Effect of Ce doping on the performance of MoSn catalyst in the selective oxidation of dimethyl ether to methyl formate
-
摘要: 采用无沉淀剂一步水热法合成了不同含量Ce掺杂的MoSn催化剂,考察了Ce的掺杂量对其二甲醚(DME)选择氧化催化性能的影响。结果表明,当Ce含量为0.5%时,MoSn催化剂的活性最高,在130℃下DME的转化率达11.8%,MF的选择性为92.2%。采用氮吸附、XRD、NH3-TPD、CO2-TPD、H2-TPR、XPS和in situ-IR等表征技术对催化剂进行了表面性质和结构分析,发现Ce的掺杂虽然没有从根本上改变MoSn催化剂的结构,但明显提高了MoSn催化剂中五价钼的含量。Abstract: A series of MoSn catalysts doped with different Ce loadings were prepared by hydrothermal method and their catalytic performance in the selective oxidation of dimethyl ether (DME) to methyl formate (MF) was investigated. The results indicate that the introduction of 0.5% Ce can significantly improve the activity of the MoSn catalyst over the 0.5%Ce-MoSn catalyst, the conversion of DME reaches 11.8% at 130 ℃, with a selectivity of 92.2% for MF. The MoSn catalysts were characterized by nitrogen sorption, XRD, NH3-TPD, CO2-TPD, H2-TPR, XPS and in situ-IR; the results illustrate that a small amount of Ce in the MoSn catalyst can obviously increase the quantity of Mo5+, though it has little effect on the structure of the MoSn catalyst.
-
Key words:
- MoSn catalyst /
- dimethyl ether /
- selective oxidation /
- methyl formate /
- hydrothermal method
-
表 1 Ce含量对MoSn催化剂上DME氧化制备甲酸甲酯的影响
Table 1 Effect of the Ce loading on the activity of Ce-doped MoSn catalyst in the selective oxidation of DME to MF
Ce
wmol/%DME
con. x/ %Selectivity sC-mol/% MF CH3OH CO FA CO2 0 10.3 84.6 14.3 1.1 0 0 0.5 11.8 92.2 6.1 1.7 0 0 1 10.6 84.5 14.1 1.4 0 0 5 7.8 78.2 21.1 0.7 0 0 10 6. 8 76. 2 23. 4 0. 4 0 0 reaction conditions: 130 ℃; GHSV = 1800 h-1; DME/O2 = 1; Mo/Sn = 1:2 表 2 反应温度对0.5%Ce-Mo1Sn2催化剂上DME氧化制备甲酸甲酯的影响
Table 2 Effect of reaction temperature on the selective oxidation of DME to MF over the 0.5%Ce-Mo1Sn2 catalyst
tR/℃ DME
con. x/ %Selectivity sC-mol/% MF CH3OH CO FA CO2 150 90.2 1.2 0.3 98.5 0 0 150* 10.8 78.2 8.3 13.5 0 0 140 15.4 86.9 0.7 12.4 0 0 130 11.8 92.2 6.1 1.7 0 0 120 7.8 89.5 4.4 6.1 0 0 110 7.2 77.1 17.6 5.3 0 0 100 5.8 65.2 31.6 3.2 0 0 reaction conditions: GHSV = 1800 h-1; DME/O2 = 1; Mo/Sn=1:2; *: for the reaction over the undoped Mo1Sn2 catalyst 表 3 不同Ce含量的MoSn催化剂的织构性质
Table 3 Textural properties of the Ce-doped MoSn catalysts with different Ce loadings
Catalyst Surface area A / (m2·g-1) Pore volume v / (cm3·g-1) Average pore diameter d / nm Mo1Sn2 141.16 0.118 3.52 0.5%Ce-Mo1Sn2 140.87 0.139 3.57 5% Ce-Mo1Sn2 136.05 0.130 3.61 10% Ce-Mo1Sn2 120.07 0.112 3.48 表 4 不同Ce含量的MoSn催化剂XPS-Mo 3d谱图
Table 4 Analysis of the Mo 3d XPS spectra of the Ce-doped MoSn catalysts with different Ce loadings
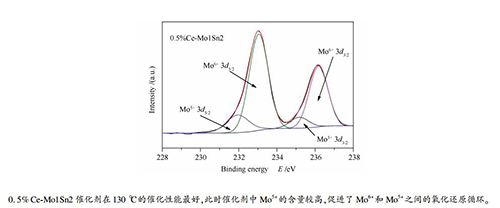
Catalyst Mo6+3d3 / 2 Mo6+3d5 / 2 Mo5+3d3 / 2 Mo5+3d3 / 2 Mo6+ / % Mo5+ / % Mo1Sn2 236.18 233.05 235.17 231.95 82.6 17.4 0. 5% Ce-Mo1Sn2 236.18 233.05 235.17 231.95 79.9 20.1 5% Ce-Mo1Sn2 236.12 233.05 235.17 231.95 83.1 16.9 -
[1] WANG D S, HAN Y Z, TAN Y S, TSUBAKI N. Effect of H2O on Cu-based catalyst in one-step slurry phase dimethyl ether synthesis[J]. Fuel Process Technol, 2009, 90(3):446-451. doi: 10.1016/j.fuproc.2008.11.007 [2] 孙明, 余林, 孙长勇, 宋一兵, 孙健.二甲醚的应用及下游产品开发[J].精细化工, 2003, 20(11):695-699. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jxhg200311017SUN Ming, YU Lin, SUN Chang-yong, SONG Yi-bing, SUN Jian. Application of dimethyl ether and development of its downstream products[J]. Fine Chem, 2003, 20(11):695-699. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jxhg200311017 [3] 黄秀敏, 徐奕德, 申文杰.负载型MoOx和VOx催化剂上二甲醚选择氧化制甲醛反应[J].催化学报, 2004, 25(4):267-271. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cuihuaxb200404006HUANG Xiu-min, XU yi-de, SHEN Wen-jie. Selective oxidation of dimethyl ether to formaldehyde over supported MoOx and VOx catalysts[J]. Chin J Catal, 2004, 25(4):267-271. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cuihuaxb200404006 [4] HUANG X M, LIU J L, CHEN J L, XU Y D, SHEN W J. Mechanistic study of selective oxidation of dimethyl ether to formaldehyde over Alumina-supported molybdenum oxide catalyst[J]. Catal Lett, 2006, 108(1/2):79-86. doi: 10.1007/s10562-006-0019-9 [5] LIU H C, CHEUNG P, IGLESIA E. Structure and support effects on the selective oxidation of dimethyl ether to formaldehyde catalyzed by MoOx domains[J]. J Catal, 2003, 217(1):222-232. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=180d6d3f82f0282854bdd2714ae015e2 [6] LIU H C, IGLESIA E. Selective oxidation of dimethyl ether to formaldehyde on small molybdenum oxide domains[J]. J Catal, 2002, 208(1):1-5. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=7d22cf6c8d4e827a2925e781c26f5eeb [7] 汪多仁.甲酸甲酯的生产发展与应用前景[J].石油与天然气化工, 1998, 27(3):149-151. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199800540043WANG Duo-ren. Production development and application prospects of methyl formate[J]. Chem Eng Oil Gas, 1998, 27(3):149-151. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199800540043 [8] 周寿祖.甲酸甲酯的生产技术和应用前景[J].化工科技市场, 2003, 26(2):13-18. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgkjsc200302004ZHOU Shou-zu. Production technology and application foreground of methyl formate[J]. Chem Technol Market, 2003, 26(2):13-18. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgkjsc200302004 [9] 李振香.浅谈甲醇羰基化合成甲酸甲酯[J].小氮肥设计技术, 2003, 24(1):60-62. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=xdfsjjs200301023LI Zhen-xiang. Brief discussion on methanol carbonylation to synthesize methyl formate[J].Small Nitrogen Fert Des Technol, 2003, 24(1):60-62. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=xdfsjjs200301023 [10] 汪青松, 李国儒, 李工.研磨法和浸渍法制备Cu/SBA-15和Cu-ZnO/SBA-15及对甲醇脱氢制甲酸甲酯的催化性能[J].常州大学学报, 2014, 26(1):23-26. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jssyhgxyxb201401006WANG Qing-song, LI Guo-ru, LI Gong. Catalytic activity of methanol dehydrogenation to MF over Cu/SBA-15 and Cu-ZnO/SBA-15 prepared by grinding and impregnation[J]. J Changzhou Univ, 2014, 26(1):23-26. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jssyhgxyxb201401006 [11] TONNER S P, TRIMM D L, WAINWEIGHT M S, CANT N W. Dehydrogenation of methanol to methyl formate over copper-catalysts[J]. Ind Eng Chem Prod Res Dev, 1984, 23(3):384-388. doi: 10.1021/i300015a012 [12] AI M. The production of methyl formate by the vapor-phase oxidation of methanol[J]. Chem Technol Market, 2003, 26(2):13-18. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=e3f3f3492468b3d76c12c7ba1bdb1945 [13] AI M. The reaction of formaldehyde on various metal-oxide catalysts[J]. J Catal, 1982, 77(1):279-288. http://www.sciencedirect.com/science/article/pii/0021951783900374 [14] 刘广波, 张清德, 韩怡卓, 椿范立, 谭猗生. MoSn催化剂上二甲醚低温氧化高选择性制备甲酸甲酯[J].燃料化学学报, 2013, 41(2):223-227. http://www.ccspublishing.org.cn/article/id/11894LIU Guang-bo, ZHANG Qing-de, HAN Yi-zhuo, TSUBAKI Noritatsu, TAN Yi-sheng. Low-temperature oxidation of dimethyl ether to methyl formate with high selectivity over MoSn catalysts[J]. J Fuel Chem Technol, 2013, 41(2):223-227. http://www.ccspublishing.org.cn/article/id/11894 [15] LIU G B, ZHANG Q D, HAN Y Z, TSUBAKI N, TAN Y S. Selective oxidation of dimethyl ether to methyl formate over trifunctional MoSn catalyst under mild conditions[J]. Green Chem, 2013, 15(6):1501-1504. doi: 10.1039/c3gc40279g [16] LIU G B, ZHANG Q D, HAN Y Z, TSUBAKI N, TAN Y S. Effects of the MoO3 structure of Mo-Sn catalysts on dimethyl ether oxidation to methyl formate under mild conditions[J]. Green Chem, 2015, 17(2):1057-1064. doi: 10.1039/C4GC01591F [17] ZHANG Z Z, ZHANG Q D, HAN Y Z, TSUBAKI N, TAN Y S. The effects of the Mo-Sn contact interface on the oxidation reaction of dimethyl ether to methyl formate at a low reaction temperature[J]. Catal Sci Technol, 2016, 6(15):6109-6117. doi: 10.1039/C6CY00460A [18] ZHANG Z Z, ZHANG Q D, HAN Y Z, TSUBAKI N, TAN Y S. Effect of MoO3 crystalline structure of MoSn catalysts on selective oxidation of glycol dimethyl ether to 1, 2-propandiol[J]. 2016, 6(6):1842-1849. [19] ZHANG Z Z, ZHANG Q D, HAN Y Z, TSUBAKI N, TAN Y S. Effect of tetrahedral molybdenum oxide species and MoOx domains on the selective oxidation of dimethyl ether under mild condition[J]. Catal Sci Technol, 2016, 6(9):2975-2983. doi: 10.1039/C5CY01569C [20] 杨奇, 高秀娟, 冯茹, 李明杰, 张俊峰, 张清德, 韩怡卓, 谭猗生.水热合成的MoSn催化剂催化氧化二甲醚的性能研究[J].燃料化学学报, 2019, 47(8):934-941. http://www.ccspublishing.org.cn/article/id/f54ec827-f8ff-45c8-9d34-ac36adcfacf3YANG Qi, GAO Xiu-juan, FENG Ru, LI Ming-jie, ZHANG Jun-feng, ZHANG Qing-de, HAN Yi-zhuo, TAN Yi-sheng. MoSn catalyst prepared by hydrothermal synthesis method for dimethyl ether catalytic oxidation[J]. J Fuel Chem Technol, 2019, 47(8):934-941. http://www.ccspublishing.org.cn/article/id/f54ec827-f8ff-45c8-9d34-ac36adcfacf3 [21] STAMPF S, CHEN Y, DUMESIC J A, HILL C G. Interactions of molybdenum oxide with various oxide supports:Calcination of mechanical mixtures[J]. J Catal, 1987, 105(2):445-454. http://www.sciencedirect.com/science/article/pii/0021951787900728 [22] MENG Y L, WANG T, CHEN S, GONE J L. Selective oxidation of methanol to dimethoxymethane on V2O5-MoO3/γ-Al2O3 catalysts[J]. Appl Catal B:Environ, 2014, 160-161(1):161-172. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=de3bc6bf39bb6c7cf988ad6f4ae76b0e [23] WHITING G T, KONDRAT S A, HAMMOND C, DIMITRATOS N, HUTCHINGS G J. Methyl formate formation from methanol oxidation using supported Gold-Palladium nanoparticles[J]. ACS Catal, 2015, 5(2):637-644. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=abc2df00fda2795c07bf97bc61991bc3 [24] GONCALVES F, MEDEIROS P R S, EON J G, APPEL L G. Active sites for ethanol oxidation over SnO2-supported molybdenum oxides[J]. Appl Catal A:Gen, 2000, 193(1/2):195-202. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=3e9f3819bf721b2b4608bb2ff36b370f [25] HABER J, LALIK E. Catalytic properties of MoO3 revisited[J]. Catal Today, 1997, 33(1/3):119-137. http://www.sciencedirect.com/science/article/pii/S0920586196001071 -





 下载:
下载: