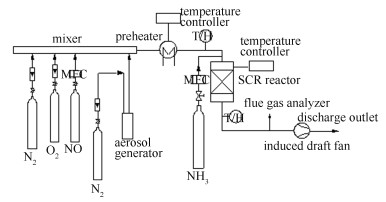
Mechanism of CaSO4 poisoning commercial V2O5-WO3/TiO2 catalyst for flue gas selective catalytic reduction of NOx with NH3
-
摘要: 基于商业V2O5-WO3/TiO2脱硝催化剂, 设计了两种模拟CaSO4中毒的方法, 通过比表面积测定(BET)、X射线衍射(XRD)、程序升温还原(H2-TPR)、扫描电子显微镜(SEM)、原位漫反射傅里叶变换红外光谱(in situ DRIFTS) 等表征技术并结合固定床脱硝性能测试平台, 对中毒前后催化剂的微观结构、氧化还原能力及表面性质的变化与脱硝活性进行了对比研究, 探索硫酸钙中毒机理。研究表明, CaSO4会堵塞催化剂孔径, 孔径小于2.7 nm和孔径大于17.8 nm时CaSO4的影响更大, 从而使催化剂的比表面积和孔体积变小; CaSO4中毒会导致Br∅nsted酸位数量和强度的降低, 同时Lewis酸强度也会减弱, 从而阻碍了NH3的吸附, CaSO4引起催化剂氧化还原能力的降低。
-
关键词:
- 机理研究 /
- CaSO4中毒 /
- V2O5-WO3/TiO2 /
- 选择性催化还原(SCR)
Abstract: Based on commercial V2O5-WO3/TiO2 catalyst, two methods to simulate CaSO4 poisoning were designed, and the physico-chemical properties of fresh and poisoned catalysts were investigated by BET specific surface area measurement, X-ray diffraction (XRD), H2 temperature-programmed reduction (H2-TPR), scanning electron microscope (SEM) and in-situ diffuse reflectance infrared spectrometry (in situ DRIFTS). Meanwhile, the catalytic performance for selective catalytic reduction of NO with NH3(NH3-SCR) in a fixed bed was also explored comparatively. SEM results show that CaSO4 plugs the small hole (pore width smaller than 2.7 nm) and big hole (pore width bigger than 17.8 nm), causing the loss of surface area and pore volume. CaSO4 could weaken the intensity of both Br∅nsted acid Sites and Lewis acid sites, particularly the active centers of Br∅nsted acid sites, which hinders the absorption of NH3 and reduces the redox abilities.-
Key words:
- mechanism /
- CaSO4 poisoning /
- V2O5-WO3/TiO2 /
- selective catalytic reduction (SCR)
-
表 1 催化剂的比表面积、孔径和孔容
Table 1 BET surface area, pore diameter and pore volume of samples
Sample BET surface area A/(m2·g-1) Pore volume v/(cm3·g-1) Average pore diamater d/nm V2O5-WO3/TiO2 60.863 0.327 24.749 V2O5-WO3/TiO2(sample 1) 51.921 0.287 20.925 V2O5-WO3/TiO2(sample 2) 39.338 0.196 18.994 V2O5-WO3(0.1CaSO4)/TiO2 57.092 0.308 21.632 V2O5-WO3(0.2CaSO4)/TiO2 52.686 0.259 23.392 V2O5-WO3(0.3CaSO4)/TiO2 49.113 0.237 19.364 V2O5-WO3(0.4CaSO4)/TiO2 42.951 0.228 21.298 -
[1] SHI Y, XIA Y F, LU B H, LIU N, ZHANG L, LI S J, LI W. Emission inventory and trends of NOx for China, 2000-2020[J]. J Zhejiang Univ Sci A, 2014, 15(6): 454-464. doi: 10.1631/jzus.A1300379 [2] 龚立贤, 王韬明.火电厂大气污染物排放标准GB13223-2011修订情况及除尘、除二氧化硫、除氮氧化物设备的选择[C]//大机组供热改造与优化运行技术2012年会论文集.无锡:中国电机工程学会, 2012: 27-30.GONG Li-xian, WANG Tao-ming. The air pollutants emission standards of the coal-fired power plant GB13223-2011 revision and the selection of dust, sulfur dioxide and nitrogen oxides removal equipment[C]//Renovation and Optimal Operation of Heating Technology in the Large Sets of Proceedings 2012 Annual Meeting Memoir. Wuxi: CSEE, 2012: 27-30. [3] 桑绮, 乐园园, 徐晗.火电厂大气污染物排放标准、现状及减排技术[J].浙江电力, 2011, 30(12): 42-46. http://www.cnki.com.cn/Article/CJFDTOTAL-ZJDL201112013.htmSANG Qi, LE Yuan-yuan, XU Han. Emission standards、current situation and reduction technology of air pollutants for thermal power plants[J]. Zhejiang Elect Power, 2011, 30(12): 42-46. http://www.cnki.com.cn/Article/CJFDTOTAL-ZJDL201112013.htm [4] 蔡小峰, 李晓芸. SNCR-SCR烟气脱硝技术及其应用[J].电力环境保护, 2008, 24(3): 26-29. http://www.cnki.com.cn/Article/CJFDTOTAL-DLHB200803008.htmCAI Xiao-feng, LI Xiao-yun. Hybrid SNCR-SCR denitrification technique and its application[J]. Elect Power Environ Prot, 2008, 24(3): 26-29. http://www.cnki.com.cn/Article/CJFDTOTAL-DLHB200803008.htm [5] 仲兆平, 张茜芸, 杨碧源, 姚杰. V2O5/TiO2烟气脱硝催化剂的钾、钠、锌、磷中毒及再生[J].东南大学学报(自然科学版), 2013, 43(3): 548-552.ZHONG Zhao-ping, ZHANG Xi-yun, YANG Bi-yuan, YAO Jie. Effects of potassium, sodium, zinc and phosphorus on flue gas de-NOx performance over V2O5/TiO2 catalysts[J]. J Southeast Univ (Nat Sci Ed), 2013, 43(3): 548-552. [6] 云端, 邓斯理, 宋蔷, 姚强. V2O5-WO3/TiO2系SCR催化剂的钾中毒及再生方法[J].环境科学研究, 2009, 22(6): 730-735.YUN Duan, DENG Si-li, SONG Qiang, YAO Qiang. Potassium deactivation and regeneration method of V2O5-WO3/TiO2 SCR catalyst [J]. Res Environ Sci, 2009, 22(6): 730-735. [7] 沈伯雄, 熊丽仙, 刘亭, 王静, 田晓娟.纳米负载型V2O5-WO3/TiO2催化剂碱中毒及再生研究[J].燃料化学学报, 2010, 38(1): 85-90. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17543.shtmlSHEN Bo-xiong, XIONG Li-xian, LIU Ting, WANG Jing, TIAN Xiao-juan. Alkali deactivation and regeneration of nano V2O5-WO3/TiO2 catalysts[J]. J Fuel Chem Technol, 2010, 38(1): 85-90. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17543.shtml [8] BENSON S A, LAUMB J D, CROCKER C R, PAVLISH J H. SCR catalyst performance in flue gases derived from subbituminous and lignite coals[J]. Fuel Process Technol, 2005, 86(5): 577-613. doi: 10.1016/j.fuproc.2004.07.004 [9] NICOSIA D, CZEKAJ I, KROCHER O. Chemical deactivation of V2O5-WO3/TiO2 SCR catalysts by additives and impurities from fuels, lubrication oils and urea solution: Part Ⅱ. Characterization study of the effect of alkali and alkaline earth metals[J]. Appl Catal B: Environ, 2008, 77(3): 228-236. [10] 沈伯雄, 邓黎丹, 马娟, 左琛, 郝晓翠. Mn-CeOx/钛基层柱粘土(Ti-PILCs) 低温选择性催化还原催化剂的钙中毒研究[J].环境污染与防治, 2011, 33(3): 1-5, 10.SHEN Bo-xiong, DENG Li-dan, MA Juan, ZUO Chen, HAO Xiao-cui. Calcium deactivation of Mn-CeOx/Ti-PILCs low-temperature SCR catalyst[J]. Environ Pollut Control, 2011, 33(3): 1-5, 10. [11] 商雪松, 陈进生, 姚源, 胡恭任.商业SCR烟气脱硝催化剂钙中毒研究[J].环境工程学报, 2013, 7(2): 624-630. http://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201302040.htmSHANG Xue-song, CHEN Jin-sheng, YAO Yuan, HU Gong-ren. Study on calcium poisoning of commercial SCR de-NOx catalyst[J]. Chin J Environ Eng, 2013, 7(2): 624-630. http://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201302040.htm [12] 张秋林. CaO对SCR催化剂选择的影响[J].能源研究与利用, 2006, 6: 008. http://www.cnki.com.cn/Article/CJFDTOTAL-NYYJ200606008.htmZHANG Qiu-Lin. Influence of CaO to the selection of SCR catalyst[J]. Energy Res Util, 2006, 6: 008. http://www.cnki.com.cn/Article/CJFDTOTAL-NYYJ200606008.htm [13] 高凤雨, 唐晓龙, 易红宏, 赵顺征, 李东, 马玎, 张佟佟.商用SCR催化剂的钠中毒及再生[J].中南大学学报(自然科学版), 2015, 46(6): 2382-2390. http://www.cnki.com.cn/Article/CJFDTOTAL-ZNGD201506052.htmGAO Feng-yu, TANG Xiao-long, YI Hong-hong, ZHAO Shun-zheng, LI Dong, MA Ding, MA Tong-tong. Studies on calcium poisoning mechanism and regeneration of commercial DeNOx SCR catalysts[J]. J Cent South Univ (Nat Sci Ed), 2015, 46(6): 2382-2390. http://www.cnki.com.cn/Article/CJFDTOTAL-ZNGD201506052.htm [14] MARIANA A, RAMOS L, LUIS C C. Effect of sulfates and reduced-vanadium species on oxidative desulfurization (ODS) with V2O5/TiO2 catalyst[J]. Ind Eng Chem Res, 2011, 50(5): 2641-2649. doi: 10.1021/ie1006728 [15] 陈婷, 管斌, 林赫, 朱霖.原位漫反射傅里叶变换红外光谱研究锰铁基催化剂上低温选择性催化还原反应机理[J].催化学报, 2014, 35(3): 294-301. doi: 10.1016/S1872-2067(12)60730-XCHEN Ting, GUAN Bin, LIN He, ZHU Lin. In situ DRIFTS study of the mechanism of low temperature selective catalytic reduction over manganese-iron oxides[J]. Chin J Catal, 2014, 35(3): 294-301. doi: 10.1016/S1872-2067(12)60730-X [16] BONINGARI T, PANAGIOTIS G S. Nickel-doped Mn/TiO2 as an efficient catalyst for the low-temperature SCR of NO with NH3: Catalytic evaluation and characterizations[J]. J Catal, 2012, 288: 74-83. doi: 10.1016/j.jcat.2012.01.003 [17] SI Z C, WENG D, WU X D, LI J, LI G. Structure, acidity and activity of CuOx/WOx-ZrO2 catalyst for selective catalytic reduction of NO by NH3[J]. J Catal, 2010, 271(1): 43-51. doi: 10.1016/j.jcat.2010.01.025 [18] 李倩, 谷华春, 李萍, 周钰浩, 刘莹, 齐中囡, 辛颖, 张昭良.非晶Ce-Ti氧化物用于NH3选择性催化还原NO的原位红外研究[J].催化学报, 2014, 35(8): 1289-1298. doi: 10.1016/S1872-2067(14)60154-6LI Qian, GU Hua-chun, LI Ping, ZHOU Yu-hao, LIU Ying, QI Zhong-nan, XIN Ying, ZHANG Zhao-liang, In situ IR studies of selective catalytic reduction of NO with NH3 on Ce-Ti amorphous oxides[J]. Chin J Catal, 2014, 35(8): 1289-1298. doi: 10.1016/S1872-2067(14)60154-6 [19] TSYGANENKO A A, POZDNYAKOV D V, FILIMONV V N. Infrared study of surface species arising from ammonia adsorption on oxides surfaces[J]. J Mol Struct, 1975, 29(2): 299-318. doi: 10.1016/0022-2860(75)85038-1 [20] NICOSIA D, CZEKAJ I, KMCHER O. Chemical deactivation of V2O5-WO3/TiO2 SCR catalysts by additives and impurities from fuels, 1ubrication oils and urea solution, part II. Characterization study of the effect of alkali and alkaline earth metals[J]. Appl Catal B: Environ, 2008, 77(3/4): 228-236. [21] 辛勤, 罗孟飞.现代催化研究方法[M].北京:科学出版社, 2009: 83.XIN Qin, LUO Meng-fei. Modern Catalytic Research Methods[M]. Beijing: Science Press, 2009: 83. -





 下载:
下载: