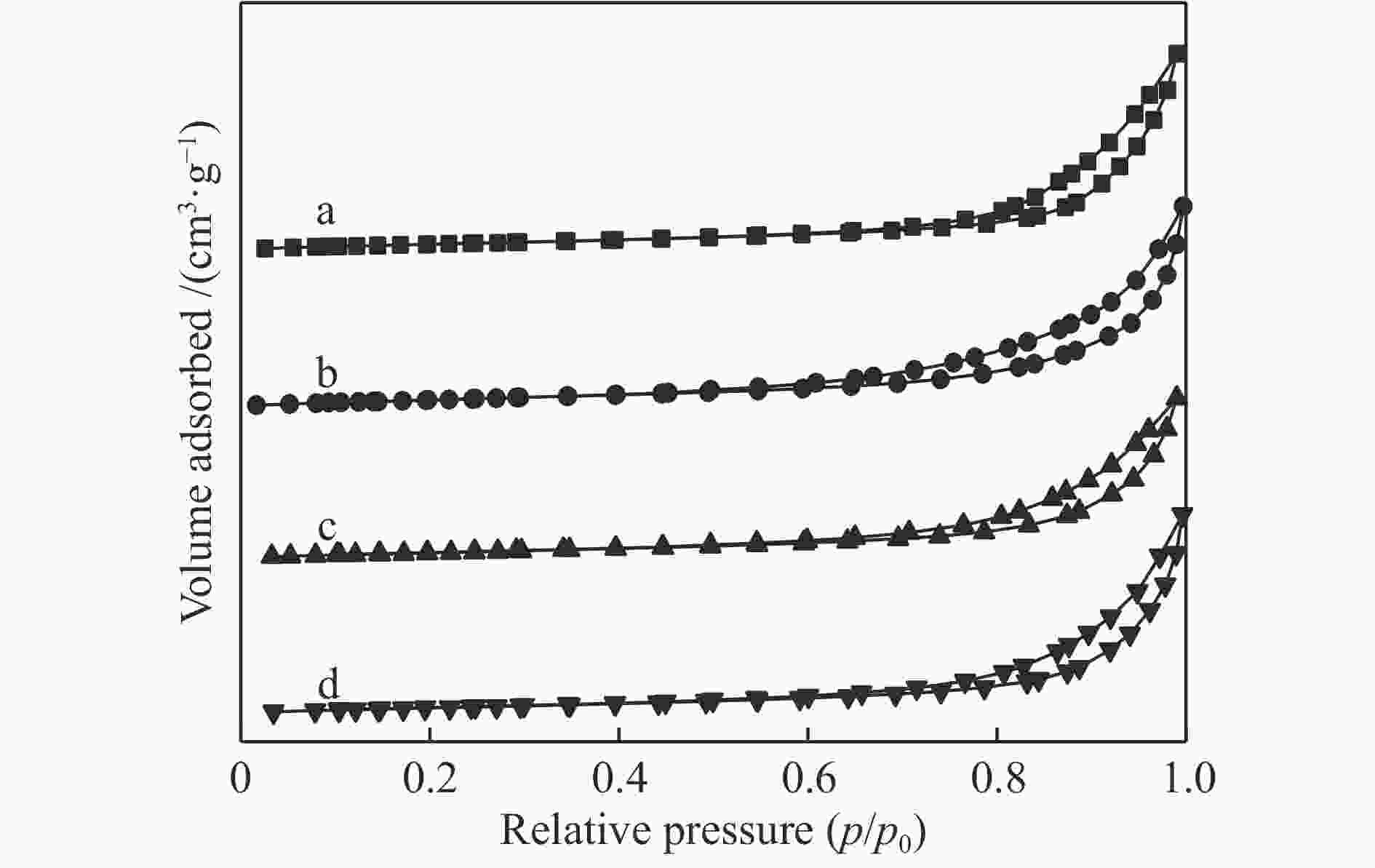
表 1 LaFeO3-t的结构参数表
Table 1 Structural parameters of LaFeO3-t
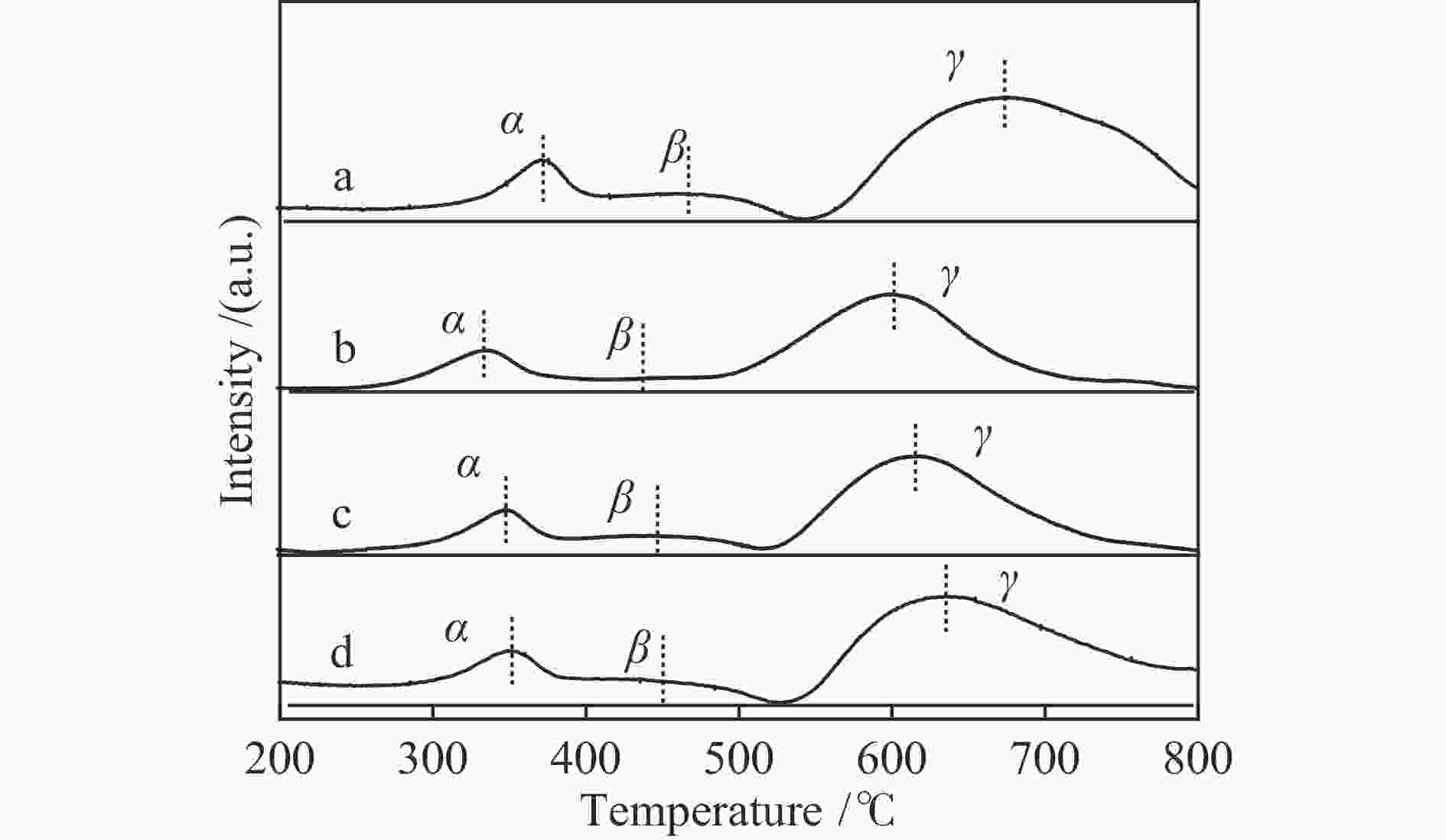
Catalyst LaFeO3-4 LaFeO3-6 LaFeO3-8 LaFeO3-10 Specific surface area /(m2·g−1) 15.8 18.2 14.7 14.7 Pore volume /(cm3·g−1) 0.10 0.08 0.08 0.09 Average crystallite size /nm 20.7 18.9 20.0 20.9 表 2 LaFeO3-t催化材料的还原峰位置
Table 2 Reduction of LaFeO3-t catalyst
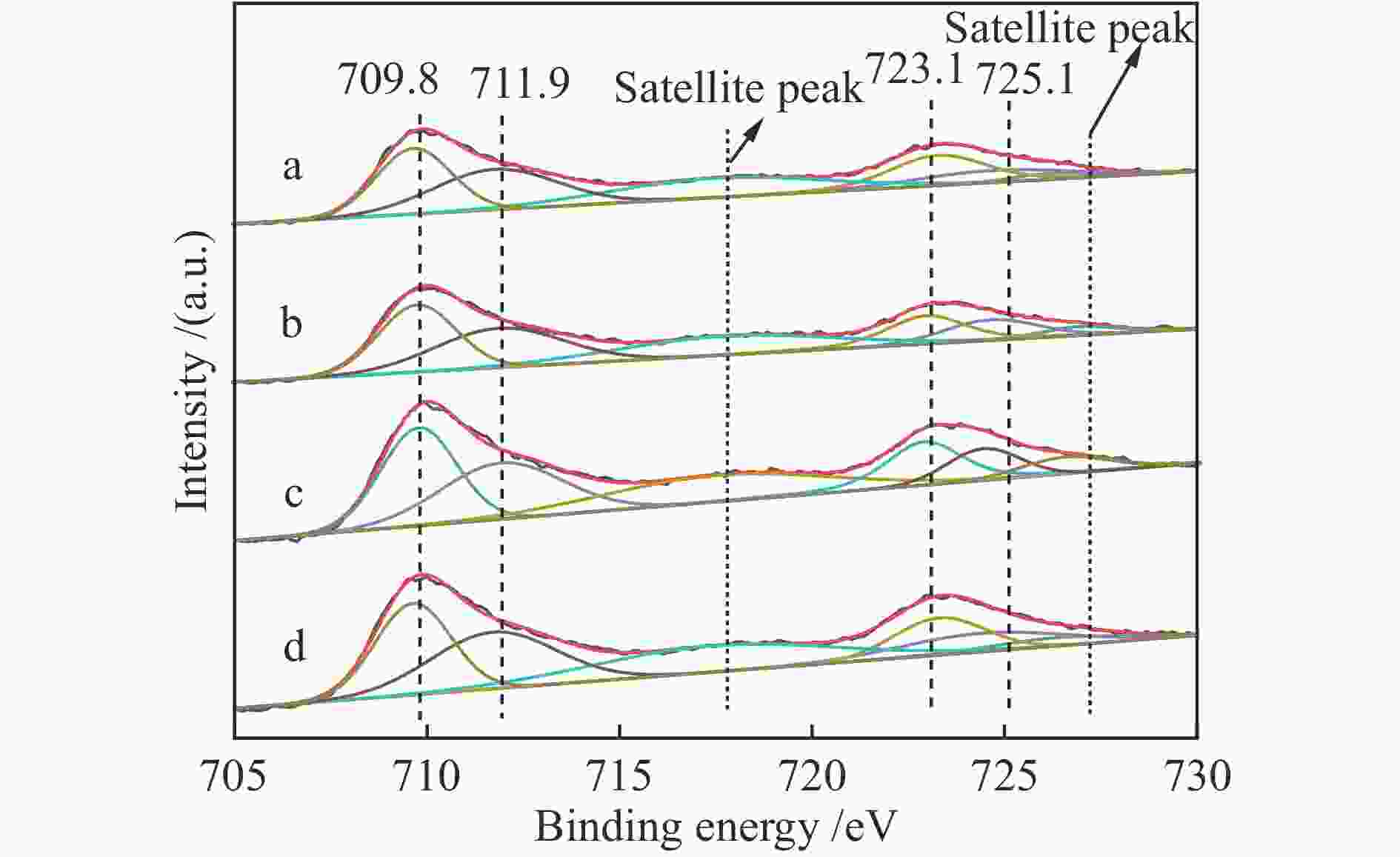
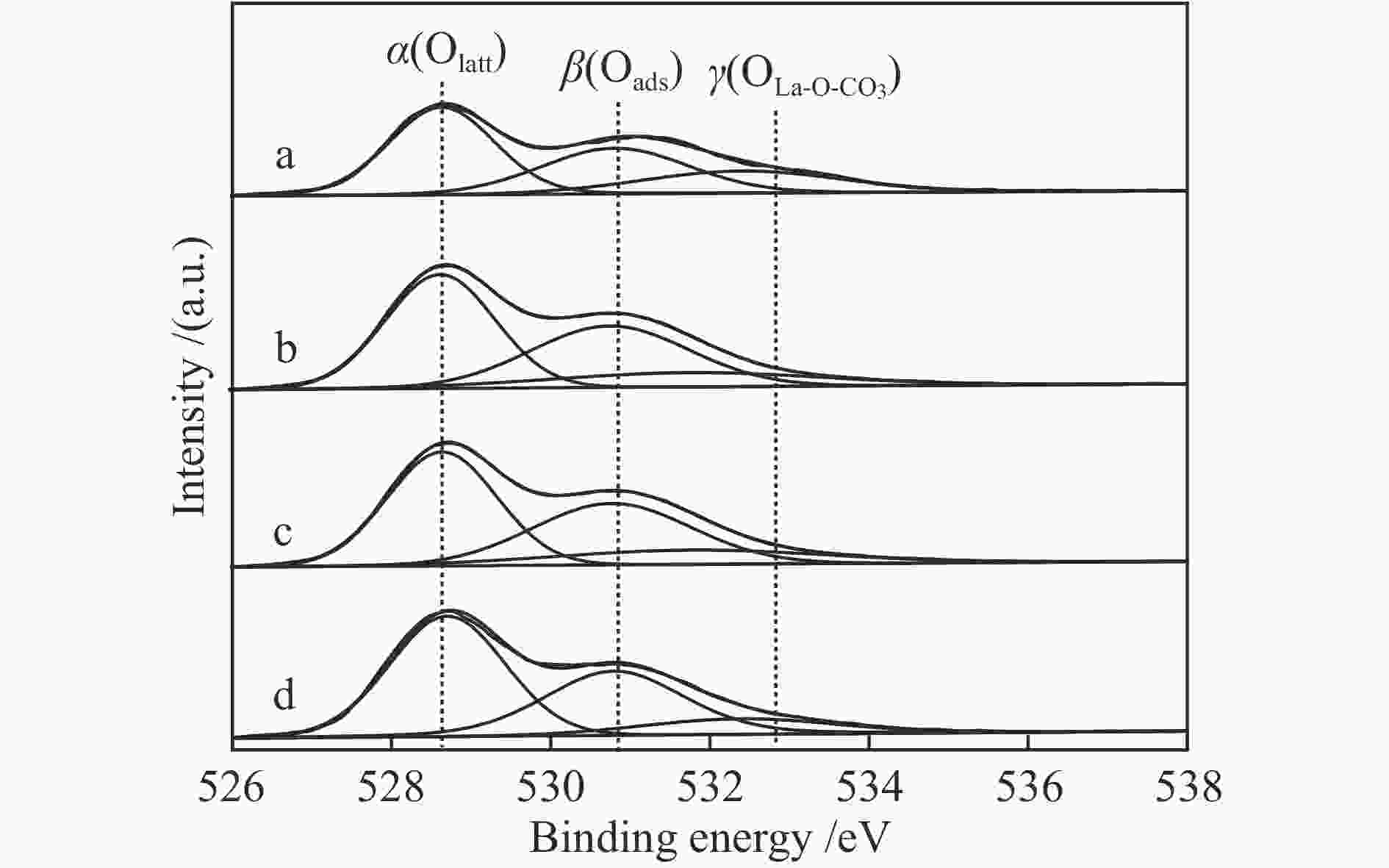
Catalyst Peak temperature t/℃ α peak β peak γ peak LaFeO3-4 371 467 673 LaFeO3-6 333 436 601 LaFeO3-8 351 448 616 LaFeO3-10 353 451 636 表 3 LaFeO3-t催化剂中O 1s XPS曲线拟合
Table 3 O 1s XPS curve-fitting results of LaFeO3-t catalysts
Catalyst Oads/(Oads+OLa-O-CO3+Olatt)(%) LaFeO3-4 37.4 LaFeO3-6 40.6 LaFeO3-8 40.3 LaFeO3-10 39.5 表 4 催化材料的重复使用性
Table 4 Repetition of the catalyst
Reuse time 1 2 3 4 COD removal rate/% 94.5 91.7 88.2 85.8 -
[1] 蒋博龙, 史顺杰, 蒋海林, 封鑫, 孙好芬. 金属有机框架材料吸附处理苯酚污水机理研究进展[J]. 化工进展,2021,40(8):4525−4539.JIANG Bo-long, SHI Shun-jie, JIANG Hai-lin, FENG Xin, SUN Hao-fen. Research progress in phenol adsorption mechanism over metal-organic framework from wastewater[J]. Chem Ind Eng Prog,2021,40(8):4525−4539. [2] 于喜洋, 顾贵洲, 李政. 水滑石材料处理工业废水的研究进展[J]. 石油化工高等学校学报,2021,34(6):16−21. doi: 10.3969/j.issn.1006-396X.2021.06.003YU Xi-yang, GU Gui-zhou, LI Zheng. Research progress of industrial wastewater treatment by hydrotalcite[J]. J Petrochem Univ,2021,34(6):16−21. doi: 10.3969/j.issn.1006-396X.2021.06.003 [3] MA C J, WEN Y Y, YUE Q Q, LI A Q, FU J L, ZHANG N W, GAI H J, ZHENG J B, CHEN B H. Oxygen-vacancy-promoted catalytic wet air oxidation of phenol from MnO2-CeO2[J]. RSC Adv,2017,7(43):27079−27088. doi: 10.1039/C7RA04037G [4] 王建兵, 祝万鹏, 王伟, 杨少霞. 颗粒Ru催化剂催化湿式氧化乙酸和苯酚[J]. 中国环境科学,2007,27(2):179−183.WANG Jian-jun, ZHU Wan-peng, WANG Wei, YANG Shao-xia. Catalytic wet air oxidation of acetic acid and phenol with pelletized ruthenium catalyst Ru/ZrO2-CeO2 catalysts[J]. Chin Environ Sci,2007,27(2):179−183. [5] PLEŞA C R, COTEŢ L C, MĂICĂNEANU A, VASILESCU M, VULPOI A. Preparation, characterization, and testing of metal-doped carbon xerogels as catalyst for phenol CWAO[J]. Environ Sci Pollut Res,2016,24(3):2980−2986. [6] 张宣娇, 孙羽, 刘明, 郝书敏, 杨涛, 张磊, 白金, 韩蛟. CeO2形貌结构对催化湿式空气氧化苯酚性能的影响[J]. 中国环境科学,2020,40(10):4330−4334. doi: 10.3969/j.issn.1000-6923.2020.10.016ZHANG Xuan-jiao, SUN Yu, LIU Ming, HAO Shu-min, YANG Tao, ZHANG Lei, BAI Jin, HAN Jiao. Effect of morphology on the performance of CeO2 for catalytic wet air oxidation of phenol[J]. Chin Environ Sci,2020,40(10):4330−4334. doi: 10.3969/j.issn.1000-6923.2020.10.016 [7] WANG D Z, LUO X Y, YANG S P, XUE G. Tourmaline/Perovskite composite material as heterogeneous catalysts for activation peroxymonosulfate to remove organic dye in water[J]. J Environ Chem Eng,2021,9(54):105221. [8] JIMÉNEZ R, ZAMORE R, PECCHI G, GARCIAGARCIA X, GORDON A L. Effect of Ca-substitution in La1−xCaxFeO3 perovskites on the catalytic activity for soot combustion[J]. Fuel Process Technol,2010,91(5):546−549. doi: 10.1016/j.fuproc.2009.12.017 [9] VIJAYARAGHAVAN T, ALTHAF R, BABU P, PARIDA K M, VADIVEL S, ASHOK A M. Visible light active LaFeO3 nano perovskite-RGO-NiO composite for efficient H2 evolution by photocatalytic water splitting and textile dye degradation[J]. J Environ Chem Eng,2021,9(1):104675. doi: 10.1016/j.jece.2020.104675 [10] WU S H, LIN Y, YANG S P, DU C, TENG Q, MA Y, ZHANG D M, NIE L J, ZHONG Y Y. Enhanced activation of peroxymonosulfte by LaFeO3 perovskite supported on Al2O3 for degradation of organic pollutants[J]. Chemosphere,2019,237:124478. doi: 10.1016/j.chemosphere.2019.124478 [11] SUN W J, LV H X, MA L, TAN X D, JIN C Y, WU H L, CHEN L L, LIU M Y, WEI H Z, SUN C L. Use of catalytic wet air oxidation (CWAO) for pretreatment of high-salinity high-organic wastewater[J]. J Environ Sci,2022,120:105−114. doi: 10.1016/j.jes.2021.06.027 [12] 白金, 丁力, 刘鑫尧, 王新明, 张磊, 韩蛟, 刘道胜, 王香攀. 原位合成CuO/ZnO-Al2O3水滑石衍生催化剂催化湿式空气氧化苯酚[J]. 环境科学学报,2018,38(6):2360−2366.BAI Jin, DING Li, LIU Xin-yao, WANG Xin-ming, ZHANG Lei, HAN Jiao, LIU Dao-sheng, WANG Xiang-pan. In-situ systhesis of CuO /ZnO-Al2O3 catalysts derived from hydrotalcite precursor for catalytic wet air oxidation of phenolic[J]. Acta Sci Circum,2018,38(6):2360−2366. [13] 邓禺南, 陈炜鸣, 罗梓尹, 崔瑜旗, 李启彬. MnO2催化O3处理准好氧矿化垃圾床渗滤液尾水中难降解有机物[J]. 中国环境科学,2018,38(11):132−142.DENG Yu-nan, CHEN Wei-ming, LUO Zi-yin, CUI Yu-qi, LI Qi-bin. Removal of refractory organics from SAARB treated landfill leachate by O3/MnO2 process[J]. Chin Environ Sci,2018,38(11):132−142. [14] 傅潇然. 一种新型在线紫外水质分析仪的研制[D]. 天津: 天津大学, 2008.FU Xiao-Ran. Development of a new kind of on-line ultraviolet spectroscopic water quality analyaer[D]. Tianjin: Tianjin University, 2008. [15] SENNA M, BILLIK P, YERMAKOV A Y, ŠKRÁTEK M, MAJEROVÁ M, ČAPLOVIčOVÁ M, NOSKO M. Synthesis and magnetic properties of CuAlO2 from high-energy ball-milled Cu2O-Al2O3 mixture[J]. J Alloy Compd,2017,695:2314−2323. doi: 10.1016/j.jallcom.2016.11.097 [16] CHAUDHARI V, SHIRSATH S E, MANE M L, KADAM R H, SHELKE S B, MANE D R. Crystallographic, magnetic and electrical properties of Ni0.5Cu0.25Zn0.25LaxFe2−xO4 nanoparticles fabricated by sol-gel method[J]. J Alloy Compd,2013,549:213−220. doi: 10.1016/j.jallcom.2012.09.060 [17] 徐俊峰, 刘坚, 赵震, 张桂臻, 段爱军, 姜桂元, 徐春明. 三维有序大孔钙钛矿LaFeO3催化剂的制备及其催化炭黑颗粒燃烧性能[J]. 催化学报,2010,31(2):236−241.XU Jun-feng, LIU Jian, ZHAO Zhen, ZHANG Gui-zhen, DUAN Ai-jun, JIANG Gui-yuan, XU Chun-ming. Preparation and catalytic performance of three-dimensionally ordered macroporous perovskite-type LaFeO3 catalyst for soot combustion[J]. Chin J Catal,2010,31(2):236−241. [18] 胡瑞生, 谭立志, 王欣, 孙艳, 胡佳楠, 赵文岩. 掺杂型稀土双钙钛矿光催化剂La2FeTiO6葡萄糖络合法制备与表征[J]. 中国稀土学报,2012,30(5):550−555.HU Rui-sheng, TAN Li-zhi, WANG Xin, SUN Yan, HU Jia-nan, ZHAO Wen-yan. Preparation and characterization of doped rare earth double perovskite-type photocatalyst La2FeTiO6 by glucose complex method[J]. J Chin Rare Earth Soc,2012,30(5):550−555. [19] LEVASSEUR B, KALIAGUINE S. Methanol oxidation on LaBO3 (B=Co, Mn, Fe) perovskite-type catalysts prepared by reactive grinding[J]. Appl Catal A: Gen,2008,343(1/2):29−38. doi: 10.1016/j.apcata.2008.03.016 [20] 杨彩虹, 韩怡卓, 李文彬. Ni-La2O3/C催化剂上甲醇羰基化反应性能的研究[J]. 燃料化学学报,2000,28(5):392−395. doi: 10.3969/j.issn.0253-2409.2000.05.003YANG Cai-hong, HAN Yi-zhuo, LI Wen-bin. Effect of La2O3 on Ni/C catalysts for vapor phase carbonylation of methanol[J]. J Fuel Chem Technol,2000,28(5):392−395. doi: 10.3969/j.issn.0253-2409.2000.05.003 [21] MICKEVIČIUS S, GREBINSKIJ S, BONDARENKA V, VENGALIS B, ŠLIUŽIENĖ K, ORLOWSKI B A, DRUBE W. Investigation of epitaxial LaNiO3−x thin films by high-energy XPS[J]. J Alloy Compd,2006,423(1):107−111. [22] HONMA T, BENINO Y, FUJIWARA T, KOMATSU T, SATO R, DIMITROV V. Electronic polarizability, optical basicity, and interaction parameter of La2O3 and related glasses[J]. J Appl Phys,2002,91(5):2942−2950. doi: 10.1063/1.1436292 [23] WU M D, MA S W, CHEN S Y, XIANG W G. Fe–O terminated LaFeO3 perovskite oxide surface for low temperature toluene oxidation[J]. J Clean Prod,2020,277:123224. doi: 10.1016/j.jclepro.2020.123224 [24] WANG H H, GUO Z Q, HAO W T, ZHANG Y J, CAO E S. Ethanol sensing characteristics of BaTiO3/LaFeO3 nanocomposite[J]. Mater Lett,2018,234:40−44. [25] CAO E S, QIN Y R, CUI T T, SUN L, HAO W T, ZHANG Y J. Influence of Na doping on the magnetic properties of LaFeO3 powders and dielectric properties of LaFeO3 ceramics prepared by citric sol-gel method[J]. Ceram Int,2017,43(10):7922−7928. doi: 10.1016/j.ceramint.2017.03.119 -




 下载:
下载: