Density functional theory study of CO2 reduction on Cu13, Cu12Zr and Cu12Zn clusters
-
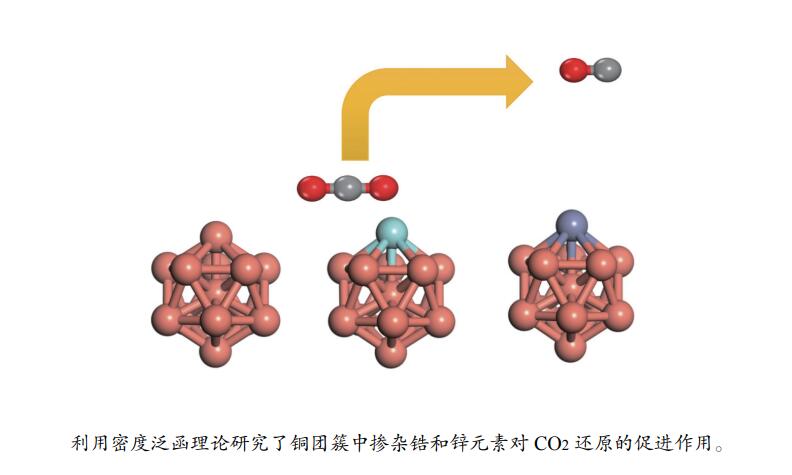
摘要: 本研究采用密度泛函理论研究了Cu13、Cu12Zn和Cu12Zr团簇的CO2还原反应的吸附和活化能力,计算结果表明相比于Cu13团簇,Cu12Zr增强了对反应物和中间体的吸附能力,而Cu12Zn团簇降低了对反应物和中间体的吸附能力。计算了Cu13、Cu12Zr和Cu12Zn团簇上CO2还原为CO的能垒为分别为0.65、0.35和0.58 eV,CO2加氢生成HCOO的能垒为0.87、0.77和0.49 eV,而CO2加氢生成COOH的能垒为1.67、1.89和0.99 eV。Zn和Zr元素的掺杂提高了铜团簇的CO2催化还原能力,其中,表现为Cu12Zr团簇有利于CO2解离生成CO,Cu12Zn团簇有利于CO2加氢生成HCOO。Abstract: In this study, we used density functional theory to study the adsorption and activation capacity of Cu13, Cu12Zn, and Cu12Zr clusters for CO2 reduction. The calculated results showed that Cu12Zr enhanced the adsorption capacity of reactants and intermediates compared with Cu13 clusters, while Cu12Zn clusters decreased the adsorption capacity of reactants and intermediates. We calculated that the energy barriers for CO2 reduction to CO on Cu13, Cu12Zr, and Cu12Zn clusters were 0.65, 0.35 and 0.58 eV, respectively, and the energy barriers for CO2 plus H to generate HCOO were 0.87, 0.77 and 0.49 eV, while the energy barriers of CO2 hydrogenation to COOH were 1.67, 1.89 and 0.99 eV. The doping of Zn and Zr elements improved the CO2 catalytic reduction ability of the Cu clusters, which showed that the Cu12Zr clusters were favorable for the dissociation of CO2 to form CO, and the Cu12Zn clusters were favorable for the hydrogenation of CO2 to HCOO.
-
Key words:
- density functional theory /
- CO2 reaction /
- copper-based alloy /
- adsorption and activation
-
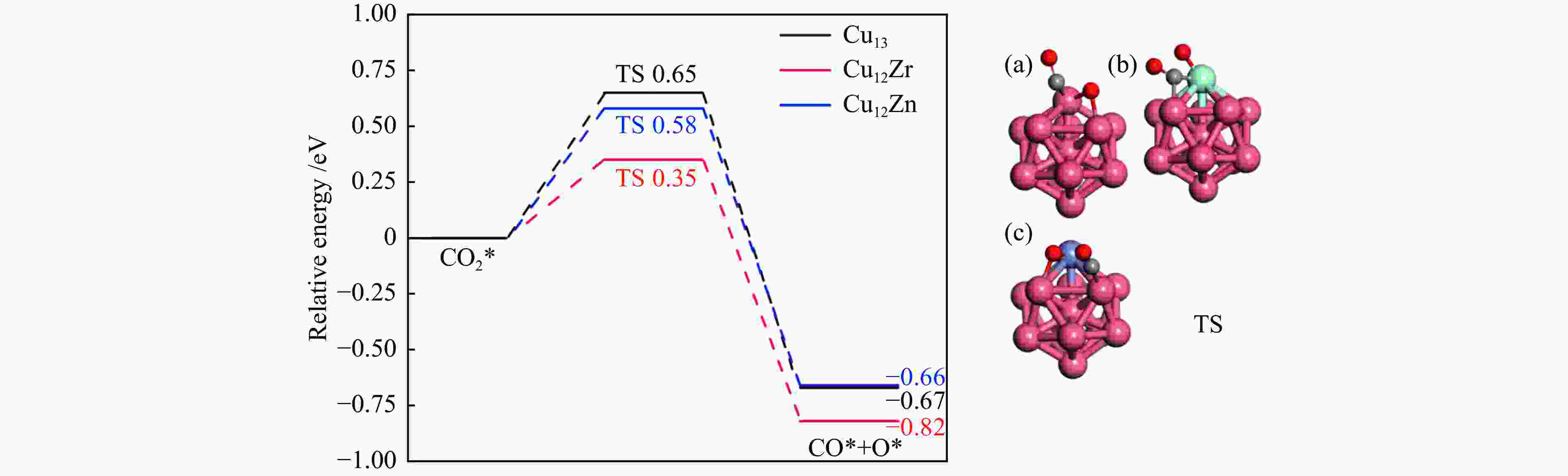
图 3 CO2*解离成CO和O势能图及(a)Cu13、(b)Cu12Zr和(c)Cu12Zn团簇表面过渡态结构的对应描述C、O和H原子以灰色、红色和白色显示
Figure 3 CO2* dissociation into CO and O potential energy diagrams and corresponding descriptions of the surface transition state structures of (a) Cu13, (b) Cu12Zr and (c) Cu12Zn clusters (C, O and H atoms are shown in grey, red and white)
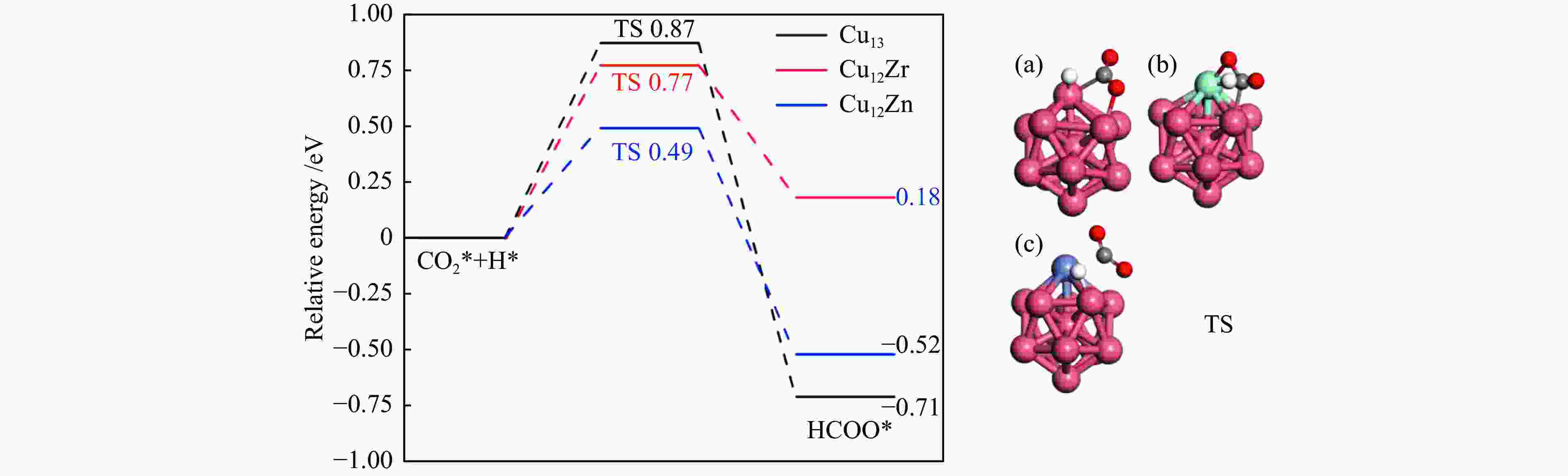
图 4 CO2*加氢生成HCOO*势能图以及(a)Cu13、(b)Cu12Zr和(c)Cu12Zn团簇表面过渡态结构的相应描述C、O和H原子以灰色、红色和白色显示
Figure 4 Hydrogenation of CO2* to HCOO* potential energy diagram and corresponding descriptions of the surface transition state structures of (a) Cu13, (b) Cu12Zr and (c) Cu12Zn clusters (C, O and H atoms are shown in grey, red and white)
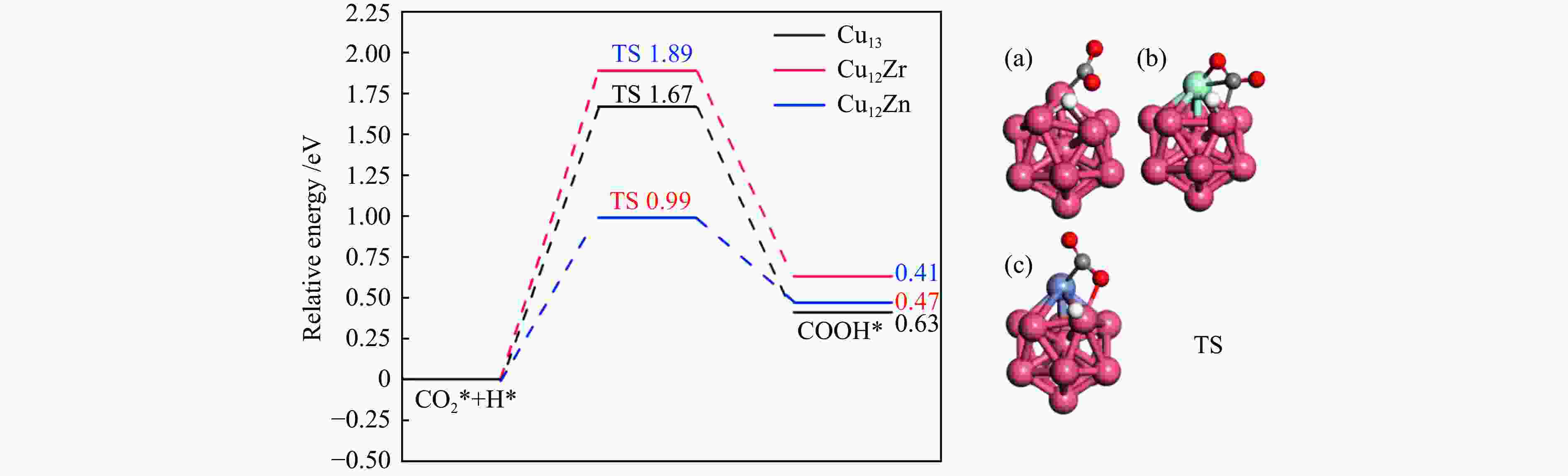
图 5 CO2*加氢生成COOH*势能图以及(a)Cu13、(b)Cu12Zr和(c)Cu12Zn团簇表面过渡态结构的相应描述C、O和H原子以灰色、红色和白色显示
Figure 5 Hydrogenation of CO2* to COOH* potential energy diagram and corresponding descriptions of the surface transition state structures of (a) Cu13, (b) Cu12Zr and (c) Cu12Zn clusters (C, O and H atoms are shown in grey, red and white)
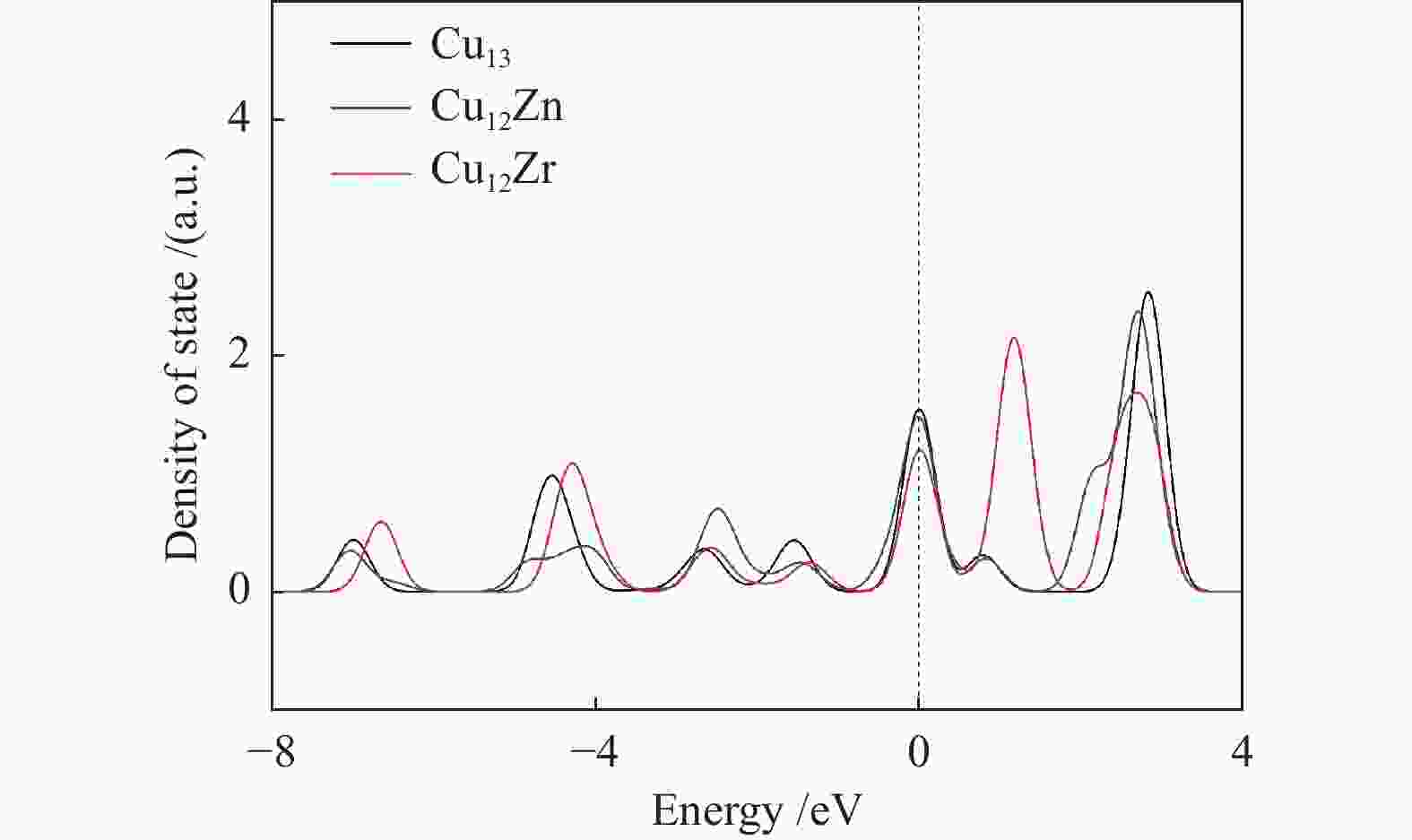
图 7 Cu13表面的Cu原子(黑色)、Cu12Zn表面的Zn原子(蓝色)和Cu12Zr表面的Zr原子(红色)的d电子轨道态密度图
Figure 7 The d electron orbital density of states of Cu atoms (black) on the surface of Cu13, Zn atoms (blue) on the surface of Cu12Zn, and Zr atoms (red) on the surface of Cu12Zr, respectively (the dashed line at 0 eV represents the Fermi level)
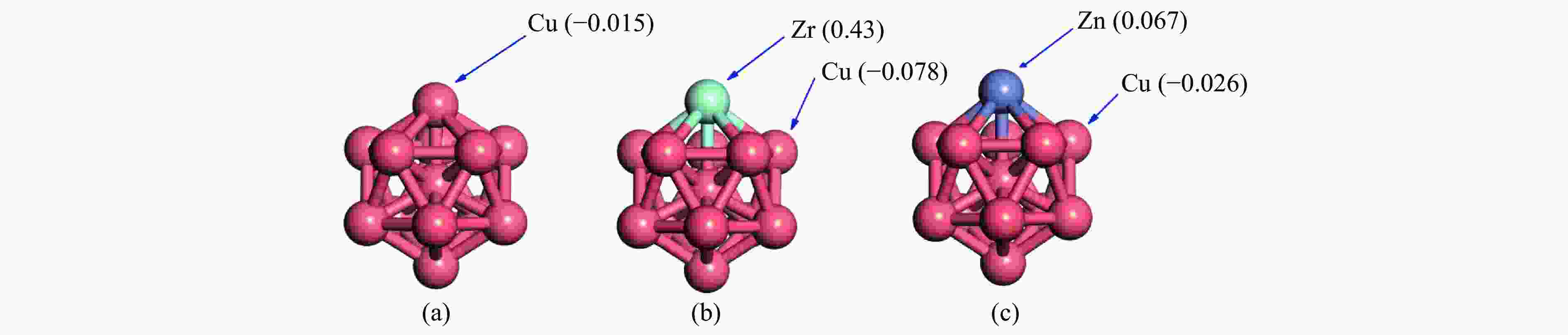
表 1 Zr和Zn掺杂在Cu13团簇表面的偏析能SE和CO2吸附参数
Table 1 Segregation energy SE and CO2 adsorption parameters of Zr and Zn doping on the surface of Cu13 cluster
Species SE/eV EadsCO2/eV Charge/e d(C=O)/Å ∠OCO(°) Cu13 − −0.39 −0.018 1.246 137 Cu12Zr −2.4 −1.95 0.432 1.345 127 Cu12Zn −0.86 −0.17 0.067 1.182 178 表 2 中间产物在Cu13、Cu12Zr和 Cu12Zn 簇上的吸附构型和吸附能
Table 2 Adsorption configuration and adsorption energy (in eV) of intermediates on Cu13, Cu12Zr and Cu12Zn clusters
Species Cu13 Cu12Zr Cu12Zn CO 


Eads/eV −1.84 −2.10 −1.72 HCOO 


Eads/eV −4.11 −4.83 −3.82 COOH 


Eads/eV −2.85 −3.97 −2.42 O 


Eads/eV −2.20 −4.23 −1.98 H 


Eads/eV −0.67 −0.86 −0.41 表 3 Cu13、Cu12Zr和Cu12Zn团簇CO2还原的活化能Ea和反应能ΔE
Table 3 Activation energy Ea and reaction energy ΔE of CO2 reduction on Cu13, Cu12Zr and Cu12Zn clusters
Elementary step Cu13/eV Cu12Zr/eV Cu12Zn/eV Ea ΔE Ea ΔE Ea ΔE CO2*→CO* + O* 0.65 −0.67 0.35 −0.82 0.58 −0.66 CO2* + H*→HCOO* 0.87 −0.71 0.77 0.18 0.49 −0.52 CO2* + H*→COOH* 1.67 0.41 1.89 0.63 0.99 0.47 -
[1] 卢思宇, 杨海艳, 杨承广, 高鹏, 孙予罕. In2O3/SSZ-13催化CO2加氢高选择性合成液化石油气[J]. 燃料化学学报,2021,49(8):1132−1139. doi: 10.1016/S1872-5813(21)60057-9LU Si-yu, YANG Hai-yan, YANG Cheng-guang, GAO Peng, SUN Yu-han. Highly selective synthesis of LPG from CO2 hydrogenation over In2O3/SSZ-13 binfunctional catalyst[J]. J Fuel Chem Technol,2021,49(8):1132−1139. doi: 10.1016/S1872-5813(21)60057-9 [2] HE J, JIN Z, GAN F, XIE L, GUO J, ZHANG S, JIA C Q, MA D, DAI Z, JIANG X. Liquefiable biomass-derived porous carbons and their applications in CO2 capture and conversion[J]. Green Chem,2022,24:3376−3415. doi: 10.1039/D1GC04746A [3] WEI J, YAO R, HAN Y, GE Q, SUN J. Towards the development of the emerging process of CO2 heterogenous hydrogenation into high-value unsaturated heavy hydrocarbons[J]. Chem Soc Rev,2021,50:10764−10805. doi: 10.1039/D1CS00260K [4] WANG G, CHEN J, DING Y, CAI P, YI L, LI Y, TU C, HOU Y, WEN Z, DAI L. Electrocatalysis for CO2 conversion: from fundamentals to value-added products[J]. Chem Soc Rev,2021,50(8):4993−5061. doi: 10.1039/D0CS00071J [5] GAUTAM S, DHARAMVIR K, GOEL N. CO2 adsorption and activation over medium sized Cun(n=7, 13 and 19) clusters: a density functional study[J]. Comput Theor Chem,2013,1009:8−16. doi: 10.1016/j.comptc.2012.12.010 [6] PADAMA A A B, OCON J D, NAKANISHI H, KASAI H. Interaction of CO, O, and CO2 with Cu cluster supported on Cu(111): a density functional theory study[J]. J Phys Condens Matter,2019,31(41):415201. doi: 10.1088/1361-648X/ab2b66 [7] ZHENG H, NARKHEDE N, HAN L, ZHANG H, LI Z. Methanol synthesis from CO2: A DFT investigation on Zn-promoted Cu catalyst[J]. Res Chem Intermediat,2020,46(3):1749−1769. doi: 10.1007/s11164-019-04061-2 [8] JO D Y, LEE M W, HAM H C, LEE K Y. Role of the Zn atomic arrangements in enhancing the activity and stability of the kinked Cu(211) site in CH3OH production by CO2 hydrogenation and dissociation: First-principles microkinetic modeling study[J]. J Catal,2019,373:336−350. doi: 10.1016/j.jcat.2019.04.009 [9] LI H, SHEN Y Y, DU H N, LI J, XU C X. Insight into the mechanisms of CO2 reduction to CHO over Zr-doped Cu nanoparticle[J]. Chem Phys,2021,540:111012. doi: 10.1016/j.chemphys.2020.111012 [10] 申艳阳. 铜锆合金纳米团簇催化性能的第一性原理研究[D]. 太原: 太原理工大学, 2021.SHEN Yan-yang. A first-principles study on the catalytic performance of copper-zirconium alloy nanoclusters[D]. Taiyuan: Taiyuan University of Technology, 2021. [11] DELLEY B. From molecules to solids with the DMol3 approach[J]. J Chem Phys,2000,113(18):7756−7764. doi: 10.1063/1.1316015 [12] DELLEY B. An all-electron numerical method for solving the local density functional for polyatomic molecules[J]. J Chem Phys,1990,92(1):508−517. doi: 10.1063/1.458452 [13] PERDEW J P, BURKE K, ERNZERHOF M. Generalized gradient approximation made simple[J]. Phys Rev Lett,1996,77(18):3865. doi: 10.1103/PhysRevLett.77.3865 [14] PERDEW J P, WANG Y. Accurate and simple analytic representation of the electron-gas correlation energy[J]. Phys Rev B,1992,45(23):13244. doi: 10.1103/PhysRevB.45.13244 [15] DELLEY B. Hardness conserving semilocal pseudopotentials[J]. Phys Rev B,2002,66(15):155125. doi: 10.1103/PhysRevB.66.155125 [16] GRIMME S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction[J]. J Comput Chem,2006,27(15):1787−1799. doi: 10.1002/jcc.20495 [17] HALGREN T A, LIPSCOMB W N. The synchronous-transit method for determining reaction pathways and locating molecular transition states[J]. Chem Phys Lett,1977,49(2):225−232. doi: 10.1016/0009-2614(77)80574-5 [18] GOVIND N, PETERSEN M, FITZGERALD G, KING-SMITH D, ANDZELM J. A generalized synchronous transit method for transition state location[J]. Comp Mater Sci,2003,28(2):250−258. doi: 10.1016/S0927-0256(03)00111-3 [19] OU L, CHEN S. DFT calculation analysis of oxygen reduction activity and stability of bimetallic catalysts with Pt-segregated surface[J]. Sci China Chem,2015,58(4):586−592. doi: 10.1007/s11426-015-5324-y -




 下载:
下载: