Preparation of monolithic cordierite supported Cu-SSZ-13 catalyst and its performance in the selective catalytic reduction of NOx with NH3
-
摘要: 以聚乙烯醇(PVA)或拟薄水铝石(SB粉)为涂覆助剂,采用超声波辅助分散法将Cu-SSZ-13分子筛涂覆于蜂窝状堇青石(Cordierite)上,制备了整体式堇青石负载的Cu-SSZ-13分子筛催化剂(Cu-SSZ-13/Cordierite),用于氨选择性催化还原(NH3-SCR)脱硝,并结合XRD、氮气吸附、SEM及H2-TPR等表征手段,研究了涂覆助剂对该催化剂的Cu-SSZ-13涂层稳定性、NH3-SCR脱硝活性、水热稳定性和抗SO2毒化能力的影响。结果表明,以PVA为涂覆助剂制备的整体式Cu-SSZ-13(PVA)/Cordierite催化剂,其Cu-SSZ-13涂层稳定、脱落率低,选择性催化还原脱硝活性与Cu-SSZ-13原粉基本相当;同时,该Cu-SSZ-13(PVA)/Cordierite催化剂也具有较好的耐高温水热稳定性和抗硫中毒能力,在移动源和固定源脱硝方面有较好应用前景。Abstract: A series of monolithic cordierite supported Cu-SSZ-13 catalysts (Cu-SSZ-13/Cordierite) was prepared by coating the Cu-SSZ-13 molecular sieves on the cellular cordierite through ultrasonic dispersion with polyvinyl alcohol (PVA) or pseudo boehmite powder (SB) as assisting agent and used in the selective catalytic reduction of NOx with NH3 (NH3-SCR). With the help of XRD, nitrogen sorption, SEM and H2-TPR characterization techniques, the influence of coating assistant agent on the firmness of the Cu-SSZ-13 layer, catalytic activity in the NH3-SCR of NO, hydrothermal stability and resistance against SO2 poisoning was then investigated. The results indicate that by using PVA as the coating assistant agent, a firm layer of Cu-SSZ-13 can be formed on the surface of monolithic Cu-SSZ-13/Cordierite catalyst. Moreover, the Cu-SSZ-13(PVA)/Cordierite exhibits high catalytic activity in the NH3-SCR of NOx (close to that of the pristine Cu-SSZ-13 molecular sieve), high hydrothermal stability and good tolerance to SO2, displaying great potential as a practicable catalyst for the removal of NOx from exhausts of both the mobile and the stationary sources.
-
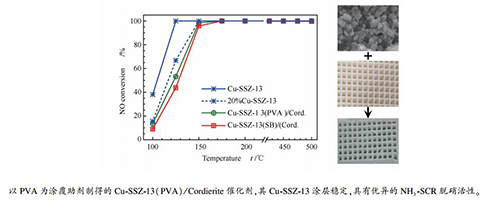
图 1 堇青石载体和Cu-SSZ-13/Cordierite催化剂的图片和SEM照片
Figure 1 Photographs of the original cordierite support (a), Cu-SSZ-13(PVA)/Cordierite catalyst (b), and Cu-SSZ-13(SB)/Cordierite catalyst (c); SEM images of the Cu-SSZ-13 molecular sieve (d), the original cordierite (e), the acid-treated cordierite (f), the Cu-SSZ-13(PVA)/Cordierite catalyst (g), and the Cu-SSZ-13(SB)/Cordierite catalyst (h)
图 4 Cu-SSZ-13分子筛和Cu-SSZ-13/Cordierite催化剂的NH3-SCR活性比较
Figure 4 A comparison of the pristine Cu-SSZ-13 molecular [JP2]sieve (a), Cu-SSZ-13 (20%) + quartz sand (b); Cu-SSZ-13[JP](PVA)/Cordierite catalyst (c), Cu-SSZ-13(SB)/Cordierite catalyst (d), Cu-SSZ-13(PVA)/Cordierite catalyst subjected to hydrothermal treatment at 800 ℃ for 12 h (e), and Cu-SSZ-13(SB)/Cordierite catalyst subjected to hydrothermal treatment at 800 ℃ for 12 h (f), in their activity for the selective catalytic reduction (SCR) of NO by NH3 (0.05% NH3, 0.05% NO, 6% O2 and balanced N2, with a space velocity of 20000 h-1)
图 5 Cu-SSZ-13/Cordierite催化剂用于NH3-SCR脱硝时的抗水和耐SO2性能
Figure 5 NO conversion for the NH3-SCR over the Cu-SSZ-13(PVA)/Cordierite (a) and Cu-SSZ-13(SB)/Cordierite (b), for the NH3-SCR in the presence of 5% water over the Cu-SSZ-13(PVA)/Cordierite (c) and Cu-SSZ-13(SB)/Cordierite (d), and for the NH3-SCR in the presence of 0.01%SO2 over the Cu-SSZ-13(PVA)/Cordierite (e) and Cu-SSZ-13(SB)/Cordierite (f)
表 1 涂覆助剂及涂覆次数对堇青石上Cu-SSZ-13涂层增量及涂层稳定性的影响
Table 1 Influence of coating assistant agent and coating times on the weight increment of coating and the stability of Cu-SSZ-13 layer on the cordierite support
Coating
assistant agentCoating weight increment /% Cu-SSZ-13
content /%Coating exfoliation rate /% 1st 2nd 3rd total 1st 2nd 3rd overall PVA 6.2 6.9 7.6 20.7 20.7 4.9 0.7 0.1 5.7 SB 14.0 15.2 - 29.2 20.1 7.7 2.3 0.7 10.7 表 2 堇青石载体和Cu-SSZ-13/Cordierite催化剂的比表面积及孔体积
Table 2 Surface area and pore volume of the cordierite support and the Cu-SSZ-13/Cordierite catalysts
Sample Surface area
A/(m2·g-1)Pore volume
v/(cm3·g-1)Cordierite, raw <1 - Cordierite, acid treated 10 0. 041 Cu-SSZ-13 500 0. 324 Cu-SSZ-13(PVA) / Cordierite 79 0. 062 Cu-SSZ-13(SB) / Cordierite 76 0. 080 -
[1] BOSCH H, JANSSEN F. Formation and control of nitrogen oxides[J]. Catal Today, 1988, 2(4):369-379. http://www.sciencedirect.com/science/article/pii/0920586188800026 [2] HERNÁNDEZ-GIMÉNEZ A M, LOZANO-CASTELLÓ D, BUENO-LÓPEZ A. Effect of CO2, H2O and SO2 in the ceria-catalyzed combustion of soot under simulated diesel exhaust conditions[J]. Appl Catal B:Environ, 2014, 148-149:406-414. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=47ff4da17d95a70ba331013d86609d0b [3] 刘小文, 李俊, 李加兴, 倪钟孝, 易秀乐, 陆新.烟气脱硝技术综述[J].广州化工, 2019, 47(24):53-55. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gzhg201924020LIU Xiao-wen, LI Jun, LI Jia-xing, NI Zhong-xiao, YI Xiu-le, LU Xin. Overview of flue gas denitrification technology[J]. Guangzhou Chem Ind (China), 2019, 47(24):53-55. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gzhg201924020 [4] QI G, YANG R, RINALDI F. Selective catalytic reduction of nitric oxide with hydrogen over Pd-based catalysts[J]. J Catal, 2006, 237(2):381-392. http://www.sciencedirect.com/science/article/pii/S0021951705004550 [5] LI J, CHANG H, MA L, HAO J, YANG R T. Low-temperature selective catalytic reduction of NOx with NH3 over metal oxide and zeolite catalysts-A review[J]. Cataly Today, 2011, 175(1):147-156. http://www.sciencedirect.com/science/article/pii/S0920586111002434 [6] BRANDENBERGER S, KRÖCHER O, TISSLER A, ALTHOFF R. The state of the art in selective catalytic reduction of NOx by ammonia using metal-exchanged zeolite catalysts[J]. Catal Rev-Sci Eng, 2008, 50(4):492-531. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=277a26bc87ad6c96721f19cc69d98d90 [7] BONINGARI T, KOIRALA R, SMIRNIOTIS P G. Low-temperature selective catalytic reduction of NO with NH3 over V/ZrO2 prepared by flame-assisted spray pyrolysis:Structural and catalytic properties[J]. Appl Catal B:Environ, 2012, 127:255-264. http://www.sciencedirect.com/science/article/pii/S0926337312003566 [8] CHEN Z, YANG Q, LI H, LI X, WANG L, CHI TSANG S. Cr-MnOx mixed-oxide catalysts for selective catalytic reduction of NOx with NH3 at low temperature[J]. J Catal, 2010, 276(1):56-65. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=8109262 [9] SCHNEIDER H, MACIEJEWSKI M, KOHLER K, WOKAUN A, BAIKER A. Chromia supported on titania:Ⅳ. Properties of different chromium oxide phases in the catalytic reduction of no by NH3 studied by in situ diffuse reflectance FTIR spectroscopy[J]. J Catal, 1995, 157:312-320. http://www.sciencedirect.com/science/article/pii/S0021951785712961 [10] SMIRNIOTIS P G, PENÃ D A, UPHADE B S. Low-temperature selective catalytic reduction (SCR) of NO with NH3 by using Mn, Cr, and Cu oxides supported on hombikat TiO2[J]. Angew Chem Int Ed, 2001, 40(13):2479-2482. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=e6162682f1b94e539588446374a86834 [11] XIN Y, LI Q, ZHANG Z. Zeolitic materials for DeNOx selective catalytic reduction[J]. ChemCatChem, 2018, 10(1):29-41. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10.1002/cctc.201700854 [12] CHEN L, LI J, GE M. DRIFT study on cerium-tungsten titiania catalyst for selective catalytic reduction of NOx with NH3[J]. Environ Sci Technol, 2010, 44(24):9590-9596. doi: 10.1021/es102692b [13] LIU F, ASAKURA K, HE H, SHAN W, SHI X, ZHANG C. Influence of sulfation on iron titanate catalyst for the selective catalytic reduction of NOx with NH3[J]. Appl Catal B:Environ, 2011, 103(3/4):369-377. http://www.sciencedirect.com/science/article/pii/S0926337311000622 [14] ZHU Q, KONDO J N, OHNUMA R, KUBOTA Y, YAMAGUCHI M, TATSUMI T. The study of methanol-to-olefin over proton type aluminosilicate CHA zeolites[J]. Microporous Mesoporous Mater, 2008, 112(1/3):153-161. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cc7d73c543f52e0afd687143c7a618d7 [15] BLEKEN F, BJØRGEN M, PALUMBO L, BORDIGA S, SVELLE S, LILLERUD K-P, OLSBYE U. The effect of acid strength on the conversion of methanol to olefins over acidic microporous catalysts with the CHA topology[J]. Top Catal, 2009, 52(3):218-228. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=e8edb27e4d70101cb50692e2c2cbaeb6 [16] FICKEL D W, LOBO R F. Copper coordination in Cu-SSZ-13 and Cu-SSZ-16 investigated by variable-temperature XRD[J]. J Phys Chem C, 2010, 114(3):1633-1640. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=90670d2b7c29e13e5559d3c55fbb7e16 [17] DEKA U, JUHIN A, EILERTSEN E A, EMERICH H, GREEN M A, KORHONEN S T, WECKHUYSEN B M, BEALE A M. Confirmation of isolated Cu2+ ions in SSZ-13 zeolite as active sites in NH3-selective catalytic reduction[J]. J Phys Chem C, 2012, 116(7):4809-4818. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=591f7753708a4d216d8108acb395827c [18] FICKEL D W, D'ADDIO E, LAUTERBACH J A, LOBO R F. The ammonia selective catalytic reduction activity of copper-exchanged small-pore zeolites[J]. Appl Catal B:Environ, 2011, 102(3/4):441-448. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=0fb9994b0a77714b0529c2edbe512293 [19] BOGER T, HEIBEL A K, SORENSEN C M. Monolithic catalysts for the chemical industry[J]. Ind Eng Chem Res, 2004, 43(16):4602-4611. doi: 10.1002/chin.200439250 [20] LIU Q, LIU Z, WU W. Effect of V2O5 additive on simultaneous SO2 and NO removal from flue gas over a monolithic cordierite-based CuO/Al2O3 catalyst[J]. Catal Today, 2009, 147S:S285-S289. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f57363d3c198098483ab07e883079a20 [21] ZONES S I. Zeolite SSZ-13 and its method of preparation: US, 4544538[P]. 1985. [22] 范超, 罗莉, 吴志伟, 朱华青, 秦张峰.整体式Pd/ZSM-5/Cordierite催化剂的制备及其低浓度甲烷燃烧催化性能[J].陕西师范大学学报(自然科学版), 2019, 47(1):94-100. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=sxsfdxxb201901014FAN Chao, LUO Li, WU Zhi-wei, ZHU Hua-qing, QIN Zhang-feng. Preparation of monolithic Pd/ZSM-5/Cordierite catalyst for the combustion of lean methane[J]. J Shaanxi Normal Univ (Nat Sci Ed), 2019, 47(1):94-100. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=sxsfdxxb201901014 [23] HE D, WANG Z, DENG D, DENG S, HE H, LIU L. Synthesis of Cu-SSZ-13 catalyst by using different silica sources for NO-SCR by NH3[J]. Mol Catal, 2020, 484:110738. http://www.sciencedirect.com/science/article/pii/S2468823119306029 [24] WANG Y, XIE L, LIU F, RUAN W. Effect of preparation methods on the performance of CuFe-SSZ-13 catalysts for selective catalytic reduction of NOx with NH3[J]. J Environ Sci, 2019, 81:195-204. doi: 10.1016/j.jes.2019.01.013 [25] RICHTER M, FAIT M, ECKELT R, SCHNEIDER M, RADNIK J, HEIDEMANN D, FRICKE R. Gas-phase carbonylation of methanol to dimethyl carbonate on chloride-free Cu-precipitated zeolite Y at normal pressure[J]. J Catal, 2007, 245(1):11-24. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=4e01dd1282792c4a80ab722518b459a6 [26] GAO F, WALTER E D, KARP E M, LUO J, TONKYN R G, KWAK J H, SZANYI J, PEDEN C H F. Structure-activity relationships in NH3-SCR over Cu-SSZ-13 as probed by reaction kinetics and EPR studies[J]. J Catal, 2013, 300:20-29. doi: 10.1016/j.jcat.2012.12.020 [27] KEFIROV R, PENKOVA A, HADJⅡVANOV K, DZWIGAJ S, CHE M. Stabilization of Cu+ ions in BEA zeolite:Study by FTIR spectroscopy of adsorbed CO and TPR[J]. Microporous Mesoporous Mater, 2008, 116(1/3):180-187. http://www.sciencedirect.com/science/article/pii/S1387181108001765 [28] 刘世成, 秦瑞香, 肖瑞, 张春雪, 王单, 刘通, 王金波.铜基ZSM-5分子筛催化氮氧化物的NH3-SCR反应性能[J].重庆科技学院学报(自然科学版), 2019, 21(5):108-111. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cqsygdzkxxxb201905027LIU Shi-cheng, QIN Rui-xiang, XIAO Rui, ZHANG Chun-xue, WANG Dan, LIU Tong, WANG Jin-bo. NH3-SCR reaction of nitrogen oxides catalyzed by Cu-based ZSM-5 zeolite[J]. J Chongqing Univ Sci Technol(Nat Sci Ed), 2019, 21(5):108-111. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cqsygdzkxxxb201905027 [29] DOU B, LV G, WANG C, HAO Q, HUI K. Cerium doped copper/ZSM-5 catalysts used for the selective catalytic reduction of nitrogen oxide with ammonia[J]. Chem Eng J, 2015, 270:549-556. doi: 10.1016/j.cej.2015.02.004 [30] SULTANA A, SASAKI M, SUZUKI K, HAMADA H. Tuning the NOx conversion of Cu-Fe/ZSM-5 catalyst in NH3-SCR[J]. Catal Commun, 2013, 41:21-25. doi: 10.1016/j.catcom.2013.06.028 [31] SJÖVALL H, OLSSON L, FRIDELL E, BLINT R J. Selective catalytic reduction of NOx with NH3 over Cu-ZSM-5-The effect of changing the gas composition[J]. Appl Catal B:Environ, 2006, 64:180-188. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=dfc71c904d667ae8c88f000dc1b6c14b [32] WANG C, WANG C, WANG J, WANG J, SHEN M, LI W. Effects of Na+ on Cu/SAPO-34 for ammonia selective catalytic reduction[J]. J Environ Sci, 2018, 70:20-28. [33] XIE K, LEISTNER K, WIJAYANTI K, KUMAR A, KAMASAMUDRAM K, OLSSON L. Influence of phosphorus on Cu-SSZ-13 for selective catalytic reduction of NOx by ammonia[J]. Catal Today, 2017, 297:46-52. http://www.sciencedirect.com/science/article/pii/S0920586117304996 [34] HAMMERSHØI P S, JANGJOU Y, EPLING W S, JENSEN A D, JANSSENS T V W. Reversible and irreversible deactivation of Cu-CHA NH3-SCRcatalysts by SO2 and SO3[J]. Appl Catal B:Environ, 2018, 226:38-45. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c25108693c8e96c4d365404b3fdcdda1 [35] HAMMERSHØI P S, VENNESTRØM P N R, FALSIG H, JENSEN A D, JANSSENS T V W. Importance of the Cu oxidation state for the SO2-poisoning of a Cu-SAPO-34 catalyst in the NH3-SCR reaction[J]. Appl Catal B:Environ, 2018, 236:377-383. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=76dda9bf0ff01d8c0b89ea3c33ba46dc [36] JANGJOU Y, DO Q, GU Y, LIM L-G, SUN H, WANG D, KUMAR A, LI J, GRABOW L C, EPLING W S. Nature of Cu active centers in Cu-SSZ-13 and their responses to SO2 exposure[J]. ACS Catal, 2018, 8(2):1325-1337. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d8e0d423adb8ade658e9999615bcf17b [37] ZHANG L, WANG D, LIU Y, KAMASAMUDRAM K, LI J, EPLING W. SO2 poisoning impact on the NH3-SCR reaction over a commercial Cu-SAPO-34 SCR catalyst[J]. Appl Catal B:Environ, 2014, 156-157:371-377. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=4e74299b00dc25a2f8e041fc9c08dbf7 [38] JANGJOU Y, WANG D, KUMAR A, LI J, EPLING W S. SO2 poisoning of the NH3-SCR reaction over Cu-SAPO-34:Impact of ammonium sulfate versus other S-containing species[J]. ACS Catal, 2016, 6(10):6612-6622. http://smartsearch.nstl.gov.cn/paper_detail.html?id=ac8656054151718e654efb7b72c2d617 [39] FERRIZZ R M, GORTE R J, VOHS J M. TPD and XPS investigation of the interaction of SO2 with model ceria catalysts[J]. Catal Lett, 2002, 82:123-129. doi: 10.1023/A:1020512713021 [40] KONG L, ZOU S, MEI J, GENG Y, ZHAO H, YANG S. Outstanding resistance of H2S-modified Cu/TiO2 to SO2 for capturing gaseous Hg(0) from nonferrous metal smelting flue gas:Performance and reaction mechanism[J]. Environ Sci Technol, 2018, 52(17):10003-10010. http://smartsearch.nstl.gov.cn/paper_detail.html?id=c6e5758536eee1cfdb9e0a4e24468bc7 [41] THEGE I K. DSC investigation of the thermal behaviour of (NH4)2SO4, NH4HSO4 and NH4NH2SO3[J]. Thermochim Acta, 1983, 60(2):149-159. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10.1002/chin.198314019 -





 下载:
下载: