-
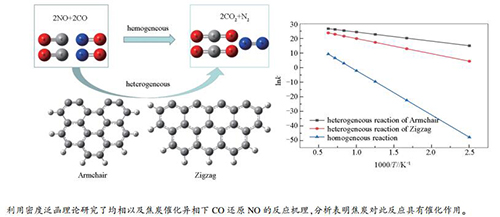
摘要: 利用密度泛函理论,研究了焦炭催化作用下CO还原NO的化学反应机理,优化得到了均相反应路径以及在Zigzag和Armchair型焦炭表面上的异相反应路径中所有驻点的几何构型与能量,并对三条反应路径进行了动力学分析。结果表明,均相NO还原反应的活化能为254.06 kJ/mol,而Zigzag型与Armchair型焦炭表面NO异相还原反应的活化能分别为86.94与52.16 kJ/mol,说明焦炭在NO还原反应中能够起到催化作用。在焦炭表面进行的CO还原NO的反应路径经历N2形成、N2释放及两步CO2释放四个阶段,最终生成一个N2分子与两个CO2分子。此外,通过对比不同路径下异相反应的能量变化与动力学参数可知,焦炭表面结构对NO还原反应特性存在较大影响;与Zigzag型焦炭表面相比,基于Armchair型焦炭表面的NO还原反应决速步能垒值更低且反应速率更快,表明在Armchair型焦炭表面上的NO还原反应更易进行。Abstract: The mechanism of NO reduction with CO catalyzed by char was studied via density function theory (DFT). The optimized configurations and stationary points of homogeneous and heterogeneous reactions on the char surfaces of Zigzag and Armchair were obtained. Finally, kinetic analysis for both homogeneous and heterogeneous reactions were carried out. The results show that the activation energy of homogeneous NO reduction reaction is 254.06 kJ/mol, while only 86.94 and 52.16 kJ/mol for heterogeneous reaction on Zigzag and Armchair models, respectively. This indicates that char is able to play an activating role in the NO reduction reaction. The NO reduction reaction on the char surface undergoes four stages of N2 formation, N2 release, and two stages of CO2 release, and finally generates one N2 molecule and two CO2 molecules. Moreover, the surface structure of char has a great influence on the characteristics of NO reduction reaction. Compared to Zigzag char surface, NO reduction reaction with Armchair-type has a lower energy barriers and higher reaction rate constants. That is, the NO reacts easily with CO on the Armchair surface.
-
Key words:
- NO /
- CO /
- heterogeneous reduction /
- char /
- density functional theory /
- reaction kinetics
-
表 1 反应动力学参数
Table 1 Reaction kinetic parameters
Reaction Pre-exponential factor A/s-1 Activation energy Ea/(kJ·mol-1) Arrhenius equation Homogeneous reaction 1.43×1012 254.06 k=1.43×1012e-30467.12/T Heterogeneous reaction of Zigzag 1.05×1013 86.94 k=1.05×1013e-10385.94/T Heterogeneous reaction of Armchair 1.48×1013 52.16 k=1.48×1013e-6253.50/T -
[1] 李文秀, 王宝凤, 任杰, 张锴, 杨凤玲, 程芳琴.贫煤O2/CO2气氛下燃烧时内在矿物质对SO2和NOx排放特性的影响[J].燃料化学学报, 2017, 45(10):1200-1208. http://www.ccspublishing.org.cn/article/id/e8eab9d5-d207-45f1-ac96-2f83146ef0e3LI Wen-xiu, WANG Bao-feng, REN Jie, ZHANG Kai, YANG Feng-ling, CHENG Fang-qin. Effect of intrinsic minerals on SO2 and NOx emission characteristics during combustion in lean coal O2/CO2 atmosphere[J]. J Fuel Chem Technol, 2017, 45(10):1200-1208. http://www.ccspublishing.org.cn/article/id/e8eab9d5-d207-45f1-ac96-2f83146ef0e3 [2] 信晶.煤焦-NO反应过程中氮转化机理与试验研究[D].北京: 华北电力大学, 2015.XIN Jing. Mechanism and experimental study of nitrogen conversion in coal char-NO reaction process[D]. Beijing: North China Electric Power University, 2015. [3] 谈冠希, 迟姚玲, 李双, 易玉峰, 靳广洲.锰锆复合氧化物CO催化还原NO性能研究[J].燃料化学学报, 2019, 47(10):1258-1264. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201910013TAN Guan-xi, CHI Yao-ling, LI Shuang, YI Yu-feng, JIN Guang-zhou. Study on the performance of Mn-Zr composite oxide for CO reduction of NO[J]. J Fuel Chem Technol, 2019, 47(10):1258-1264. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201910013 [4] 张秀霞.焦炭燃烧过程中氮转化机理与低NOx燃烧技术的开发[D].杭州: 浙江大学, 2012.ZHANG Xiu-xia. Nitrogen conversion mechanism during coke combustion and development of low NOx combustion technology[D]. Hangzhou: Zhejiang University, 2012. [5] MA Z, DENG J, LI Z, LI Q, ZHAO P, WANG L, SUN Y, ZHENG H, PAN L, ZHAO S, JIANG J, WANG S, DUAN L. Characteristics of NOx emission from Chinese coal-fired power plants equipped with new technologies[J]. Atmos Environ, 2016, 131:164-170. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=b9584c9aea73676eb02ab0100fe5f332 [6] BURCH T E, CHEN W, LESTER T W, STERLING A M. Interaction of fuel nitrogen with nitric oxide during reburning with coal[J]. Combust Flame, 1994, 98(4):391-401. http://cn.bing.com/academic/profile?id=6c8e7215951c5cc49017dfd1c18b4ba0&encoded=0&v=paper_preview&mkt=zh-cn [7] JOHNSSON J E. Formation and reduction of nitrogen oxides in fluidized-bed combustion[J]. Fuel, 1994, 73(9):1398-1415. doi: 10.1016-0016-2361(94)90055-8/ [8] ZHANG H, JIANG X, LIU J, SHEN J. Application of density functional theory to the nitric oxide heterogeneous reduction mechanism in the presence of hydroxyl and carbonyl groups[J]. Energy Convers Manage, 2014, 83:167-176. http://cn.bing.com/academic/profile?id=83e4361b66c8d6af7b1f2af4260dd8ea&encoded=0&v=paper_preview&mkt=zh-cn [9] FENG B, LIU H, YUAN J, LIN Z, LIU D, LECKNER B. Mechanisms of N2O formation from char combustion[J]. Energy Fuels, 1996, 10(1):203-208. doi: 10.1021/ef9500898 [10] 王春波, 岳爽, 许旭斌, 李一鹏. O2/CO2气氛下煤焦恒温燃烧NOx释放特性[J].煤炭学报, 2018, 43(1):257-264. http://www.cnki.com.cn/Article/CJFDTotal-MTXB201801032.htmWANG Chun-bo, YUE Shuang, XU Xu-bin, LI Yi-peng. NOx release characteristics of coal coke in constant temperature combustion under O2/CO2 atmosphere[J]. J China Coal Soc, 2018, 43(1):257-264. http://www.cnki.com.cn/Article/CJFDTotal-MTXB201801032.htm [11] 王贲, 孙路石, 苏胜, 向军, 胡松, 费华. O2/CO2气氛中低氧浓度下煤粉和煤焦的NO生成规律[J].煤炭学报, 2012, 37(3):501-505. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=mtxb201203027WANG Ben, SUN Lu-shi, SU Sheng, XIANG Jun, HU Song, FEI Hua. NO formation law of pulverized coal and coal char under low oxygen concentration in O2/CO2 atmosphere[J]. J China Coal Soc, 2012, 37(3):501-505. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=mtxb201203027 [12] LÓPEZ D, CALO J. The NO-carbon reaction:The influence of potassium and CO on reactivity and populations of oxygen surface complexes[J]. Energy Fuels, 2007, 21(4):1872-1877. http://cn.bing.com/academic/profile?id=9cfb487e141ae89e83355668b85d69cb&encoded=0&v=paper_preview&mkt=zh-cn [13] 梁磊, 孙章, 魏侦凯, 郭瑞.冶金焦炭结构及组成的研究进展[J].燃料与化工, 2019, 50(2):18-22. http://d.old.wanfangdata.com.cn/Periodical/rlyhg201902005LIANG Lei, SUN Zhang, WEI Zhen-kai, GUO Rui. Progress in research of structure and composition of metallurgical coke[J]. Fuel Chem Processes, 2019, 50(2):18-22. http://d.old.wanfangdata.com.cn/Periodical/rlyhg201902005 [14] 钟俊, 高正阳, 丁艺, 余岳溪, 杨维结. Zigzag煤焦表面异相还原N2O反应[J].煤炭学报, 2017, 42(11):3028-3034. doi: 10.13225/j.cnki.jccs.2017.0228ZHONG Jun, GAO Zheng-yang, DING Yi, YU Yue-xi, YANG Wei-jie. Heterogeneous reduction of N2O reaction on Zigzag coal char surface[J]. J China Coal Soc, 2017, 42(11):3028-3034. doi: 10.13225/j.cnki.jccs.2017.0228 [15] 张秀霞, 周志军, 周俊虎, 姜树栋, 刘建忠, 岑可法. N2O在焦炭表面异相生成和分解机理的密度泛函理论研究[J].燃料化学学报, 2011, 39(11):806-811. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201111002ZHANG Xiu-xia, ZHOU Zhi-jun, ZHOU Jun-hu, JIANG Shu-dong, LIU Jian-zhong, CEN Ke-fa. Density functional theory study on the mechanism of heterogeneous phase formation and decomposition of N2O on coke surface[J]. J Fuel Chem Technol, 2011, 39(11):806-811. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201111002 [16] 余岳溪, 高正阳, 季鹏, 李方勇, 杨维结.煤焦异相还原N2O的反应机理[J].化工学报, 2017, 68(1):369-374. http://d.old.wanfangdata.com.cn/Periodical/hgxb201701044YU Yue-xi, GAO Zheng-yang, JI Peng, LI Fang-yong, YANG Wei-jie. Reaction mechanism of heterogeneous reduction of N2O from coal coke[J]. CIESC J, 2017, 68(1):369-374. http://d.old.wanfangdata.com.cn/Periodical/hgxb201701044 [17] SENDT K, HAYNES B S. Density functional study of the reaction of O2 with a single site on the zigzag edge of graphene[J]. Proc Combust Inst, 2011, 33(2):1851-1858. doi: 10.1016/j.proci.2010.06.021 [18] CHEN G, CHEN P, GU M, LIU F, LIN Y. DFT study on the reaction mechanism of N2O reduction with CO catalyzed by char[J]. Fuel, 2019, 254:115666. doi: 10.1016/j.fuel.2019.115666 [19] GAO Z, YANG W, DING X, DING Y, YAN W. Theoretical research on heterogeneous reduction of N2O by char[J]. Appl Therm Eng, 2017, 126:28-36. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=39883cac9b1d097d475518a0bf79d159 [20] JIAO A, ZHANG H, LIU J, JIANG X. Quantum chemical and kinetics calculations for the NO reduction with char(N):Influence of the carbon moNOx ide[J]. Combust Flame, 2018, 196:377-385. doi: 10.1016/j.combustflame.2018.06.029 [21] 张秀霞, 周志军, 周俊虎, 姜树栋, 刘建忠, 岑可法.含氮焦炭异相还原NO反应机理的密度泛函理论研究[J].化工学报, 2011, 62(4):1048-1054. http://d.old.wanfangdata.com.cn/Periodical/hgxb201104024ZHANG Xiu-xia, ZHOU Zhi-jun, ZHOU Jun-hu, JIANG Shu-dong, LIU Jian-zhong, CEN Ke-fa. A density functional theory study on the heterogeneous reduction mechanism of nitrogen-containing coke by NO[J]. CIESC J, 2011, 62(4):1048-1054. http://d.old.wanfangdata.com.cn/Periodical/hgxb201104024 [22] PERRY S T, HAMBLY E M, FLETCHER T H, SOLUM M S, PUGMIRE R J. Solid-state 13C NMR characterization of matched tars and chars from rapid coal devolatilization[J]. Proc Combust Inst, 2000, 28(2):2313-2319. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=CC028008586 [23] NAKADA K, FUJITA M, DRESSELHAUS G, DRESSELHAUS M S. Edge state in graphene ribbons:Nanometer size effect and edge shape dependence[J]. Phys Rev B, 1996, 54(24):17954-17961. http://cn.bing.com/academic/profile?id=62a95b9eb5c2a79f98ffcc5987e4a411&encoded=0&v=paper_preview&mkt=zh-cn [24] ENOKI T, FUJII S, TAKAI K. Zigzag and armchair edges in graphene[J]. Carbon, 2012, 50(9):3141-3145. http://d.old.wanfangdata.com.cn/OAPaper/oai_arXiv.org_1008.2496 [25] GIRIT C O, MEYER J C, ERNI R, ROSSELL M D, KISIELOWSKI C, YANG L, PARK C H, CROMMIE M F, COHEN M L, LOUIE S G, ZETTL A. Graphene at the Edge:Stability and dynamics[J]. Science, 2009, 323(5922):1705-1708 http://d.old.wanfangdata.com.cn/OAPaper/oai_arXiv.org_1104.3371 [26] ESPINAL J F, MONDRAGÓN F, TRUONG T N. Mechanisms for methane and ethane formation in the reaction of hydrogen with carbonaceous materials[J]. Carbon, 2005, 43(9):1820-1827. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6407267133d788e72454fbc5fa064580 [27] 高正阳, 杨维结, 阎维平.煤焦催化HCN还原NO的反应机理[J].燃料化学学报, 2017, 45(9):1043-1048. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201709003GAO Zheng-yang, YANG Wei-jie, YAN Wei-ping. Reaction mechanism of coal coke catalyzing hcn reduction[J]. J Fuel Chem Technol, 2017, 45(9):1043-1048. http://d.old.wanfangdata.com.cn/Periodical/rlhxxb201709003 [28] J F M, W T G, B S H, E S G, A R M. Gaussian 16, Revision D.01[CP]. Gaussian, Inc., Wallingford, CT, 2009. [29] ZHANG H, LIU J, SHEN J, JIANG X. Thermodynamic and kinetic evaluation of the reaction between NO (nitric oxide) and char(N) (char bound nitrogen) in coal combustion[J]. Energy, 2015, 82:312-321. [30] KYOTANI T, TOMITA A. Analysis of the reaction of carbon with NO/N2O using Ab initio molecular orbital theory[J]. J Phys Chem B, 1999, 103(17):3434-3441. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=514b7debbf6b0292216d45f3a3d5f185 -





 下载:
下载: