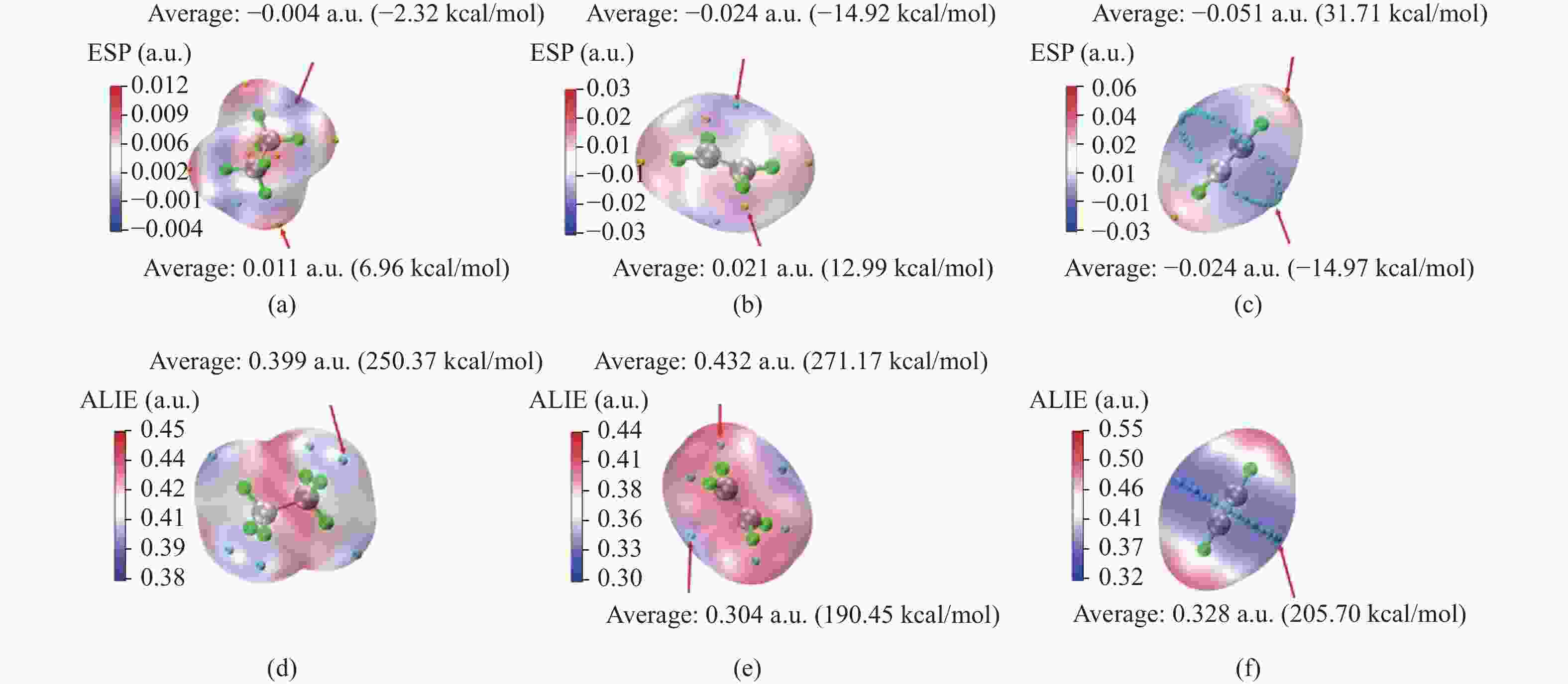
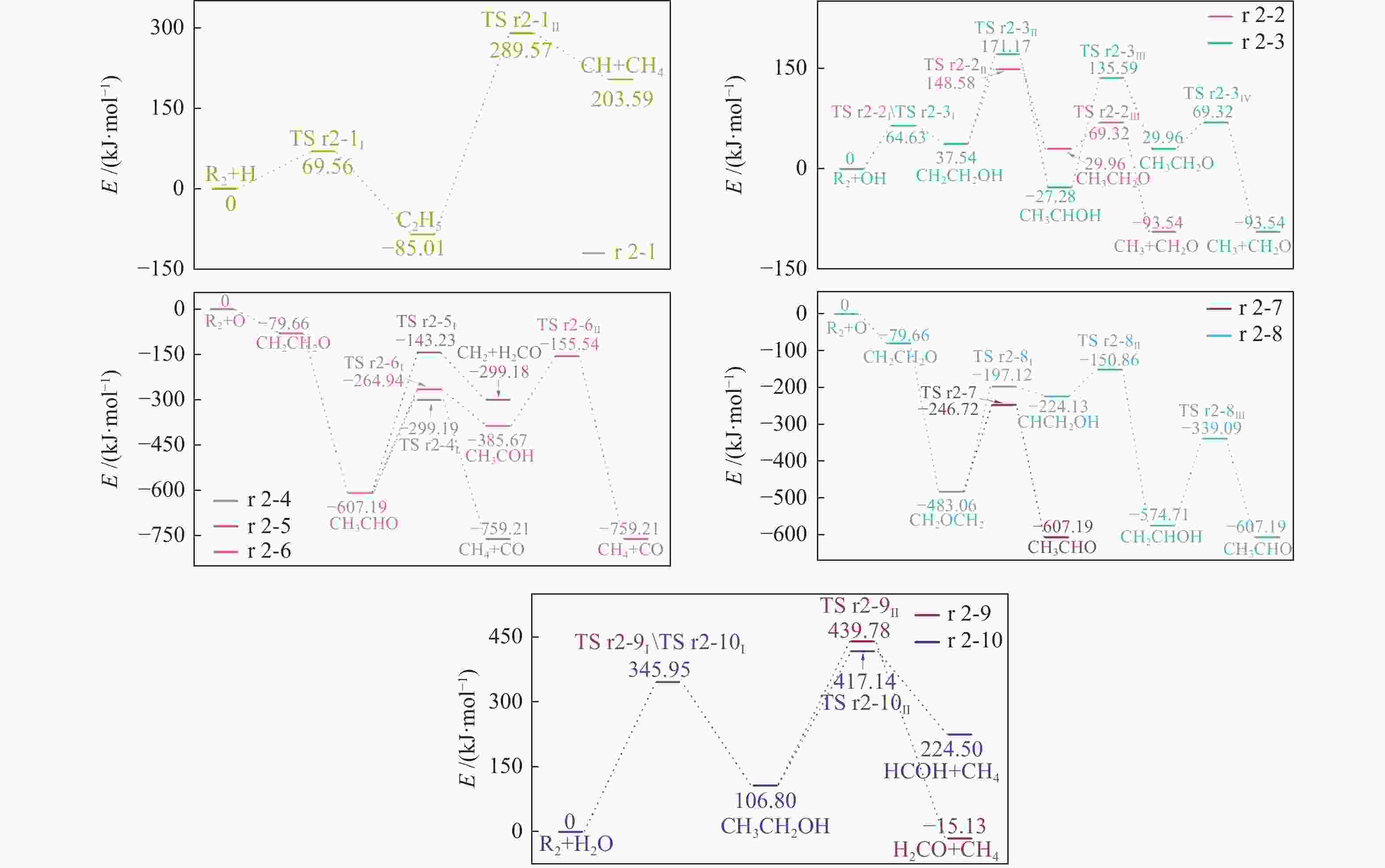
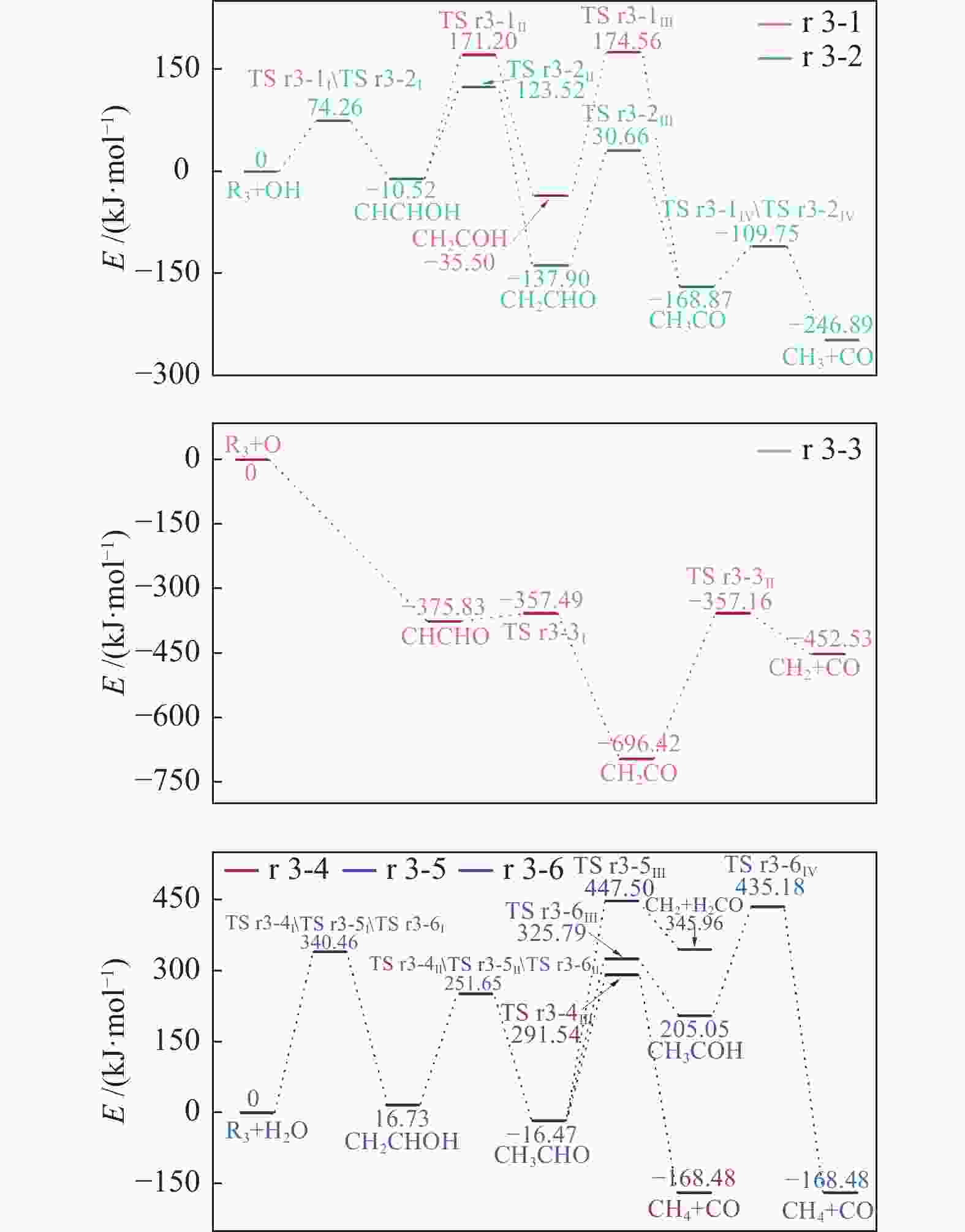
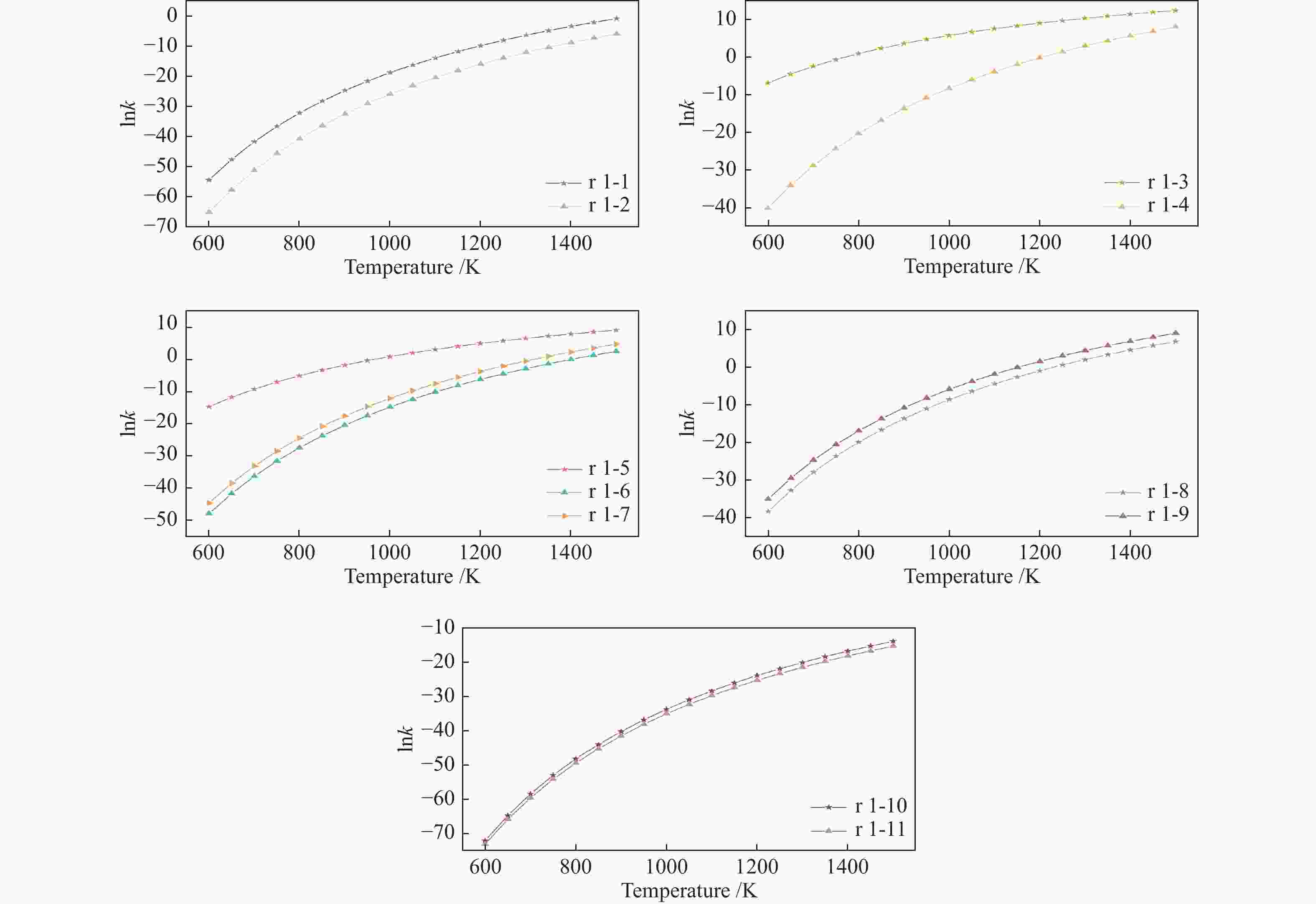
| [1] |
MARCULESCU C, CENUŞĂ V, ALEXE F. Analysis of biomass and waste gasification lean syngases combustion for power generation using spark ignition engines[J]. Waste Manage,2016,47:133−140. doi: 10.1016/j.wasman.2015.06.043
|
| [2] |
ARENA U. Process and technological aspects of municipal solid waste gasification. A review[J]. Waste Manage,2012,32(4):625−639. doi: 10.1016/j.wasman.2011.09.025
|
| [3] |
BROWN R C. The role of pyrolysis and gasification in a carbon negative economy[J]. Processes,2021,9(5):882. doi: 10.3390/pr9050882
|
| [4] |
BOGUSH A A, STEGEMANN J A, WILLIAMS R, WOOD I G. Element speciation in UK biomass power plant residues based on composition, mineralogy, microstructure and leaching[J]. Fuel,2018,211:712−725. doi: 10.1016/j.fuel.2017.09.103
|
| [5] |
SCHEFTELOWITZ M, BECKER R, THRÄN D. Improved power provision from biomass: A retrospective on the impacts of German energy policy[J]. Biomass Bioenergy,2018,111:1−12. doi: 10.1016/j.biombioe.2018.01.010
|
| [6] |
JÅSTAD E O, BOLKESJØ T F, TRØMBORG E, RØRSTAD P K. The role of woody biomass for reduction of fossil GHG emissions in the future North European energy sector[J]. Appl Energy,2020,274:115360. doi: 10.1016/j.apenergy.2020.115360
|
| [7] |
FUTAMURA S, KABASHIMA H, ANNADURAI G. Roles of CO2 and H2O as oxidants in the plasma reforming of aliphatic hydrocarbons[J]. Catal Today,2006,115(1/4):211−216. doi: 10.1016/j.cattod.2006.02.032
|
| [8] |
TANG F, CHI Y, JIN Y, ZHU Z, MA J. Gasification characteristics of a simulated waste under separate and mixed atmospheres of steam and CO2[J]. Fuel,2022,317:123527. doi: 10.1016/j.fuel.2022.123527
|
| [9] |
CHENG Y, THOW Z, WANG C. Biomass gasification with CO2 in a fluidized bed[J]. Powder Technol,2016,296:87−101. doi: 10.1016/j.powtec.2014.12.041
|
| [10] |
PINTO F, ANDRÉ R, MIRANDA M, NEVES D, VARELA F, SANTOS J. Effect of gasification agent on co-gasification of rice production wastes mixtures[J]. Fuel,2016,180:407−416. doi: 10.1016/j.fuel.2016.04.048
|
| [11] |
EBADI A G, HISORIEV H. Gasification of algal biomass (Cladophora glomerata L. ) with CO2/H2O/O2 in a circulating fluidized bed[J]. Environ Technol,2019,40(6):749−755. doi: 10.1080/09593330.2017.1406538
|
| [12] |
YOON S J, CHOI Y, LEE J. Hydrogen production from biomass tar by catalytic steam reforming[J]. Energy Convers Manage,2010,51(1):42−47. doi: 10.1016/j.enconman.2009.08.017
|
| [13] |
LOPEZ G, ARTETXE M, AMUTIO M, ALVAREZ J, BILBAO J, OLAZAR M. Recent advances in the gasification of waste plastics. A critical overview[J]. Renewable Sustainable Energy Rev,2018,82:576−596. doi: 10.1016/j.rser.2017.09.032
|
| [14] |
SIVARAMAKRISHNAN R, MICHAEL J V, RUSCIC B. High-temperature rate constants for H/D + C2H6 and C3H8[J]. Int J Chem Kinet,2012,44(3):194−205. doi: 10.1002/kin.20607
|
| [15] |
MICHAEL J V, SU M C, SUTHERLAND J W, HARDING L B, WAGNER A F. Rate constants for D + C2H4→C2H3D + H at high temperature: implications to the high pressure rate constant for H + C2H4→C2H5[J]. Proc Combust Inst,2005,30(1):965−973. doi: 10.1016/j.proci.2004.08.213
|
| [16] |
MIYOSHI A, OHMORI K, TSUCHIYA K, MATSUI H. Reaction rates of atomic oxygen with straight chain alkanes and fluoromethanes at high temperatures[J]. Chem Phys Lett,1993,204(3/4):241−247. doi: 10.1016/0009-2614(93)90003-J
|
| [17] |
MICHAEL J V. Rate constants for the reaction O + D2→OD + D by the flash photolysis-shock tube technique over the temperature range 825–2487 K: The H2 to D2 isotope effect[J]. J Chem Phys,1989,90(1):189−198. doi: 10.1063/1.456513
|
| [18] |
FETHI K, BINOD R G, MILÁN S, BÉLA V, AAMIR F. An experimental and theoretical study on the kinetic isotope effect of C2H6 and C2D6 reaction with OH[J]. Chem Phys Lett,2015,641:158−162. doi: 10.1016/j.cplett.2015.10.057
|
| [19] |
VASU S S, HONG Z, DAVIDSON D F, HANSON R K, GOLDEN D M. Shock tube/laser absorption measurements of the reaction rates of OH with ethylene and propene[J]. J Phys Chem A,2010,114(43):11529−11537. doi: 10.1021/jp106049s
|
| [20] |
张红梅, 林枫, 任铭琪, 李金莲, 郝玉兰, 吴红军, 赵晶莹, 赵亮, 贺永殿. 小分子烃类蒸汽热裂解自由基机理模型研究方法的探讨[J]. 化工学报,2017,68(4):1423−1433.ZHANG Hong-mei, LIN Feng, REN Ming-qi, LI Jin-lian, HE Yu-lan, WU Hong-jun, ZHAO Jing-ying, ZHAO Liang, HE Yong-dian. Free radical models of small molecular alkane pyrolysis[J]. CIESC J,2017,68(4):1423−1433.
|
| [21] |
MANION J A, HUIE R E, LEVIN R D, JR. BURGESS D R, ORKIN V L, TSANG W, MCGIVERN W S, HUDGENS J W, KNYAZEV V D, ATKINSON D B, CHAI E, TEREZA A M, LIN C Y, ALLISON T C, MALLARD W G, WESTLEY F, HERRON J T, HAMPSON R F, FRIZZELL D H. NIST Chemical Kinetics Database[EB/OL]. https://kinetics.nist.gov/kinetics/index.jsp
|
| [22] |
RAMAZANI S. Direct-dynamics VTST study of hydrogen or deuterium abstraction and C–C bond formation or dissociation in the reactions of CH3 + CH4, CH3 + CD4, CH3D + CD3, CH3CH3 + H, and CH3CD3 + D[J]. J Chem Phys,2013,138(19):194305. doi: 10.1063/1.4803862
|
| [23] |
NGUYEN T L, VEREECKEN L, PEETERS J. Quantum chemical and theoretical kinetics study of the O(3P) + C2H2 reaction: A multistate process[J]. J Phys Chem A,2006,110(21):6696−6706. doi: 10.1021/jp055961k
|
| [24] |
DING J, ZHANG L, KELI H. Thermal rate constants of the pyrolysis of n-heptane[J]. Combust Flame,2011,158(12):2314−2324. doi: 10.1016/j.combustflame.2011.04.015
|
| [25] |
DIAMANTI A, ADJIMAN C S, PICCIONE P M, REA A M, GALINDO A. Development of predictive models of the kinetics of a hydrogen abstraction reaction combining quantum-mechanical calculations and experimental data[J]. Ind Eng Chem Res,2016,56(4):815−831.
|
| [26] |
OGLIARO F, BEARPARK M J, HEYD J J, BROTHERS E N, KUDIN K N, STAROVEROV V N, KEITH T A, KOBAYASHI R, NORMAND J, RAGHAVACHARI K[CP]. Gaussian 16, Revision C. 01, Gaussian. Inc. : Wallingford, CT, USA. 2016.
|
| [27] |
LU T, CHEN F. Quantitative analysis of molecular surface based on improved Marching Tetrahedra algorithm[J]. J Mol Graphics Modell,2012,38:314−323. doi: 10.1016/j.jmgm.2012.07.004
|
| [28] |
LU T, CHEN F. Multiwfn: A multifunctional wavefunction analyzer[J]. J Comput Chem,2012,33(5):580−592. doi: 10.1002/jcc.22885
|
| [29] |
ZHANG J. Libreta: Computerized optimization and code synthesis for electron repulsionintegral evaluation[J]. J Chem Theory Comput,2018,14(2):572−587. doi: 10.1021/acs.jctc.7b00788
|
| [30] |
HUMPHREY W, DALKE A, SCHULTEN K. VMD: Visual molecular dynamics[J]. J Mol Graphics Modell,1996,14(1):33−38. doi: 10.1016/0263-7855(96)00018-5
|
| [31] |
LU T, CHEN Q. Shermo: A general code for calculating molecular thermochemistry properties[J]. Comput Theor Chem,2021,1200:113249. doi: 10.1016/j.comptc.2021.113249
|
| [32] |
付蓉, 卢天, 陈飞武. 亲电取代反应中活性位点预测方法的比较[J]. 物理化学学报,2014,30(4):628−639. doi: 10.3866/PKU.WHXB201401211FU Rong, LU Tian, CHEN Fei-wu. Comparing methods for predicting the reactive site of electrophilic substitution[J]. Acta Phys-Chim Sin,2014,30(4):628−639. doi: 10.3866/PKU.WHXB201401211
|
| [33] |
BARTLETT R, MUSIA M. Coupled-cluster theory in quantum chemistry[J]. Rev Mod Phys,2007,79(1):291−352. doi: 10.1103/RevModPhys.79.291
|
| [34] |
杨振丽. 烷基过氧自由基和芳香烃双环过氧自由基与HO2的化学反应动力学理论研究[D]. 合肥: 中国科学技术大学, 2020.YANG Zhen-li. A theoretical study on the chemical reaction kinetics of the alkyl peroxy radicals and aromatic bicyclic peroxy radicals with HO2 reactions[D]. Hefei: University of Science and Technology of China, 2020.
|
| [35] |
MIHÁLY K, JÜRGEN G. Approximate treatment of higher excitations in coupled-cluster theory. II. Extension to general single-determinant reference functions and improved approaches for the canonical Hartree-Fock case[J]. J Chem Phys,2008,129(14):144101. doi: 10.1063/1.2988052
|
| [36] |
BOMBLE Y J, STANTON J F, KÁLLAY M, GAUSS J. Coupled-cluster methods including noniterative corrections for quadruple excitations[J]. J Chem Phys,2005,123(5):54101. doi: 10.1063/1.1950567
|
| [37] |
DAVIDSON E R. Comment on comment on Dunning's correlation-consistent basis sets[J]. Chem Phys Lett,1996,260(3):514−518.
|
| [38] |
MERRICK J P, MORAN D, RADOM L. An evaluation of harmonic vibrational frequency scale factors[J]. J Phys Chem A,2007,111(45):11683. doi: 10.1021/jp073974n
|
| [39] |
SKODJE R T, TRUHLAR D G, GARRETT B C. Vibrationally adiabatic models for reactive tunneling[J]. J Chem Phys,1982,77(12):5955−5976. doi: 10.1063/1.443866
|
| [40] |
GONZALEZ C, SCHLEGEL H B. Reaction-path following in mass-weighted internal coordinates[J]. J Phys Chem,1990,94:5523−5527. doi: 10.1021/j100377a021
|
| [41] |
黄罗仪, 翁约约, 黄旭慧, 王朝杰. 车前草中黄酮类成分结构和性质的理论研究[J]. 高等学校化学学报,2021,42(9):2752−2765. doi: 10.7503/cjcu20210180HUANG Luo-yi, WENG Yue-yue, HUANG Xu-hui, WANG Chao-jie. Theoretical study on the structures and properties of flavonoids in plantain[J]. Chem J Chin Univ,2021,42(9):2752−2765. doi: 10.7503/cjcu20210180
|
| [42] |
POLITZER P, MURRAY J S, BULAT F A. Average local ionization energy: A review[J]. J Mol Model,2010,16(11):1731−1742. doi: 10.1007/s00894-010-0709-5
|
| [43] |
曹静思, 陈飞武. 芳香化合物亲核、亲电反应活性的理论预测和实验反应速率的相关性研究[J]. 有机化学,2016,36(10):2463−2471. doi: 10.6023/cjoc201602026CAO Jing-si, CHEN Fei-wu. Theoretical study on the correlation of the experimental nucleophilic and electrophilic reaction rates of aromatic compounds with the prediction results of theoretical methods[J]. Chin J Org Chem,2016,36(10):2463−2471. doi: 10.6023/cjoc201602026
|
| [44] |
唐海飞, 颜涛, 吴梅青. 莲心碱定量分子表面分析及反应位点预测[J]. 山西卫生健康职业学院学报,2020,30(6):7−9.TANG Hai-fei, YAN Tao, WU Mei-qing. Quantitative molecular surface analysis and reaction site prediction of Liensinine[J]. J Shanxi H Voc Coll,2020,30(6):7−9.
|
| [45] |
YAN T, HASE W L, DOUBLEDAY C. Energetics, transition states, and intrinsic reaction coordinates for reactions associated with O(3P) processing of hydrocarbon materials[J]. J Chem Phys,2004,120(19):9253−9265. doi: 10.1063/1.1705574
|
| [46] |
GARTON D J, MINTON T K, TROYA D, PASCUAL R, SCHATZ G C. Hyperthermal reactions of O(3P) with alkanes: observations of novel reaction pathways in crossed-beams and theoretical studies[J]. J Phys Chem A,2003,107(23):4583−4587. doi: 10.1021/jp0226026
|
| [47] |
GARTON D J, MINTON T K, HU W, SCHATZ G C. Experimental and theoretical investigations of the inelastic and reactive scattering dynamics of O(3P) collisions with ethane[J]. J Phys Chem A,2009,113(16):4722−4738. doi: 10.1021/jp900412w
|
| [48] |
SUN Y C, WANG I T, NGUYEN T L, LU H F, YANG X, MEBEL A M. A combined quantum chemistry and RRKM calculation predicts the O(1D) + C2H6 reaction can produce water molecule in a collision-free crossed molecular beam environment[J]. J Phys Chem A,2003,107:6986−6994.
|
| [49] |
NGUYEN T L, VEREECKEN L, HOU X J, NGUYEN M T, PEETERS J. Potential energy surfaces, product distributions and thermal rate coefficients of the reaction of O(3P) with C2H4(X1–Ag): A comprehensive theoretical study[J]. J Phys Chem A,2005,109:7489−7499. doi: 10.1021/jp052970k
|
| [50] |
TALOTTA F, MORISSET S, ROUGEAU N, LAUVERGNAT D, AGOSTINI F. Electronic structure and excited states of the collision reaction O(3P) + C2H4: A multiconfigurational perspective[J]. J Phys Chem A,2021,125(28):6075−6088. doi: 10.1021/acs.jpca.1c02923
|
| [51] |
TSANG W, HAMPSON R F. Chemical kinetic data base for combustion chemistry. Part I. methane and related compounds[J]. J Phys Chem Ref Data,1986,15(3):1087−1279. doi: 10.1063/1.555759
|
| [52] |
RAJAK K, MAITI B. Communications: direct dynamics study of the O((3)P)+C(2)H(2) reaction: contribution from spin nonconserving route[J]. J Chem Phys,2010,133(1):2093.
|
| [53] |
ZUO J, CHEN Q, HU X, HUA G, XIE D. Dissection of the multichannel reaction of acetylene with atomic oxygen: from the global potential energy surface to rate coefficients and branching dynamics[J]. Phys Chem Chem Phys,2019,21:1408−1416. doi: 10.1039/C8CP07084A
|
| [54] |
GE Y, GORDON M S, BATTAGLIA F, FOX R O. Theoretical study of the pyrolysis of methyltrichlorosilane in the gas phase. 3. Reaction rate constant calculations[J]. J Phys Chem A,2010,114(6):2384−2392. doi: 10.1021/jp911673h
|
| [55] |
YANG X, JASPER A W, KIEFER J H, TRANTER R S. The dissociation of diacetyl: a shock tube and theoretical study[J]. J Phys Chem A,2009,113(29):8318−8326. doi: 10.1021/jp903716f
|
| [56] |
OEHLSCHLAEGER M A, DA VIDSON D F, HANSON R K. High-temperature ethane and propane decomposition[J]. Proc Combust Inst,2005,30(1):1119−1127. doi: 10.1016/j.proci.2004.07.032
|
| [57] |
BACK R A. A search for a gas-phase free-radical inversion displacement reaction at a saturated carbon atom[J]. Can J Chem,2011,61(5):916−920.
|
| [58] |
HUYNH L K, PANASEWICZ S, RATKIEWICZ A, TRUONG T N. Ab initio study on the kinetics of hydrogen abstraction for the H + alkene→H2 + alkenyl reaction class[J]. J Phys Chem A,2007,111(11):2156−2165. doi: 10.1021/jp066659u
|
| [59] |
HUYNH L K, BARRIGER K, VIOLI A. Kinetics study of the OH + alkene→H2O + alkenyl reaction class[J]. J Phys Chem A,2008,112:1436−1444. doi: 10.1021/jp077028i
|
| [60] |
LI X, JASPER A W, ZÁDOR J, MILLER J A, KLIPPENSTEIN S J. Theoretical kinetics of O + C2H4[J]. Proc Combust Inst,2017,36:219−227. doi: 10.1016/j.proci.2016.06.053
|
| [61] |
DUFOUR A, MASSON E, GIRODS P, ROGAUME Y, ZOULALIAN A. Evolution of aromatic tar composition in relation to methane and ethylene from biomass pyrolysis-gasification[J]. Energy Fuels,2011,25:4182−4189. doi: 10.1021/ef200846g
|
| [62] |
DUFOUR A, VALIN S, CASTELLI P, THIERY S B, BOISSONNET G, ZOULALIAN A, GLAUDE P A. Mechanisms and kinetics of methane thermal conversion in a syngas[J]. Ing Eng Chem Res,2009,48(14):6564−6572.
|
| [63] |
WU W G, LUO Y H, YI S, ZHANG Y L, ZHAO S H, WANG Y. Nascent biomass tar evolution properties under homogeneous/heterogeneous decomposition conditions in a two-stage reactor[J]. Energy Fuels,2011,25:5394−5406. doi: 10.1021/ef2007276
|
| [64] |
MILLER J A, MELIUS C F. A theoretical analysis of the reaction between hydroxyl and acetylene[J]. Symp Combust,1989,22(1):1031−1039. doi: 10.1016/S0082-0784(89)80113-4
|
| [65] |
VANDOOREN J, TIGGELEN P. Reaction mechanisms of combustion in low pressure acetylene-oxygen flames[J]. Symp Combust,1977,16(1):1133−1144. doi: 10.1016/S0082-0784(77)80402-5
|
| [66] |
TSUBOI T, HASHIMOTO K. Shock tube study on homogeneous thermal oxidation of methanol[J]. Combust Flame,1981,42:61−76. doi: 10.1016/0010-2180(81)90142-5
|
 20221219105016_91.docx
20221219105016_91.docx





 下载:
下载: