Simulation of hydrogen bonds in low-rank coals with lignite-related complexes using dispersion corrected density functional theory
-
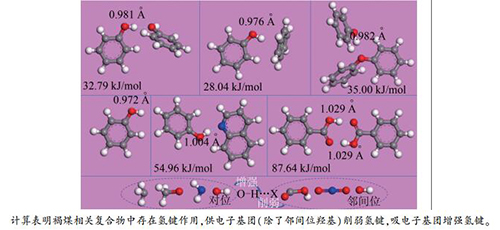
摘要: 本研究以苯酚…苯酚、苯酚…苯、苯酚…二苯醚、苯酚…喹啉和苯甲酸…苯甲酸为对象,采用色散校正的密度泛函理论分别研究褐煤中自缔合OH、OH-π、OH-醚O、OH-N和COOH-COOH之间形成的氢键。此外,还研究了氢键供体中取代基(CH3-、CH3O-、OH-、NH2-、COOH-和NO2-)对氢键的影响。对上述复合物进行了几何优化,并计算了能量、Mulliken电荷分布及振动频率。从优化的结构中可以看出上述复合物之间都存在氢键,所有复合物中O-H键键长都比苯酚中自由羟基的长,这表明这些复合物之间存在相互作用。其中,羧酸…羧酸复合物中O-H键的键长最长。此外,通过Mulliken电荷分布可看出上述复合物之间存在电荷转移。基于振动频率分析,所有的O-H键伸缩振动都发生了红移,尤其是羧酸…羧酸和苯酚…喹啉复合物,这可为煤中羟基振动的红外光谱分析提供依据。根据键能不同氢键强度按以下顺序依次递减:COOH-COOH>OH-N >自缔合OH≈OH-醚O > OH-π,这与振动频率的分析结果一致。此外,不同取代基对氢键作用的影响不同。Abstract: Phenol…phenol, phenol…benzene, phenol…oxydibenzene, phenol…quinoline, and benzoic acid…benzoic acid were selected as lignite-related complexes to investigate different hydrogen bonds formed by self-associated OH, OH-π, OH-ether O, OH-N, and COOH-COOH using density functional theory with dispersion correction, respectively. Moreover, the effects of substituents (CH3-, CH3O-, OH-, NH2-, COOH-, and NO2-) in donors on the hydrogen bonds were investigated. Geometry optimization, energy, Mulliken population, and frequency of all the complexes were calculated. It can be seen from optimized structures that there indeed are hydrogen bonds in the different complexes. Bond lengths of all O-H bonds in the different complexes become longer than that of free OH in phenol, which implies that intermolecular interactions exist in all the complexes. Among of them, bond lengths of O-H bonds in benzoic acid…benzoic acid are the longest. In addition, charge transfer can be observed via Mulliken population. Based on frequency analysis, all O-H stretching vibrations have obvious red shift, especially O-H bonds in benzoic acid…benzoic acid and phenol…quinoline, which gives evidence of using the infrared spectroscopy to analyze hydroxyl groups of coals. According to bond energies, the strength of the different hydrogen bonds decreases in the order: COOH-COOH > OH-N > self-associated OH ≈ OH-ether O > OH-π, which is consistent with the reported experimental results. Different substituents have distinct effects on the hydrogen bonds.
-
Key words:
- hydrogen bonds /
- lignite-related complexes /
- substituents /
- density functional theory
-
Table 1 Bond energies of HBs in the different complexes with PBE and PBE-D functional
Functional Bond energy E/(kJ·mol-1) phenol…phenol phenol…benzene phenol…oxydibenzene phenol…quinoline benzoic acid…benzoic acid PBE 18.19 15.37 22.08 43.43 77.82 PBE-D 32.79 28.04 35.00 54.96 87.64 -
[1] LARSEN J W, GREEN T K, KOVAC J. The nature of the macromolecular network structure of bituminous coals[J]. J Org Chem, 1985, 50(24):4729-4735. doi: 10.1021/jo00224a014 [2] LARSEN J W, SHAWVER S. Solvent swelling studies of two low-rank coals[J]. Energy Fuels, 1990, 4(1):74-77. doi: 10.1021/ef00019a013 [3] XIAO J, ZHAO Y P, FAN X, CAO J P, KANG G J, ZHAO W, WEI X Y. Hydrogen bonding interactions between the organic oxygen/nitrogen monomers of lignite and water molecules:A DFT and AIM study[J]. Fuel Process Technol, 2017, 168:58-64. doi: 10.1016/j.fuproc.2017.09.001 [4] LI Z K, YAN H L, YAN J C, WANG Z C, LEI Z P, REN S B, SHUI H F. Drying and depolymerization technologies of Zhaotong lignite:A review[J]. Fuel Process Technol, 2019, 186:88-98. doi: 10.1016/j.fuproc.2019.01.002 [5] LI D, LI W, CHEN H, LI B. The adjustment of hydrogen bonds and its effect on pyrolysis property of coal[J]. Fuel Process Technol, 2004, 85(8/10):815-825. http://www.sciencedirect.com/science/article/pii/S037838200300287X [6] LI H J, LI X H, FENG J, LI W Y. Effect of preheating treatment on oxygen migration during lignite pyrolysis[J]. J Fuel Chem Technol, 2019, 47(1):1-7. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=rlhxxb201901001 [7] HOU R, BAI Z, ZHENG H, FENG Z, YE D, GUO Z, KONG L, BAI J, LI W. Behaviors of hydrogen bonds formed by lignite and aromatic solvents in direct coal liquefaction:Combination analysis of density functional theory and experimental methods[J]. Fuel, 2020, 265:117011. doi: 10.1016/j.fuel.2020.117011 [8] LEI Z P, WU L, ZHANG Y Q, SHUI H F, WANG Z C, REN S B. Effect of noncovalent bonds on the successive sequential extraction of Xianfeng lignite[J]. Fuel Process Technol, 2013, 111:118-122. doi: 10.1016/j.fuproc.2013.02.004 [9] LEI Z P, CHENG L L, ZHANG S F, ZHANG Y Q, SHUI H F, REN S B, WANG Z C. Dissolution performance of coals in ionic liquid 1-butyl-3-methyl-imidazolium chloride[J]. Fuel Process Technol, 2015, 129:222-226. doi: 10.1016/j.fuproc.2014.09.021 [10] CHEN C, GAO J S, YAN Y J. Observation of the type of hydrogen bonds in coal by FT-IR[J]. Energy Fuels, 1998, 12(3):446-449. doi: 10.1021/ef970100z [11] PAINTER P C, SOBKOWIAK M, YOUTCHEFF J. FT-IR. study of hydrogen bonding in coal[J]. Fuel, 1987, 66(7):973-978. doi: 10.1016/0016-2361(87)90338-3 [12] MIURA K, MAE K, HASEGAWA I, CHEN H, KUMANO A, TAMURA K. Estimation of hydrogen bond distributions formed between coal and polar solvents using in situ IR technique[J]. Energy Fuels, 2002, 16(1):23-31. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=J-STAGE_219252 [13] MIURA K, MAE K, LI W, KUSAKAWA T, MOROZUMI F, KUMANO A. Estimation of hydrogen bond distribution in coal through the analysis of OH stretching bands in diffuse reflectance infrared spectrum measured by in-situ technique[J]. Energy Fuels, 2001, 15(3):599-610. http://www.istic.ac.cn/suoguan/detailed.htm?dbname=xw_qk&wid=0220071119244056 [14] HAO P Y, MENG Y J, ZENG F G, YAN T T, XU G B. Quantitative study of chemical structures of different rank coals based on infrared spectroscopy[J]. Spectrosc Spect Anal, 2020, 40(3):787-792. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=gpxygpfx202003022 [15] HUANG X, CHU W, SUN W J, JIANG C F, FENG Y Y, XUE Y. Investigation of oxygen-containing group promotion effect on CO2-coal interaction by density functional theory[J]. Appl Surf Sci, 2014, 299:162-169. doi: 10.1016/j.apsusc.2014.01.205 [16] LI G Y, XIE Q A, ZHANG H, GUO R, WANG F, LIANG Y H. Pyrolysis mechanism of metal-ion-exchanged lignite:A combined reactive force field and density functional theory study[J]. Energy Fuels, 2014, 28(8):5373-5381. doi: 10.1021/ef501156b [17] LI L, FAN H, HU H. A theoretical study on bond dissociation enthalpies of coal based model compounds[J]. Fuel, 2015, 153:70-77. doi: 10.1016/j.fuel.2015.02.088 [18] LI G Y, WANG F, WANG J P, LI Y Y, LI A Q, LIANG Y H. ReaxFF and DFT study on the sulfur transformation mechanism during the oxidation process of lignite[J]. Fuel, 2016, 181:238-247. doi: 10.1016/j.fuel.2016.04.068 [19] LIU J, WU J, ZHU J, WANG Z, ZHOU J, CEN K. Removal of oxygen functional groups in lignite by hydrothermal dewatering:An experimental and DFT study[J]. Fuel, 2016, 178:85-92. doi: 10.1016/j.fuel.2016.03.045 [20] WU J, LIU J, YUAN S, WANG Z, ZHOU J, CEN K. Theoretical investigation of noncovalent interactions between low-rank coal and water[J]. Energy Fuels, 2016, 30(9):7118-7124. doi: 10.1021/acs.energyfuels.6b01377 [21] LI L, FAN H, HU H. Distribution of hydroxyl group in coal structure:A theoretical investigation[J]. Fuel, 2017, 189:195-202. doi: 10.1016/j.fuel.2016.10.091 [22] SUN T, WANG Y B. Calculation of the binding energies of different types of hydrogen bonds using GGA density functional and its long-range, empirical dispersion correction methods[J]. Acta Phys-Chim Sin, 2011, 27(11):2553-2558. doi: 10.3866/PKU.WHXB20111017 [23] JANESKO B G. Modeling interactions between lignocellulose and ionic liquids using DFT-D[J]. Phys Chem Chem Phys, 2011, 13(23):11393-11401. doi: 10.1039/c1cp20072k [24] JOSA D, RODRÍGUEZ-OTERO J, CABALEIRO-LAGO E M, RELLÁN-PIÑEIRO M. Analysis of the performance of DFT-D, M05-2X and M06-2X functionals for studying π-π interactions[J]. Chem Phys Lett, 2013, 557:170-175. doi: 10.1016/j.cplett.2012.12.017 [25] LI B, LIU S, GUO J, ZHANG L. Interaction between low rank coal and kaolinite particles:A DFT simulation[J]. Appl Surf Sci, 2018, 456:215-220. doi: 10.1016/j.apsusc.2018.06.121 [26] LI Z K, ZONG Z M, YAN H L, WANG Y G, WEI X Y, SHI D L, ZHAO Y P, ZHAO C L, YANG Z S, FAN X. Alkanolysis simulation of lignite-related model compounds using density functional theory[J]. Fuel, 2014, 120:158-162. doi: 10.1016/j.fuel.2013.12.009 [27] ZHANG R, XING Y, XIA Y, LUO J, TAN J, RONG G, GUI X. New insight into surface wetting of coal with varying coalification degree:An experimental and molecular dynamics simulation study[J]. Appl Surf Sci, 2020, 511:145610. doi: 10.1016/j.apsusc.2020.145610 [28] STEINER T. The hydrogen bond in the solid state[J]. Angew Chem Int Ed, 2002, 41:48-76. doi: 10.1002/1521-3773(20020104)41:1<48::AID-ANIE48>3.0.CO;2-U [29] LIU F J, WEI X Y, GUI J, WANG Y G, LI P, ZONG Z M. Characterization of biomarkers and structural features of condensed aromatics in Xianfeng lignite[J]. Energy Fuels, 2013, 27(12):7369-7378. doi: 10.1021/ef402027g [30] LI Z K, WEI X Y, YAN H L, ZONG Z M. Insight into the structural features of Zhaotong lignite using multiple techniques[J]. Fuel, 2015, 153:176-182. doi: 10.1016/j.fuel.2015.02.117 -





 下载:
下载: