Adsorption of PbCl2 vapor in high temperature furnace by modified attapulgite: Experimental and theoretical calculations
-
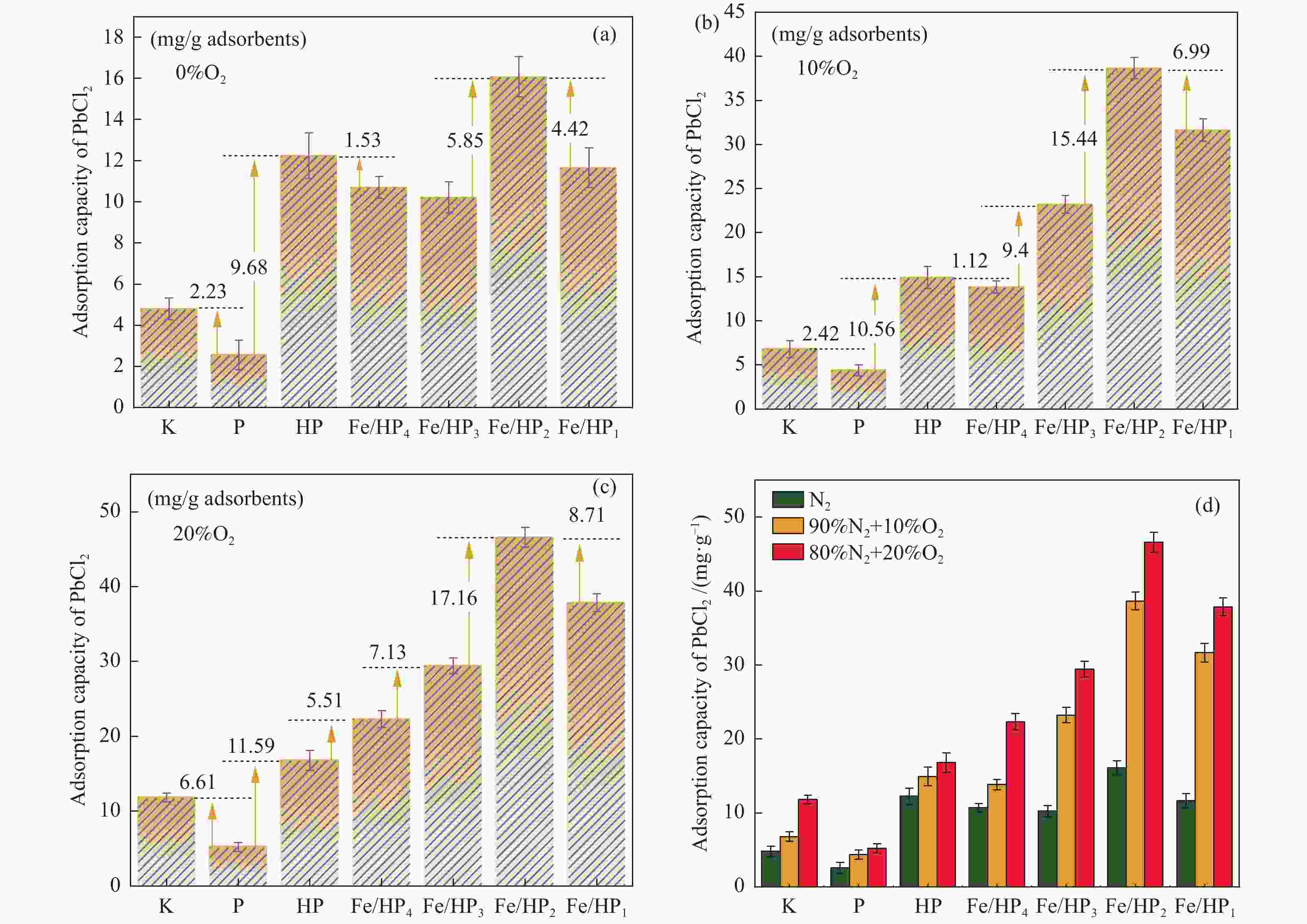
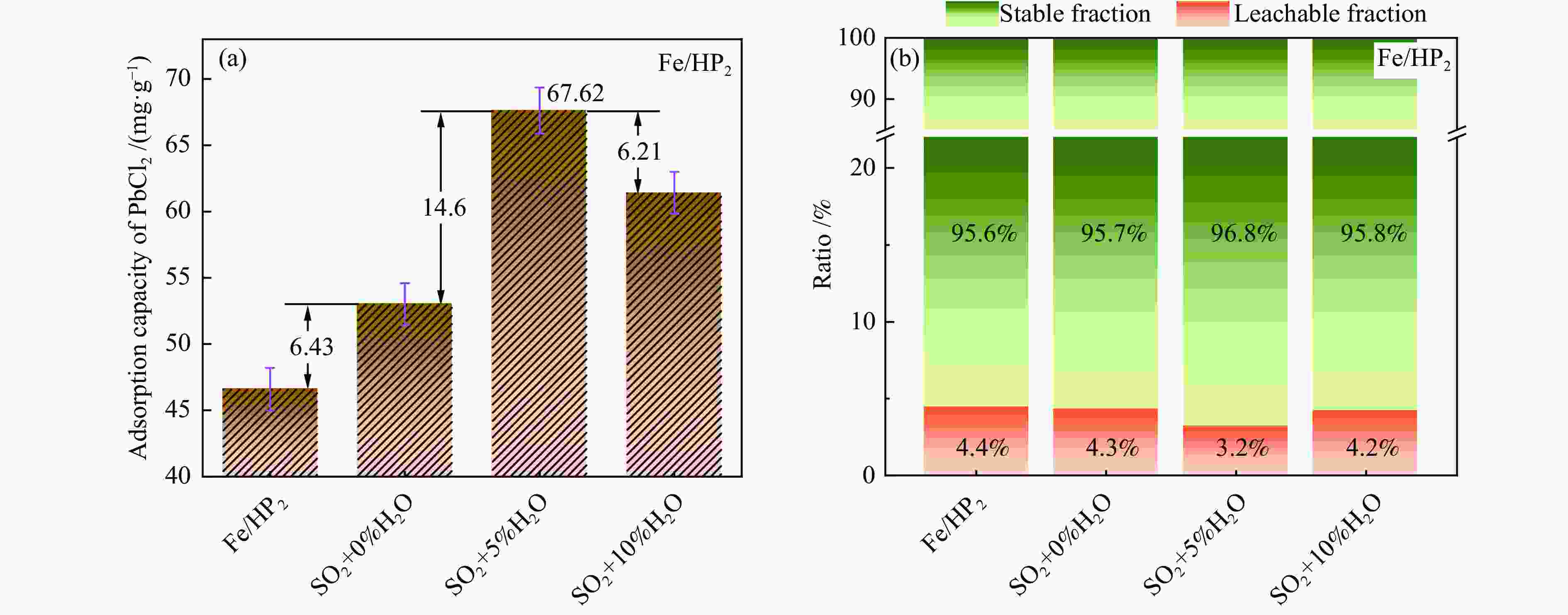
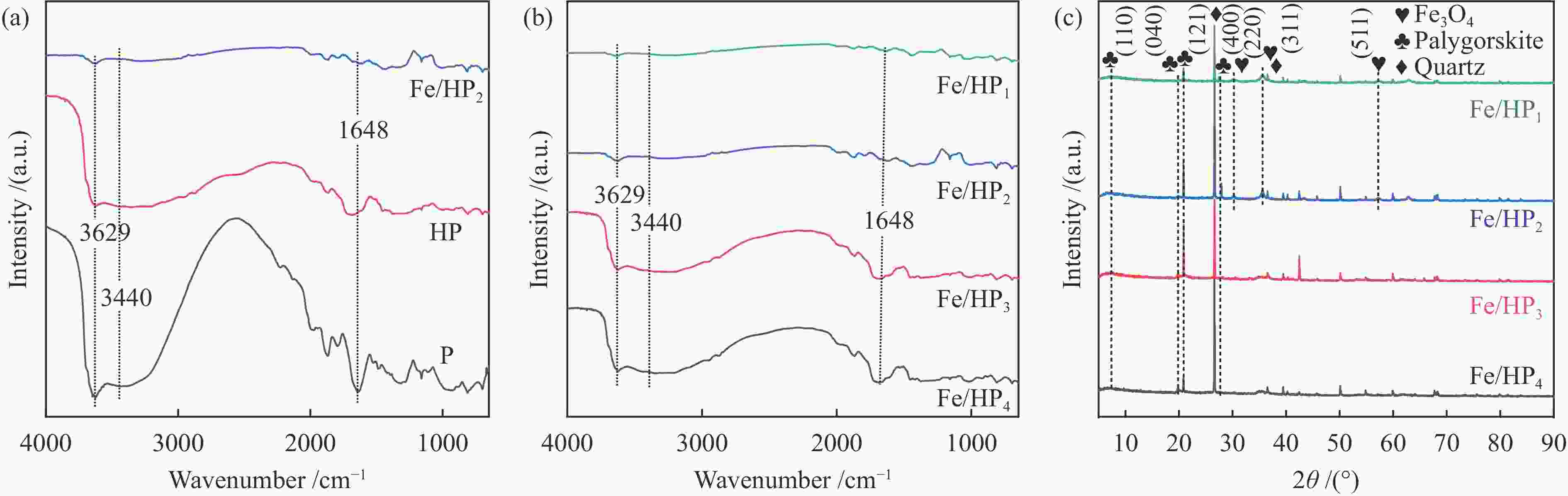
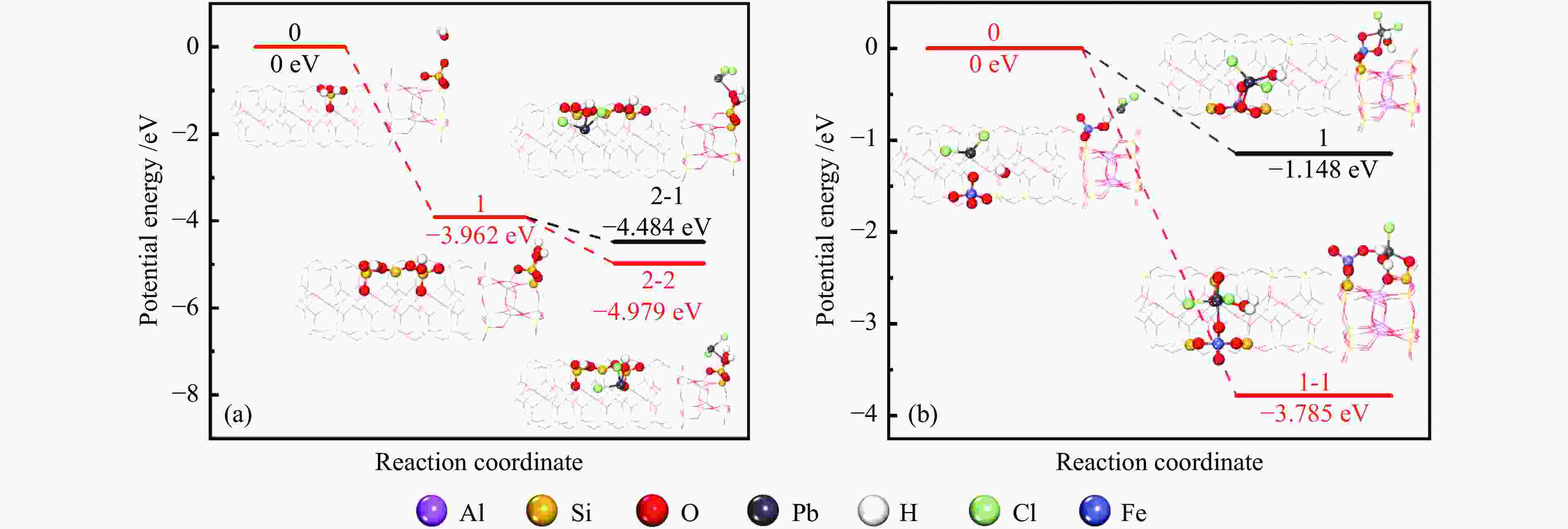
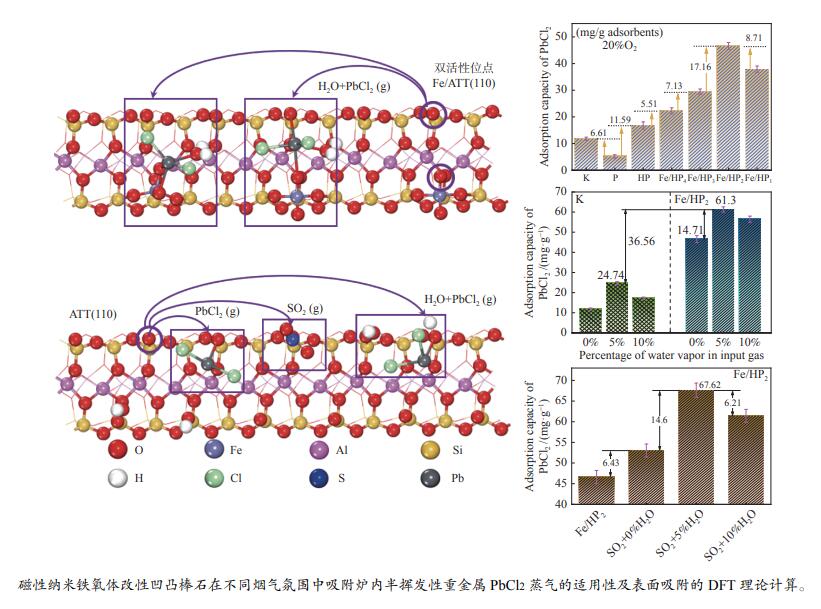
摘要: 通过酸活化和负载磁性纳米铁氧体复合改性方式获得改性凹凸棒石,探究其在不同烟气氛围中吸附炉内半挥发性重金属PbCl2蒸气的适用性,结合FT-IR、BET和XRD等表征手段以及DFT理论计算深入探究其对PbCl2蒸气的吸附机理。结果表明,酸活化通过分解原矿中杂质提高表面活性位点占比,复合改性后铁基氧化物与凹凸棒石晶格氧形成的双活性吸附位点显著增强了其PbCl2吸附容量,质量比为1∶2的Fe/HP2样品吸附容量最高达67.62(mg PbCl2/g吸附剂)。当高温烟气中含有O2、SO2和少量H2O时,会提升复合改性凹凸棒石的PbCl2吸附容量。DFT理论计算表明,H2O、O2、SO2和PbCl2在ATT(110)表面均发生化学吸附,同时证明了H2O通过共吸附作用促进PbCl2在ATT(110)和Fe/ATT(110)表面的吸附。PbCl2在H2O分子形成的吸附氧位点的吸附弱于在晶格氧位点的吸附,在Fe/ATT(110)表面会优先与晶格氧位点和铁氧化物团簇中氧位点双活性位点通过强相互作用成键。Abstract: The modified attapulgite was obtained by acid activation and loaded magnetic nano-ferrite composite modification. The applicability of attapulgite in the adsorption furnace of semi-volatile heavy metal PbCl2 vapor in different flue gas atmosphere was explored. Besides, the adsorption mechanism of PbCl2 vapor was investigated by combining FT-IR, BET, XRD and DFT theoretical calculation. The results show that acid activation increases the proportion of surface-active sites by decomposing impurities in the original ore, and the double active adsorption sites formed by the composite modified iron-based oxides and attapulgite lattice oxygen significantly enhance the adsorption capacity of PbCl2. The maximum adsorption capacity of Fe/HP2 samples with the mass ratio of 1∶2 is 67.62 (mg PbCl2/g adsorbent). When the high-temperature flue gas contains O2, SO2 and a small amount of H2O, it can enhance the adsorption capacity of modified attapulgite. In addition, DFT theoretical calculations show that H2O, O2, SO2 and PbCl2 all undergo chemisorption on the surface of ATT(110), and it also demonstrates that H2O promotes the adsorption of PbCl2 on the surface of ATT(110) and Fe/ATT(110) through co-adsorption. Weaker adsorption of PbCl2 at the adsorbed oxygen sites formed by H2O molecules instead of at the lattice oxygen sites can be preferentially bond to double active sites (the lattice oxygen sites and the oxygen site) in the iron oxide clusters through strong interactions on the Fe/ATT(110) surface.
-
Key words:
- PbCl2 /
- stabilization /
- adsorbents /
- adsorption mechanism /
- DFT
-
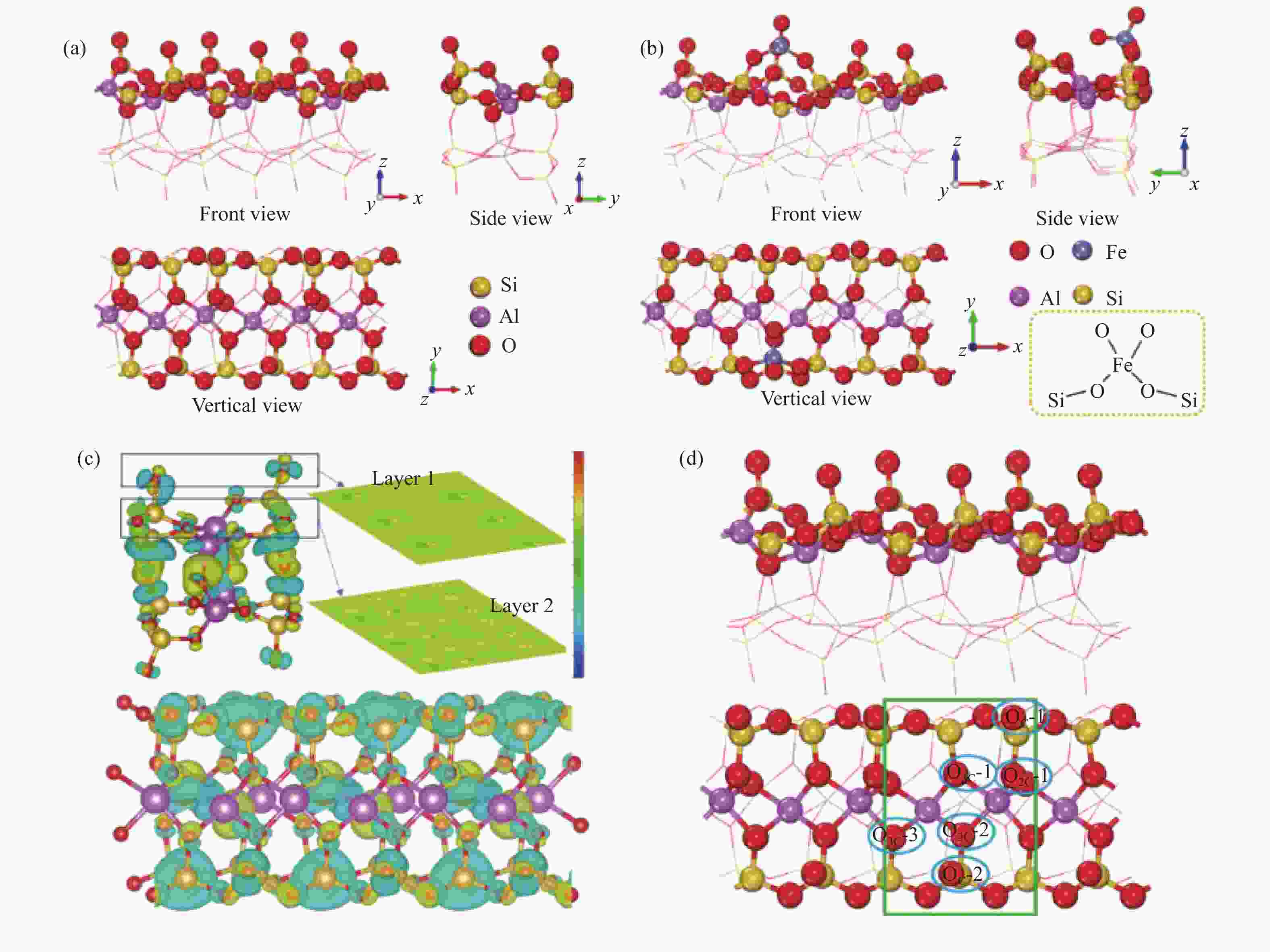
图 8 (a)ATT(110) 和(b)Fe/ATT(110)吸附模型的三视图(c)ATT(110)结构的电荷密度差分图;(d)ATT(110)表面上的晶格氧位点
Figure 8 (a) Three views of ATT(110) and (b) Fe/ATT(110) adsorption models; (c) charge density differential distribution of ATT(110) structure; (d) lattice oxygen sites on the surface of ATT(110) (green box: repeat unit; blue box: test sites)
表 1 吸附剂组成成分分析
Table 1 Analysis of the composition of the adsorbents
Sample Content w/% SiO2 Al2O3 Na2O Fe2O3 TiO2 MgO CaO K2O P2O5 Kaolinite 49.521 39.912 0.063 0.924 0.610 0.350 0.060 1.820 0.089 Attapulgite 62.942 14.274 6.064 5.099 1.265 1.252 1.117 0.848 0.139 表 2 改性吸附剂的物理化学性质
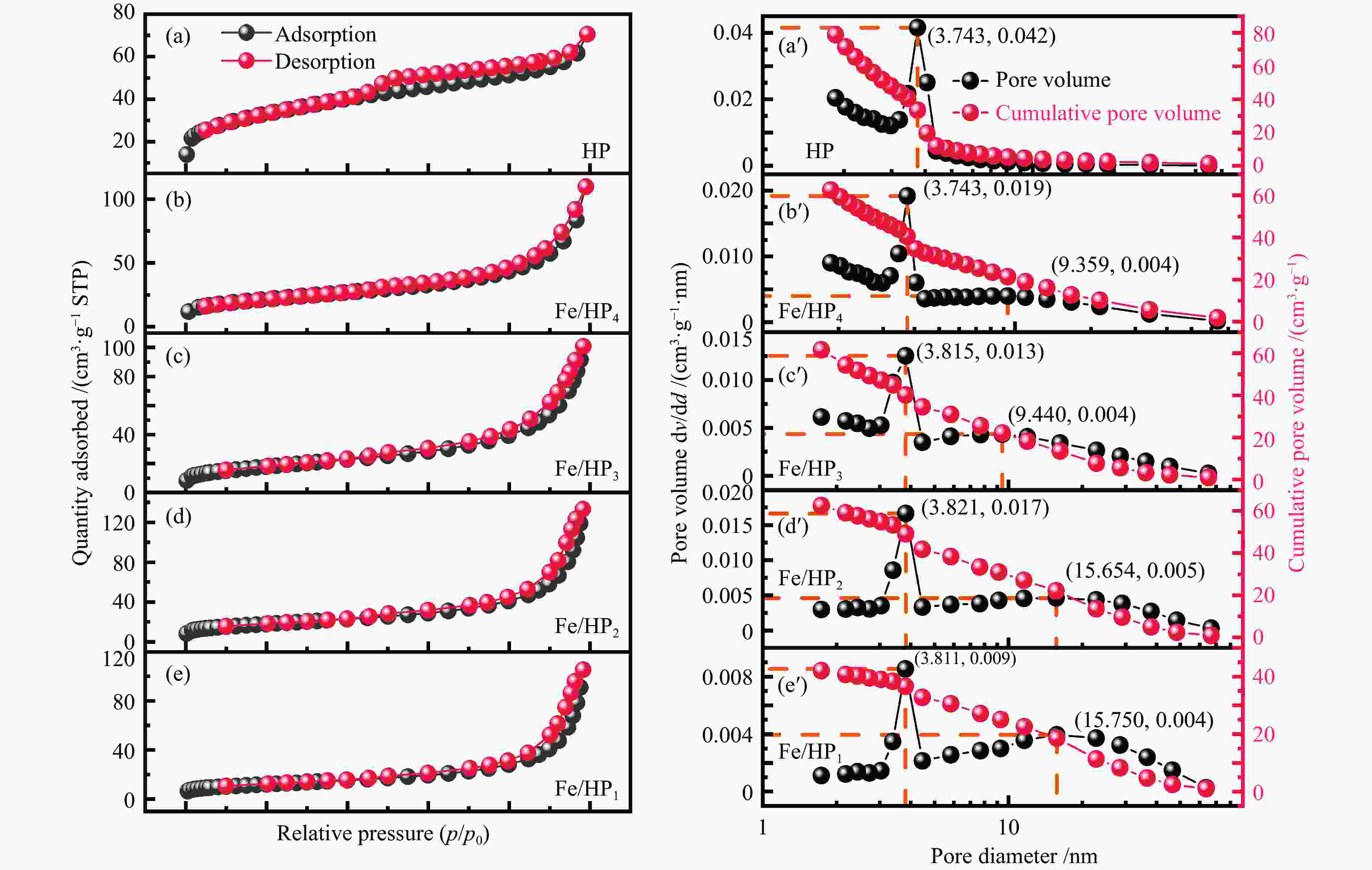
Table 2 Physicochemical characteristics of Modified adsorbents
Sample Specific surface
area/(m2·g−1)Pore volume/
(cm3·g−1)Mean pore
size/nmP 38.1941 0.046592 5.4531 HP 114.0762 0.109196 4.5113 Fe/HP4 76.0169 0.169613 9.9182 Fe/HP3 64.4017 0.156313 10.3605 Fe/HP2 64.2434 0.206309 12.9872 Fe/HP1 43.2412 0.163498 15.3436 表 3 ATT(110)表面吸附气体分子的吸附能和活性位点
Table 3 Adsorption energies and active sites of ATT(110) adsorbed gas molecules
Adsorption energy/
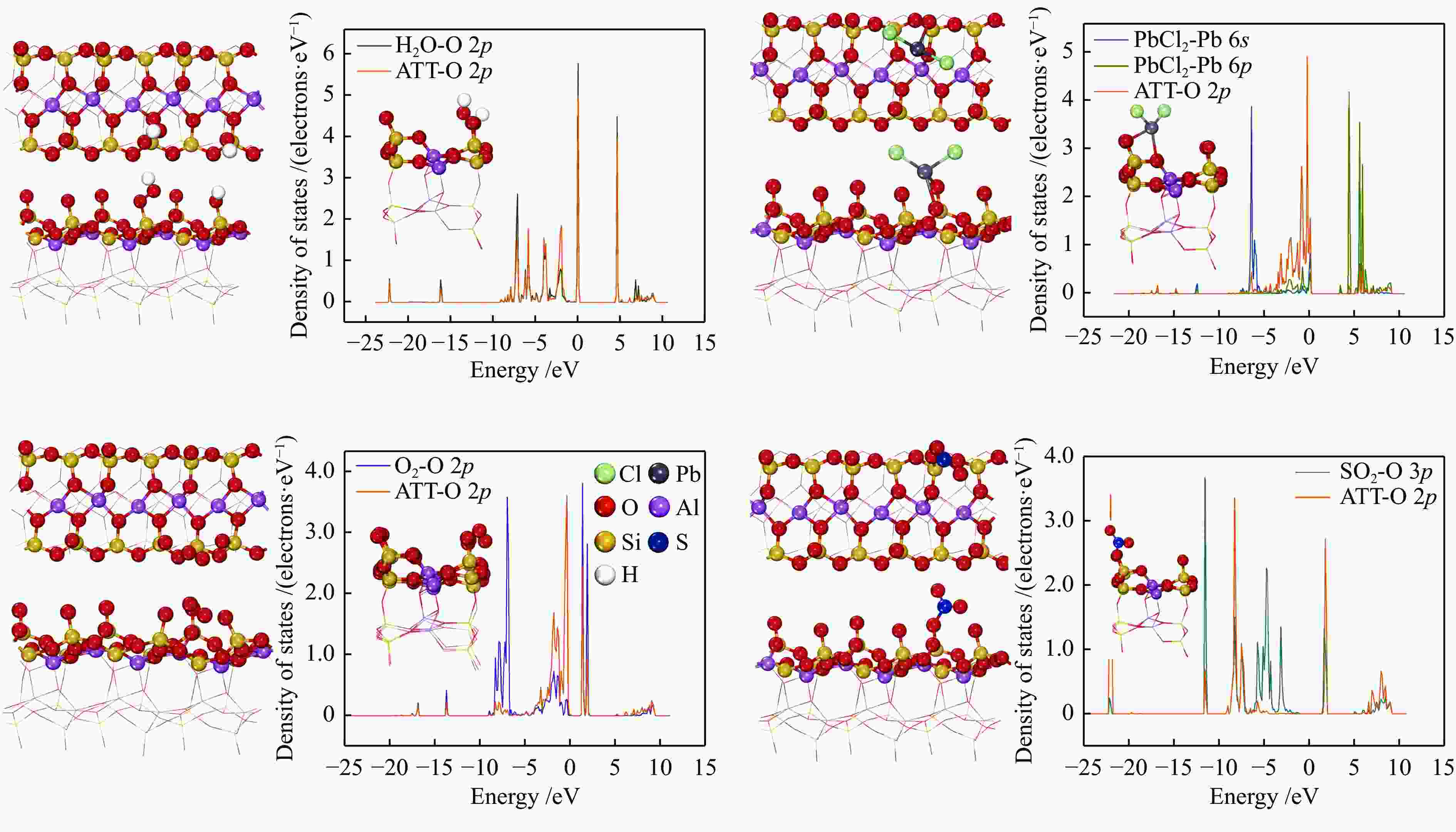
eVAdsorption site O3C-1 O3C-2 O3C-3 O2C-1 OC-1 OC-2 H2O (g) −2.906 −2.420 −1.482 −2.231 −3.962 −3.885 O2 (g) −2.404 −2.213 −2.191 −2.398 −2.821 −2.911 PbCl2 (g) −2.854 −2.378 −2.211 −3.063 −3.327 −3.378 SO2 (g) −0.233 −2.510 −2.589 −2.491 −3.969 −3.998 -
[1] ZHANG J, HUANG X, CHEN Y, LUO B, LUO J, ZHANG W, RAO Z, YANG F. Characterization of lead-containing atmospheric particles in a typical basin city of China: Seasonal variations, potential source areas, and responses to fireworks[J]. Sci Total Environ,2019,661:354−363. doi: 10.1016/j.scitotenv.2019.01.079 [2] AWUAL M R, ISLAM A, HASAN M M, RAHMAN M M, ASIRI A M, KHALEQUE M A, SHEIKH M C. Introducing an alternate conjugated material for enhanced lead (II) capturing from wastewater[J]. J Clean Prod,2019,224:920−929. doi: 10.1016/j.jclepro.2019.03.241 [3] GEORGE A, SHEN B, KANG D, YANG J, LUO J. Emission control strategies of hazardous trace elements from coal-fired power plants in China[J]. J Environ Sci -China,2020,93:66−90. doi: 10.1016/j.jes.2020.02.025 [4] YU S, ZHANG C, MA L, TAN P, FANG Q, CHEN G. Deep insight into the effect of NaCl/HCl/SO2/CO2 in simulated flue gas on gas-phase arsenic adsorption over mineral oxide sorbents[J]. J Hazard Mater,2021,403:123617. doi: 10.1016/j.jhazmat.2020.123617 [5] YU S, ZHANG C, MA L, TAN P, FANG Q, CHEN G. Geochemical mechanism of lead vapors over fly ash cenospheres in simulated flue gas[J]. Fuel,2021,285:119274. doi: 10.1016/j.fuel.2020.119274 [6] WENDT J O, LEE S J. High-temperature sorbents for Hg, Cd, Pb, and other trace metals: Mechanisms and applications[J]. Fuel,2010,89(4):894−903. doi: 10.1016/j.fuel.2009.01.028 [7] XU M, YAN R, ZHENG C, QIAO Y, HAN J, SHENG C. Status of trace element emission in a coal combustion process: A review[J]. Fuel Process. Technol.,2004,85(2/3):215−237. doi: 10.1016/S0378-3820(03)00174-7 [8] WANG J, ZHANG Y, WANG T, XU H, PAN W. Effect of modified fly ash injection on As, Se, and Pb emissions in coal-fired power plant[J]. Chem Eng J,2020,380:122561. doi: 10.1016/j.cej.2019.122561 [9] ZHA J, HUANG Y, CLOUGH P T, XIA Z, ZHU Z, FAN C, YU M, YAN Y, CHENG H. Green production of a novel sorbent from kaolin for capturing gaseous PbCl2 in a furnace[J]. J Hazard Mater,2021,404:124045. doi: 10.1016/j.jhazmat.2020.124045 [10] XU L, LIU Y, WANG J, TANG Y, ZHANG Z. Selective adsorption of Pb2 + and Cu2 + on amino-modified attapulgite: Kinetic, thermal dynamic and DFT studies[J]. J Hazard Mater,2021,404:124140. doi: 10.1016/j.jhazmat.2020.124140 [11] WANG H, HU W, WU Q, HUANG B, ZONG L, WANG A, SIEBECKER M G. Effectiveness evaluation of environmentally friendly stabilizers on remediation of Cd and Pb in agricultural soils by multi-scale experiments[J]. J Clean Prod,2021,311:127673. doi: 10.1016/j.jclepro.2021.127673 [12] XU Y, LIU X, WANG H, ZENG X, ZHANG Y, HAN J, XU M, PAN S. Influences of in-furnace kaolin addition on the formation and emission characteristics of PM2.5 in a 1000 MW coal-fired power station[J]. Environ Sci Technol,2018,52(15):8718−8724. doi: 10.1021/acs.est.8b02251 [13] LIU J, ZENG J, SUN S, HUANG S, KUO J, CHEN N. Combined effects of FeCl3 and CaO conditioning on SO2, HCl and heavy metals emissions during the DDSS incineration[J]. Chem Eng J,2016,299:449−458. doi: 10.1016/j.cej.2016.04.132 [14] WANG X, HUANG Y, PAN Z, WANG Y, LIU C. Theoretical investigation of lead vapor adsorption on kaolinite surfaces with DFT calculations[J]. J Hazard Mater,2015,295:43−54. doi: 10.1016/j.jhazmat.2015.03.020 [15] ZHANG Y, LIU X, XU Y, SUN W, XU M. Investigation of reducing ultrafine particulate matter formation by adding modified montmorillonite during coal combustion[J]. Fuel Process Technol,2017,158:264−271. doi: 10.1016/j.fuproc.2017.01.019 [16] ZHANG X, LIU H, XING H, LI H, HU H, LI A, YAO H. Improved sodium adsorption by modified kaolinite at high temperature using intercalation-exfoliation method[J]. Fuel,2017,191:198−203. doi: 10.1016/j.fuel.2016.11.067 [17] YU S, ZHANG C, ZHANG X, LI X, WEI B, TAN P, FANG Q, CHEN G, XIA J. Release and transformation characteristics of Na/Ca/S compounds of Zhundong coal during combustion/CO2 gasification[J]. J Energy Inst,2020,93(2):752−765. doi: 10.1016/j.joei.2019.05.007 [18] YU S, ZHANG C, YUAN C, XU H, MA L, FANG Q, CHEN G. Investigation on the influence of sulfur and chlorine on the initial deposition/fouling characteristics of a high-alkali coal[J]. Fuel Process Technol,2020,198:106234. doi: 10.1016/j.fuproc.2019.106234 [19] YU S, ZHANG C, MA L, FANG Q, CHEN G. Insight into As2O3 adsorption characteristics by mineral oxide sorbents: Experimental and DFT study[J]. Chem Eng J,2021,420:127593. doi: 10.1016/j.cej.2020.127593 [20] NOWAK B, ROCHA S F, ASCHENBRENNER P, RECHBERGER H, WINTER F. Heavy metal removal from MSW fly ash by means of chlorination and thermal treatment: Influence of the chloride type[J]. Chem Eng J,2012,179:178−185. doi: 10.1016/j.cej.2011.10.077 [21] LIU T, XUE L, GUO X, HUANG Y, ZHENG C. DFT and experimental study on the mechanism of elemental mercury capture in the presence of HCl on α-Fe2O3 (001)[J]. Environ Sci Technol,2016,50(9):4863−4868. doi: 10.1021/acs.est.5b06340 [22] WANG X, HUANG Y, ZHONG Z, PAN Z, LIU C. Theoretical investigation of cadmium vapor adsorption on kaolinite surfaces with DFT calculations[J]. Fuel,2016,166:333−339. doi: 10.1016/j.fuel.2015.11.004 [23] YU S, ZHANG C, MA L, FANG Q, CHEN G. Experimental and DFT studies on the characteristics of PbO/PbCl2 adsorption by Si/Al-based sorbents in the simulated flue gas[J]. J Hazard Mater,2021,407:124742. doi: 10.1016/j.jhazmat.2020.124742 [24] XU Y, LIU X, WANG H, ZHANG Y, QI J, XU M. Investigation of simultaneously reducing the emission of ultrafine particulate matter and heavy metals by adding modified attapulgite during coal combustion[J]. Energy Fuels,2018,33(2):1518−1526. [25] WANG X, HUANG Y, ZHONG Z, YAN Y, NIU M, WANG Y. Control of inhalable particulate lead emission from incinerator using kaolin in two addition modes[J]. Fuel Process Technol,2014,119:228−235. doi: 10.1016/j.fuproc.2013.11.012 [26] CHENG H, HUANG Y, ZHU Z, DONG L, ZHA J, YU M. Enhanced PbCl2 adsorption capacity of modified kaolin in the furnace using a combined method of thermal pre-activation and acid impregnation[J]. Chem Eng J,2021,414:128672. doi: 10.1016/j.cej.2021.128672 [27] WANG L, SHI Y, YAO D, PAN H, HOU H, CHEN J, CRITTENDEN J C. Cd complexation with mercapto-functionalized attapulgite (MATP): adsorption and DFT study[J]. Chem Eng J,2019,366:569−576. doi: 10.1016/j.cej.2019.02.114 [28] POST J E, HEANEY P J. Synchrotron powder X-ray diffraction study of the structure and dehydration behavior of palygorskite[J]. Am Mineral,2008,93(4):667−675. doi: 10.2138/am.2008.2590 [29] ZHAO L, SHEN K, LI B, ZHANG Y, ZHANG S, HONG Y, ZHANG J, LI Z. Exploration of novel high-temperature heavy metals adsorbent for sludge incineration process: Experiments and theoretical calculations[J]. J Environ Chem Eng,2022,10(3):107755. [30] ZHANG W, QIAN L, CHEN Y, OUYANG D, HAN L, SHANG X, LI J, GU M, CHEN M. Nanoscale zero-valent iron supported by attapulgite produced at different acid modification: Synthesis mechanism and the role of silicon on Cr (VI) removal[J]. Chemosphere,2021,267:129183. doi: 10.1016/j.chemosphere.2020.129183 [31] ZHANG T, WANG W, ZHAO Y, BAI H, WEN T, KANG S, SONG G, SONG S, KOMARNENI S. Removal of heavy metals and dyes by clay-based adsorbents: From natural clays to 1D and 2D nano-composites[J]. Chem Eng J,2021,420:127574. doi: 10.1016/j.cej.2020.127574 [32] LIU Y, LIU P, SU Z, LI F, WEN F. Attapulgite-Fe3O4 magnetic nanoparticles via co-precipitation technique[J]. Appl. Surf. Sci. 2008, 255(5, Part 1): 2020−2025. [33] PAN J, XU L, DAI J, LI X, HANG H, HUO P, LI C, YAN Y. Magnetic molecularly imprinted polymers based on attapulgite/Fe3O4 particles for the selective recognition of 2, 4-dichlorophenol[J]. Chem Eng J,2011,174(1):68−75. doi: 10.1016/j.cej.2011.08.046 [34] ZHU L, GUO J, LIU P, ZHAO S. Novel strategy for palygorskite/poly (acrylic acid) nanocomposite hydrogels from bi-functionalized palygorskite nanorods as easily separable adsorbent for cationic basic dye[J]. Appl Clay Sci,2016,121:29−35. [35] LIU Y, LIU P, SU Z, LI F, WEN F. Attapulgite-Fe3O4 magnetic nanoparticles via co-precipitation technique[J]. Appl Surf Sci,2008,255(5):2020−2025. doi: 10.1016/j.apsusc.2008.06.193 [36] TANG J, MU B, ZONG L, WANG A. One-step synthesis of magnetic attapulgite/carbon supported NiFe-LDHs by hydrothermal process of spent bleaching earth for pollutants removal[J]. J Clean Prod,2018,172:673−685. doi: 10.1016/j.jclepro.2017.10.181 [37] LIU J, ZHANG J, XING L, WANG D, WANG L, XIAO H, KE J. Magnetic Fe3O4/attapulgite hybrids for Cd (II) adsorption: performance, mechanism and recovery[J]. J Hazard Mater,2021,412:125237. doi: 10.1016/j.jhazmat.2021.125237 -




 下载:
下载: