Physicochemical characteristics and TGA of distillation residues from bio-oil
-
摘要: 利用傅里叶变换红外光谱仪、激光共焦显微拉曼光谱仪和TGA Q500热分析仪对生物油蒸馏残渣及其在不同温度处理后的热解焦炭理化性质进行表征,并对其热失重特性进行分析。结果表明,生物油蒸馏残渣主要是由脂肪族、芳香族和低聚糖类等有机化合物组成;在氮气氛围下热解主要分为三个阶段:30-145℃为小分子物质挥发析出阶段145-550℃为大分子物质裂解和氧化阶段,550-750℃为焦炭产生阶段;热处理过程中各类物质逐步有序热解析出,同时固体产物石墨化程度随着热处理终止温度的升高而升高。Abstract: The distillation residues from bio-oil and the pyrolysis char obtained at different temperatures were investigated with FT-IR, Raman spectra and TGA. The results show that the distillation residues from bio-oil consist of aliphatic, aromatic and oligosaccharides. The process of thermal weight loss of distillation residues from bio-oil in nitrogen can be separated into three stages. The first is the volatilization of low-boiling point compounds from ambient temperature to 145℃. The second is the pyrolysis, oxidation and polymerization of the heavy compounds at higher temperature and the last is the formation and combustion of coke. The graphitic carbon fraction increases with increasing the carbonization temperature while the disordered carbon amount decreases.
-
Key words:
- bio-oil /
- distillation /
- thermal weight loss
-
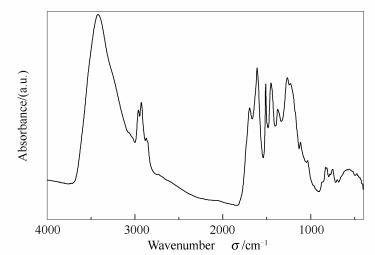
表 1 生物油蒸馏残渣的工业分析和元素分析
Table 1 Proximate and ultimate analyses of distillation residue from bio-oil
Sample Proximate analysis wad /% Ultimate analysis wad /% QHHV
/(MJ·kg-1)M A V FC C H O N BP025 1.00 0.50 72.90 25.60 74.00 6.21 18.25 1.45 29.12 表 2 生物油蒸馏残渣红外光谱定量分析
Table 2 Quantitative analysis on FT-IR spectra of distillation residues from bio-oil
Sample Content w/% C-O CH3/ CH2 C=C C-H -OH BP025 13.32 3.50 5.63 9.85 17.05 BP100 13.36 2.52 5.22 8.39 14.76 BP200 15.53 3.60 6.26 9.73 8.81 BP300 17.70 4.50 5.65 9.45 8.74 BP400 23.00 4.20 6.70 2.89 9.97 -
[1] 陈尔旺, 陈明强, 王君, 刘少敏, 王华.生物油分离技术的研究进展[J].广州化工, 2011, 39(3):3-5, 45. http://www.cnki.com.cn/Article/CJFDTOTAL-GZHA201103004.htmCHEN Er-wang, CHEN Ming-qiang, WANG Jun, LIU Shao-min, WANG Hua. Research progress of bio-oil separation[J]. Guangzhou Chem Ind, 2011, 39(3):3-5, 45. http://www.cnki.com.cn/Article/CJFDTOTAL-GZHA201103004.htm [2] 孙培勤, 臧哲学, 孙绍晖, 陈俊武.生物油的分离与分析研究进展[J].可再生能源, 2008, 26(5):35-40. http://www.cnki.com.cn/Article/CJFDTOTAL-NCNY200805014.htmSUN Pei-qin, ZANG Zhe-xue, SUN Shao-hui, CHEN Jun-wu. Seperation and analysis status on bio-oil[J]. Renewable Energy Resour, 2008, 26(5):35-40. http://www.cnki.com.cn/Article/CJFDTOTAL-NCNY200805014.htm [3] 李允超, 王贤华, 杨海平, 张世红, 陈汉平.生物油分离精制技术的研究进展[J].生物质化学工程, 2010, 44(6):46-51. http://www.cnki.com.cn/Article/CJFDTOTAL-LCHG201006015.htmLI Yun-chao, WANG Xian-hua, YANG Hai-ping, ZHANG Shi-hong, CHEN Han-ping. Progress on the separation and purification of bio-oil[J]. Biomass Chem Eng, 2010, 44(6):46-51. http://www.cnki.com.cn/Article/CJFDTOTAL-LCHG201006015.htm [4] 陶书伟, 杨松, 宋宝安, 王瑞, 周友春, 王俊.生物质热解液化制备生物基化学品[J].贵州大学学报, 2008, 25(3):299-303. http://www.cnki.com.cn/Article/CJFDTOTAL-GZDI200803021.htmTAO Shu-wei, YANG Song, SONG Bao-an, WANG Rui, ZHOU You-chun, WANG Jun. Preparation of chemicals with the technology of biomass[J]. J Guizhou Univ, 2008, 25(3):299-303. http://www.cnki.com.cn/Article/CJFDTOTAL-GZDI200803021.htm [5] 朱锡锋, 朱昌朋.生物质热解液化与美拉德反应[J].燃料化学学报, 2013, 41(8):911-916. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18225.shtmlZHU Xi-feng, ZHU Chang-peng. Biomass fast pyrolysis and Maillard reaction[J]. J Fuel Chem Technol, 2013, 41(8):911-916. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18225.shtml [6] LU Q, YANG X L, ZHU X F. Analysis on chemical and physical properties of bio-oil pyrolyzed from rice husk[J]. J Anal Appl Pyrolysis, 2008, 82(2):191-198. doi: 10.1016/j.jaap.2008.03.003 [7] CZERNIK S, BRIDGWATER A V. Overview of applications of biomass fast pyrolysis oils[J]. Energy Fuels, 2004, 18(2):590-598. doi: 10.1021/ef034067u [8] 朱满洲, 朱锡锋, 郭庆祥, 朱清时.以玉米秆为原料的生物质热解油的特性分析[J].中国科学技术大学学报, 2006, 36(4):374-377. http://www.cnki.com.cn/Article/CJFDTOTAL-ZKJD200604005.htmZHU Man-zhou. ZHU Xi-feng, GUO Qing-xiang, ZHU Qing-shi. Analyses and properties of pryolytic bio-oil from cornstalk[J]. J Univ Sci Technol China, 2006, 36(4):374-377. http://www.cnki.com.cn/Article/CJFDTOTAL-ZKJD200604005.htm [9] QIAN Y J, ZUO C J, HE J H.Structural analysis of bio-oils from sub-and supercritical water liquefaction of woody biomass[J]. Energy, 2007, 32:196-202. doi: 10.1016/j.energy.2006.03.027 [10] 宋春财.农作物秸秆的热解及在水中的液化研究[D].大连:大连理工大学, 2003.SONG Chun-cai. Pyrolysis of agricultural stalks and liquefaction in water[D].Dalian:Dalian University of Technology, 2003. [11] 戎欣, 黄清发, 谢丹, 许庆利, 王复, 李洪宇.生物质裂解油催化裂解精制机理研究(Ⅱ)-生物质油沥青质的分子模型[J].太阳能学报, 2012, 33(2):243-248. http://www.cnki.com.cn/Article/CJFDTOTAL-TYLX201202011.htmRONG Xin, HUANG Qing-fa, XIE Dan, XU Qing-li, WANG Fu, LI Hong-yu. Study on catalytic pyrolysis mechanism of bio-oil (Ⅱ)-molecural structure model of asphaltene in bio-oil[J]. Acta Energ Sol Sin, 2012, 33(2):243-248. http://www.cnki.com.cn/Article/CJFDTOTAL-TYLX201202011.htm [12] 翁诗甫.傅里叶变换红外光谱分析[M]. 2版.北京:化学工业出版社, 2012.WENG Shi-fu. Fourier Transform Infrared Spectrum Analysis[M]. 2nd ed. Beijing:Chemical Industry Press, 2012. [13] 冯冬冬.生物质热解焦炭理化特性的研究[D].哈尔滨:哈尔滨工业大学, 2014.FENG Dong-dong. Research on physical and chemical characteristics of biomass pyrolysis char[D]. Harbin:Harbin Institute of Technology, 2014. [14] 冯杰, 李文英, 谢克昌.傅立叶红外光谱法对煤结构的研究[J].中国矿业大学学报, 2002, 31(5):362-366. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGKD200205005.htmFENG Jie, LI Wen-ying, XIE Ke-chang. Research on coal structure using FT-IR[J]. J China Univ Min Technol, 2002, 31(5):362-366. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGKD200205005.htm [15] 丰晓, 叶奋, 黄彭.基于沥青老化的红外光谱羰基吸光度分析[J].建筑材料学报, 2008, 11(3):375-378. http://www.cnki.com.cn/Article/CJFDTOTAL-JZCX200803026.htmFENG Xiao, YE Fen, HUANG Peng. Infrared spectrum analysis of carbonyl absorbance on asphalt aging[J]. J Build Mater, 2008, 11(3):375-378. http://www.cnki.com.cn/Article/CJFDTOTAL-JZCX200803026.htm [16] 游婷婷, 马建锋, 郭思勤, 许凤.傅里叶变换拉曼光谱对芦竹碱木质素的研究[J].光谱学与光谱分析, 2014, (8):2112-2116. http://www.cnki.com.cn/Article/CJFDTOTAL-GUAN201408023.htmYOU Ting-ting, MA Jian-feng, GUO Si-Qin, XU Feng. Study of alkaline ligin from arundo donax linn Based on FT-Raman spectroscopy[J]. Spetrosc Spect Anal, 2014, (8):2112-2116. http://www.cnki.com.cn/Article/CJFDTOTAL-GUAN201408023.htm [17] WANG M R, LAI Y Q, FANG J, LI J. N-doped porous carbon derived from biomass as an advanced electro catalyst for aqueous aluminum/air battery[J]. Int J Hydrogen Energy, 2015, 40(46):16230-16237. doi: 10.1016/j.ijhydene.2015.09.054 [18] MODAK A, BHAUMIK A. Porous carbon derived via KOH activation of a hypercrosslinked porous organic polymer for efficient CO2, CH4, H2 adsorptions and high CO2/N2 selectivity[J]. J Solid State chem, 2015, 232:157-162. doi: 10.1016/j.jssc.2015.09.022 [19] SHAHTALEBI A, MAR M, GUERIN K, BHATIA S K. Effect of fluorine doping on structure and CO2 adsorption in silicon carbide-derived carbon[J]. Carbon, 2016, 96:565-577. doi: 10.1016/j.carbon.2015.09.105 [20] ZHAO Y, FANG F, XIAO H M, Preparation of pore-size controllable activated carbon fibers from bamboo fibers with superior performance for xenon storage[J]. Chem Eng J, 2015, 270:528-534. doi: 10.1016/j.cej.2015.02.054 -





 下载:
下载: