Catalytic oxidation of soot over monovalent copper modified ZSM-5
-
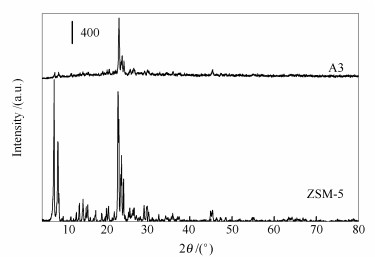
摘要: 采用固态离子交换法制备了系列一价铜改性的ZSM-5催化剂,结合多种表征手段,研究了一价铜改性对碳烟氧化反应催化活性的影响。结果表明,采用固态离子交换法可以制备出高负载量的一价铜改性Cu/ZSM-5分子筛催化剂,而不会破坏ZSM-5分子筛原有微孔结构;随催化剂中一价铜比例的增加,低温还原峰和高温还原峰均向低温段移动,且低温还原峰面积增加。改性催化剂对碳烟氧化反应的催化活性随改性元素比例变化先增加后降低;当铜改性比例超过11%后,铜物种的分散性及催化剂对碳烟氧化反应的催化活性均恶化。同时研究还发现,反应气氛中通入NO可以改善催化剂对碳烟氧化反应的催化效果。Abstract: ZSM-5 zeolites were modified with different contents of monovalent copper by solid-state ion exchange method and used as the catalysts in soot oxidation; the effect of monovalent copper content on the catalytic performance was investigated through extensive characterization. The results indicate that high content of monovalent copper can be loaded on ZSM-5 zeolite by the solid-state ion exchange method, whereas has little detriment to the original micro-structure of ZSM-5. With the increase of copper content in catalyst, the reduction peaks at low temperature and high temperature both shift to lower temperature, while the area of low temperature reduction peak is increased. The catalytic activity of Cu-modified zeolite in soot oxidation decreases after an initial increase with the increase of Cu content. When the Cu loading exceeds 11%, the dispersion of copper species deteriorates, accompanying with a decrease in the catalytic activity for soot oxidation. Meanwhile, the addition of NO to the feed (O2/He) can enhance the soot oxidation.
-
表 1 不同比例一价铜改性催化剂
Table 1 Numbering of modified ZSM-5 catalysts with different contents of monovalent copper
Numbers A0 A1 A2 A3 A4 w(Cu)/% 0 5.0 9.0 11.0 13.0 表 2 Cu/ZSM-5系列催化剂的氧化还原性质
Table 2 Redox properties of Cu/ZSM-5 catalysts
Sample δ peak α peak γ peak β peak Total uptake
/(μmol·g-1)t/℃ H2 uptake
/(μmol·g-1)t/℃ H2 uptake
/(μmol·g-1)t/℃ H2 uptake
/(μmol·g-1)t/℃ H2 uptake
/(μmol·g-1)A1 174.5 4.778 258.9 18.066 357.6 2.441 694.1 10.498 35.783 A2 173.1 8.789 252.5 19.531 335.4 7.568 535.6 22.705 58.593 A3 168.7 10.393 249.0 24.658 340.0 8.301 448.5 26.123 69.475 A4 170.9 4.395 263.7 23.403 351.3 12.033 431.9 25.879 65.710 表 3 O2/He气氛下Cu/ZSM-5系列催化剂对碳烟氧化反应的催化性能
Table 3 Catalytic performance in soot oxidation of various Cu/ZSM-5 catalysts in O2/He atmosphere
Sample In O2/He atmosphere ti/℃ tm/℃ tf/℃ Δt/℃ A0 373 621 691 318 A1 275 532 650 375 A2 287 520 646 359 A3 263 458 597 334 A4 314 516 634 320 表 4 NO/O2/He气氛下Cu/ZSM-5系列催化剂对碳烟氧化反应的催化性能
Table 4 Catalytic performance in soot oxidation of various Cu/ZSM-5 catalysts in NO/O2/He atmosphere
Sample In NO/O2/He atmosphere ti/℃ tm/℃ tf/℃ Δt/℃ A0 291 605 670 379 A1 177 336 425 248 A2 165 324 442 277 A3 144 315 407 263 A4 156 319 413 257 -
[1] ALKEMADE U G, SCHUMANN B. Engines and exhaust after treatment systems for future automotive applications[J]. Solid State Ion, 2006, 177(26/32):2291-2296. http://www.sciencedirect.com/science/article/pii/S0167273806003304 [2] HOHL Y. Retrofit kit to reduce NOx and PM emissions from diesel engines using a low-pressure EGR and a DPF-system with FBC and throttling for active regeneration without production of secondary emissions[J]. Int J Bilingualism, 2015, 19(293):627-645. [3] FAYAD M, HERREROS J M, MARTOS F J, TSOLAKIS A. The role of alternative fuels on pm characteristics and influence of the diesel oxidation catalyst[J]. Environ Sci Technol, 2015, 49(19):11967-11973. doi: 10.1021/acs.est.5b02447 [4] XU J F, LIU J, ZHAO Z, XU C M, ZHENG J X, DUAN A J, JIANG G Y. Easy synthesis of three-dimensionally ordered microporous La1-xKxCoO3 catalysts and their high activities for the catalytic combustion of soot[J]. J Catal, 2011, 282(1):1-12. doi: 10.1016/j.jcat.2011.03.024 [5] SEO P W, CHO S P, HONG S H, HONG S C. The influence of lattice oxygen in titania on selective catalytic reduction in the low temperature region[J]. Appl Catal A:Gen, 2010, 380(1):21-27. https://www.researchgate.net/publication/239153973_The_influence_of_lattice_oxygen_in_titania_on_selective_catalytic_reduction_in_the_low_temperature_region [6] OLSSON L, WIJAYANTI K, LEISTNER K, KUMAR A, JOSHI S Y, KAMASAMUDRAM K, NEAL W C, ALEKSEY Y. A kinetic model for sulfur poisoning and regeneration of Cu/SSZ-13 used for NH3-SCR[J]. Appl Catal B:Environ, 2015, 183:394-406. https://www.researchgate.net/publication/283686566_A_kinetic_model_for_sulfur_poisoning_and_regeneration_of_CuSSZ-13_used_for_NH3-SCR [7] MA A J, WANG S Z, CHENG L, XIAN H, DING Q, GUO L, MENG M, TAN Y S, TSUBAKI N, ZHANG J, ZHENG L D, LI X G. Effects of Fe dopants and residual carbonates on the catalytic activities of the perovskite-type La 0.7 Sr0.3 Co1-xFexO3, NOx, storage catalyst[J]. Appl Catal B:Environ, 2014, 146(5):24-34. [8] IWASAKI S, MIZUTANI T, MIYAIRI Y, YUUKI K, MAKINO M. New design concept for diesel particulate filter[J]. SAE Int J Engines, 2011, 4(1):527-536. doi: 10.4271/2011-01-0603 [9] YOSHIDA K, MAKINO S, SUMIYA S. Simultaneous reduction of NOx and particulate emissions from diesel engine exhaust[J]. SAE Paper, 1989, 19892046. https://www.researchgate.net/profile/Dennis_Assanis/publication/300833582_Simultaneous_Reduction_of_NOX_and_Soot_in_a_Heavy-Duty_Diesel_Engine_by_Instantaneous_Mixing_of_Fuel_and_Water/links/578d131808ae254b1de86f7d.pdf?origin=publication_detail [10] YIN F, JI S, WU P, ZHAO F, LI C, Deactivation behavior of Pd-based SBA-15 mesoporous silica catalysts for the catalytic combustion of methane[J]. J Catal, 2008, 257(1):108-116. doi: 10.1016/j.jcat.2008.04.010 [11] FIERRO G, MORETTI G, FERRARIS G, ANDREOZZI G B. A Mössbauer and structural investigation of Fe-ZSM-5 catalysts:Influence of Fe oxide nanoparticles size on the catalyticbehaviour for the NO-SCR by C3H8[J]. Appl Catal B:Environ, 2011, 102(1):215-223. [12] BIN F, SONG C L, LV G, SONG J O, WANG K P, LI X D. Soot low-temperature combustion on Cu-Zr/ZSM-5 catalysts in O2/He and NO/O2/He atmospheres[J]. Proc Combust Inst, 2013, 34(2):2303-2311. doi: 10.1016/j.proci.2012.07.075 [13] SCHWIDDER M, SANTHOSH K M, BRVCKNER A, GRVERT W. Active sites for NO reduction over Fe-ZSM-5 catalysts[J]. Chem Commun, 2005, 6(6):805-807. https://www.researchgate.net/publication/8047732_Active_sites_for_NO_reduction_over_Fe-ZSM-5_catalysts [14] SONG Z X, ZHANG Q L, NING P, LIU X, ZHANG J H, WANG Y C, XU L S, HUANG Z Z. Effect of copper precursors on the catalytic activity of Cu/ZSM-5 catalysts for selective catalytic reduction of NO by NH3[J]. Res Chem Intermed, 2016, 42(10):7429-7445. doi: 10.1007/s11164-016-2545-4 [15] YUAN E, ZHANG K, LU G, MO Z, TANG Z. Synthesis and application of metal-containing ZSM-5 for the selective catalytic reduction of NOx, with NH3[J]. J Ind Eng Chem, 2016, 42:142-148. doi: 10.1016/j.jiec.2016.07.030 [16] KANG W, CHOI B, KIM H. Characteristics of the simultaneous removal of PM and NOx, using CuNb-ZSM-5 coated on diesel particulate filter[J]. J Ind Eng Chem, 2013, 19(4):1406-1412. doi: 10.1016/j.jiec.2013.01.004 [17] TRONCONI E, NOVA I, MARCHITTI F, KOLTSAKIS G, KARAMITROS D, MALETIC B, MARKERT N, CHATTERJEE D, HEHLE M. Interaction of NOx, reduction and soot oxidation in a DPF with Cu-zeolite SCR coating[J]. Emis Control Sci Technol, 2015, 1(2):134-151. doi: 10.1007/s40825-015-0014-y [18] SHAKYA B M, HAROLD M P, BALAKOTAIAH V. Simulations and optimization of combined Fe-and Cu-zeolite SCR monolith catalysts[J]. Chem Eng J, 2015, 278:374-384. doi: 10.1016/j.cej.2014.11.029 [19] HUANG L H, ZHANG F B, WANG N, CHEN R H, ANDREW T H. Nickel-based perovskite catalysts with iron-doping via self-combustion for hydrogen production in auto-thermal reforming of ethanol[J]. Int J Hydrogen Energy, 2012, 37(2):1272-1279. doi: 10.1016/j.ijhydene.2011.10.005 [20] URQUIETAGONZÁLEZ, MARTINS E A, PEGUIN L, BATISTA RPS S M. Identification of extra-framework species on Fe/ZSM-5 and Cu/ZSM-5 catalysts typical microporous molecular sieves with zeolitic structure[J]. Mater Res, 2002, 5(3):321-327. doi: 10.1590/S1516-14392002000300017 [21] SCHWIDDER M, SANTHOSH K M, BRVCKNER A, GRVNERT W. Active sites for NO reduction over Fe-ZSM-5 catalysts[J]. Chem Commun, 2015, 6:805-807. https://www.researchgate.net/publication/8047732_Active_sites_for_NO_reduction_over_Fe-ZSM-5_catalysts [22] PANG L, FAN C, SHAO L N, SONG K P, YI J X, CAI X, WANG J, KANG M, LI T. The Ce doping Cu/ZSM-5 as a new superior catalyst to remove NO from diesel engine exhaust[J]. Chem Eng J, 2014, 25(7):394-401. http://www.sciencedirect.com/science/article/pii/S1385894714006652 [23] DEKA U, LEZCANOGONZALEZ I, WECKHUYSEN B M, BEALE A M. Local environment and nature of Cu active sites in zeolite-based catalysts for the selective catalytic reduction of NOx[J]. ACS Catal, 2013, 3(3):413-427. doi: 10.1021/cs300794s [24] ABOUL-GHEIT A K, ABOUL-FOTOUH S M, ABDEL-HAMID S M, ABOUL-GHEIT N A K. Hydroconversion of cyclohexene using H-ZSM-5 zeolite catalysts promoted via hydrochlorination and/or platinum incorporation[J]. J Mol Catal A:Chem, 2006, 245(1):167-177. https://www.researchgate.net/publication/244278304_Hydroconversion_of_cyclohexene_using_H-ZSM-5_zeolite_catalysts_promoted_via_hydrochlorination_andor_platinum_incorporation [25] GUILLÉN-HURTADO N, BUENO-LÓPEZ A, GARCÍA-GARCÍA A. Catalytic performances of ceria and ceria-zirconia materials for the combustion of diesel soot under NOx/O2, and O2. Importance of the cerium precursor salt[J]. Appl Catal A:Gen, 2012, 437-438:166-172. doi: 10.1016/j.apcata.2012.06.028 -





 下载:
下载: