Effect of preparation methods on the structure and naphthalene hydrogenation performance of Ni2P/SiO2 catalyst
-
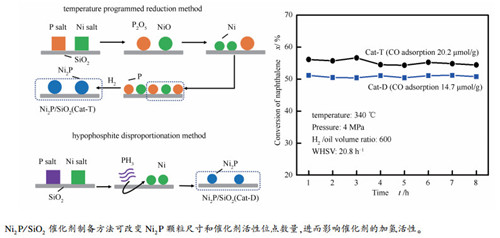
摘要: 采用程序升温还原法和次磷酸盐歧化法制备了Ni2P/SiO2催化剂,结合现代仪器分析表征技术,研究了制备方法对Ni2P/SiO2催化剂结构和萘加氢性能的影响。结果表明,两种方法均可制备出仅含Ni2P活性相的Ni2P/SiO2催化剂,在反应温度340℃、氢气压力4 MPa、空速为20.8 h-1下,程序升温还原法制备的Ni2P/SiO2催化剂表现出更高的萘加氢活性,这主要是因为程序还原法制备的Ni2P/SiO2催化剂中有更多Ni2P物种生成,提供了较多的活性位点(CO吸附量21.6 μmol/g);且催化剂表面弱酸位点多,有利于芳烃吸附。当选用程序升温还原法制备Ni2P/SiO2催化剂时,在保证生成纯相Ni2P的前提下,较低的Ni/P比更有利于合成高加氢活性的Ni2P/SiO2催化剂。Abstract: Ni2P/SiO2 catalysts were prepared by temperature-programmed reduction method and hypophosphite disproportionation method to investigate their naphthalene hydrogenation performance. The prepared catalysts were characterized by ICP-OES, X-ray diffraction, H2 temperature-programmed reduction, N2 adsorption-desorption method and transmission electron microscopy, etc. Results showed that Ni2P/SiO2 catalyst with pure Ni2P crystal phase could be successfully prepared by the temperature-programmed reduction method and hypophosphite disproportionation method. When the naphthalene hydrogenation reaction was performed at 340℃, 4 MPa, H2/oil volume ratio of 600, and a weight hourly space velocity (WHSV) of 20.8 h-1, Ni2P/SiO2 catalyst prepared by the temperature-programmed reduction method possessed superior hydrogenation activity. This result was ascribed to the advantages of temperature-programmed reduction method. It not only installed the higher number of Ni2P species (CO adsorption amount 21.6 μmol/g) over SiO2, but also obtained more weak acid sites on the catalyst surface, which promoted the adsorption of aromatic hydrocarbons and subsequently resulted in the higher hydrogenation activity. Furthermore, when the temperature-programmed reduction method was used to prepare Ni2P/SiO2 catalyst, the lower Ni/P molar ratio was more beneficial to enhance the naphthalene hydrogenation activity of the as-prepared catalyst.
-
表 1 不同制备方法Ni2P/SiO2催化剂的元素组成
Table 1 Element composition of Ni2P/SiO2 catalysts synthesized by different methods
Sample Theory content Actual content Ni w/% P w/% Ni/P (mol ratio) Ni w/% P w/% Ni/P (mol ratio) Cat-D 7.9 8.4 0.5 7.9 5.0 0.8 Cat-T(1) 7.9 4.2 1 8.1 2.5 1.7 Cat-T(1.25) 7.9 3.3 1.25 8.3 3.0 1.4 表 2 不同制备方法催化剂的比表面积和孔结构
Table 2 BET specific surface area and pore structure of the Ni2P/SiO2 catalyst synthesized by different methods
Sample BET specific
surface area
A/(m2·g-1)Total pore
volume
v/(cm3·g-1)Most probable
aperture
d/nmSiO2 209.0 2.1 32.8 Cat-D 146.8 0.5 12.5 Cat-T(1) 145.3 0.7 20.1 Cat-T(1.25) 149.5 0.8 22.3 表 3 不同制备方法所得Ni2P/SiO2催化剂中Ni 2p、P 2p的结合能和Ni/P比
Table 3 Binding energy of Ni 2p and P 2p, the atomic ratio of Ni/P on Ni2P/SiO2 catalysts prepared by different methods
Sample Binding energy E/ eV Ni/P
/(mol ratio)Ni 2p3/2 P 2p3/2 Niδ+ Ni2+ Pδ- PO43- Cat-D 853.42 857.36 129.29 133.84 0.99 Cat-T(1) 853.35 857.26 129.39 133.89 1.05 Cat-T(1.5) 853.31 857.05 129.41 133.84 1.52 -
[1] CORMA A, MARTÍNEZ A, MARTÍNEZ-SORIA V. Hydrogenation of aromatics in diesel fuels on Pt/MCM-41 catalysts[J]. J Catal, 1997, 169(2):480-489. https://www.sciencedirect.com/science/article/pii/S0021951797917371 [2] STANISLAUS A, COOPER B H. Aromatic hydrogenation catalysis:A review[J]. Catal Rev, 2006, 36(1):75-123. doi: 10.1080/01614949408013921 [3] 夏良燕, 夏芝香, 方梦祥, 唐巍, 王勤辉, 骆仲泱.煤焦油中芳烃(萘)的加氢饱和试验[J].浙江大学学报(工学版), 2015, 49(3):578-584. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zjdxxb-gx201503025XIA Liang-yan, XIA Zhi-xiang, FANG Meng-xiang, TANG Wei, WANG Qin-hui, LUO Zhong-yang. Hydrogenation saturation of aromatic compounds (naphthalene) in coal tar[J]. J Zhejiang Univ (Eng Sci)), 2015, 49(3):578-584. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=zjdxxb-gx201503025 [4] 佟瑞利, 王永刚, 张旭, 张海永, 戴谨泽, 林雄超, 许德平. P改性NiW/γ-Al2O3的低温焦油芳烃组分加氢性能研究[J].燃料化学学报, 2015, 43(12):1461-1469. doi: 10.3969/j.issn.0253-2409.2015.12.009TONG Rui-li, WANG Yong-gang, ZHANG Xu, ZHANG Hai-yong, DAI Jin-ze, LIN Xiong-chao, XU De-ping. Effect of phosphorus modification on the catalytic properties of NiW/γ-Al2O3 in the hydrogenation of aromatics from coal tar[J]. J Fuel Chem Technol, 2015, 43(12):1461-1469. doi: 10.3969/j.issn.0253-2409.2015.12.009 [5] HE T, WANG Y, MIAO P, LI J, WU J, FANG Y. Hydrogenation of naphthalene over noble metal supported on mesoporous zeolite in the absence and presence of sulfur[J]. Fuel, 2013, 106:365-371. doi: 10.1016/j.fuel.2012.12.025 [6] 李贺, 殷长龙, 赵雪萍, 李秀峥, 柳云骐, 刘晨光.萘、四氢萘和十氢萘的加氢或脱氢反应与催化剂的研究进展[J].石油化工, 2014, 23(8):971-979. doi: 10.3969/j.issn.1000-8144.2014.08.020LI He, YIN Chang-long, ZHAO Xue-ping, LI Xiu-zheng, LIU Yun-ji, LIU Chen-guang. Progresses in hydrogenation or dehydrogenation of naphthalene, tetralin and decalin, and the catalysts[J]. Petrochem Technol, 2014, 23(8):971-979. doi: 10.3969/j.issn.1000-8144.2014.08.020 [7] LIU D, WANG A, LIU C, PRINS R. Bulk and Al2O3-supported Ni2P HDS catalysts prepared by separating the nickel and hypophosphite sources[J]. Catal Commun, 2016, 77:13-17. doi: 10.1016/j.catcom.2016.01.008 [8] YUN G, GUAN Q, LI W. The synthesis and mechanistic studies of a highly active nickel phosphide catalyst for naphthalene hydrodearomatization[J]. RSC Adv, 2017, 7(14):8677-8687. doi: 10.1039/C7RA00250E [9] YANG Y, LI J, LV G, ZHANG L. Novel method to synthesize Ni2P/SBA-15 adsorbents for the adsorptive desulfurization of model diesel fuel[J]. J Alloy Compd, 2018, 745:467-476. doi: 10.1016/j.jallcom.2018.02.156 [10] D'AQUINO A I, DANFORTH S J, CLINKINGBEARD T R, ILIC B, PULLAN L, REYNOLDS M A, MURRAY B D, BUSSELL M E. Highly-active nickel phosphide hydrotreating catalysts prepared in situ using nickel hypophosphite precursors[J]. J Catal, 2016, 335:204-214. doi: 10.1016/j.jcat.2015.12.006 [11] LI X, FENG J, GUO J, WANG A, PRINS R, DUAN X, CHEN Y. Preparation of Ni2P/Al2O3 by temperature-programmed reduction of a phosphate precursor with a low P/Ni ratio[J]. J Catal, 2016, 334:116-119. doi: 10.1016/j.jcat.2015.11.007 [12] LI J, CHAI Y, LIU B, WU Y, LI X, TANG Z, LIU Y, LIU C. The catalytic performance of Ni2P/Al2O3 catalyst in comparison with Ni/Al2O3 catalyst in dehydrogenation of cyclohexane[J]. Appl Catal A:Gen, 2014, 469:434-441. doi: 10.1016/j.apcata.2013.09.047 [13] SONG H, DAI M, GUO Y T, ZHANG Y J. Preparation of composite TiO2-Al2O3 supported nickel phosphide hydrotreating catalysts and catalytic activity for hydrodesulfurization of dibenzothiophene[J]. Fuel Process Technol, 2012, 96:228-236. doi: 10.1016/j.fuproc.2012.01.001 [14] WANG S, WANG K, WANG X. Novel preparation of highly dispersed Ni2P embedded in carbon framework and its improved catalytic performance[J]. Appl Surf Sci, 2016, 386:442-450. doi: 10.1016/j.apsusc.2016.06.055 [15] LAN X, HENSEN E J M, WEBER T. Silica-supported Ni2P:Effect of preparation conditions on structure and catalytic performance in thiophene hydrodesulfurization (HDS)[J]. Catal Today, 2017, 292:121-132. doi: 10.1016/j.cattod.2016.12.040 [16] ZHANG X, ZHANG Q, ZHAO A, GUAN J, HE D, HU H, LIANG C. Naphthalene hydrogenation over silica supported nickel phosphide in the absence and presence of N-containing compounds[J]. Energy Fuels, 2010, 24(7):3796-3803. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=10848dc5c6db284ae5e7b3c1e235b42f [17] H DOIRE C E, LOUIS C, DAVIDSON A, BREYSSE M, MAUG F, VRINAT M, H DOIRE C E, LOUIS C, DAVIDSON A, BREYSSE M. Support effect in hydrotreating catalysts:Hydrogenation properties of molybdenum sulfide supported on β-zeolites of various acidities[J]. J Catal, 2003, 220(2):433-441. https://www.sciencedirect.com/science/article/pii/S0021951703003087 [18] OYAMA S T, WANG X, LEE Y K, CHUN W J. Active phase of Ni2P/SiO2 in hydroprocessing reactions[J]. J Catal, 2004, 221(2):263-273. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d02b02011f6259ee0fce72460cbef0ad [19] HUA S, MIN D, SONG H, XIA W, XU X. A novel synthesis of Ni2P/MCM-41 catalysts by reducing a precursor of ammonium hypophosphite and nickel chloride at low temperature[J]. Vacuum, 2013, 462-463(27):247-255. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=52ccf707bd8757a5597297c77cd9c953 [20] LIU D, WANG A, LIU C. Bulk and Al2O3-supported Ni2P HDS catalysts prepared by separating the nickel and hypophosphite sources[J]. Catal Commun, 2016, 77(5):13-17. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=412aa624e55137137db3b75efb902e64 [21] LIU D, WANG A, LIU C, PRINS R. Ni2P/Al2O3 hydrodesulfurization catalysts prepared by separating the nickel compound and hypophosphite[J]. Catal Today, 2017, 292(1):133-142. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=dd2fb386e39edc491b26d7af984edba9 [22] CHO K S, SEO H-R, LEE Y K. A new synthesis of highly active Ni2P/Al2O3 catalyst by liquid phase phosphidation for deep hydrodesulfurization[J]. Catal Commun, 2011, 12(6):0-474. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=23c215d18f8a69de8c8394fd30044e9b [23] XIE Y, SU H L, QIAN X F, X.M. A mild one-step solvothermal route to metal phosphides (metal=Co, Ni, Cu)[J]. J Solid State Chem, 2000, 149(1):88-91. https://www.sciencedirect.com/science/article/pii/S0022459699984992 [24] 张子毅, Ni2P/Ce-Al2O3催化剂制备及其萘饱和加氢性能研究[D].太原: 太原理工大学, 2018. http://cdmd.cnki.com.cn/Article/CDMD-10112-1018959349.htmZHANG Zi-yi. Preparation of Ni2P/Ce-Al2O3 catalyst and its saturated hydrogenation performance of naphthalene[D]. Taiyuan: Taiyuan University of Technology, 2018. http://cdmd.cnki.com.cn/Article/CDMD-10112-1018959349.htm [25] 郄志强, Ni2P/Al2O3催化剂的低温制备及其萘加氢饱和性能研究[D].太原: 太原理工大学, 2019.QIE Zhi-qiang. Low temperature synthesis of Ni2P/Al2O3 catalyst and its hydrogenation saturation performance of naphthalene[D]. Taiyuan: Taiyuan University of Technology, 2019. [26] 郄志强, 张子毅, 荆洁颖, 杨志奋, 冯杰, 李文英. Ni2P负载量对Ni2P/Ce-Al2O3催化剂结构及萘加氢性能的影响[J].燃料化学学报, 2019, 47(6):718-724. doi: 10.3969/j.issn.0253-2409.2019.06.009QIE Zhi-qiang, ZHANG Zi-yi, JING Jie-ying, YANG Zhi-fen, FENG Jie, LI Wen-ying, Effect of Ni2P loading on the structure and naphthalene hydrogenation performance of Ni2P/Ce-Al2O3 catalyst[J].J Fuel Chem Technol, 2019, 47(6):718-724. doi: 10.3969/j.issn.0253-2409.2019.06.009 [27] OYAMA S T, WANG X, LEE Y K, BANDO K, REQUEJO F G. Effect of phosphorus content in nickel phosphide catalysts studied by XAFS and other techniques[J]. J Catal, 2002, 210(1):207-217. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f07149cfa7b1d89fff3478c746e22cc5 [28] SAWHILL S J, LAYMAN K A, WYK D R V, ENGELHARD M H, BUSSELL M E. Thiophene hydrodesulfurization over nickel phosphide catalysts:Effect of the precursor composition and support[J]. J Catal, 2005, 231(2):300-313. https://www.academia.edu/4182673/Thiophene_hydrodesulfurization_over_nickel_phosphide_catalysts_effect_of_the_precursor_composition_and_support [29] HUO J, JING J, LI W. Reduction time effect on structure and performance of Ni-Co/MgO catalyst for carbon dioxide reforming of methane[J]. Int J Hydrogen Energy, 2014, 39(36):21015-21023. doi: 10.1016/j.ijhydene.2014.10.086 [30] OYAMA S T, GOTT T, ZHAO H, LEE Y-K. Transition metal phosphide hydroprocessing catalysts:A review[J]. Catal. Today, 2009, 143(1/2):94-107. https://www.sciencedirect.com/science/article/pii/S0920586108004446 [31] ZHU T, SONG H, DAI X, SONG H. Preparation of Ni2P/Al-SBA-15 catalyst and its performance for benzofuran hydrodeoxygenation[J]. Chin J Chem Eng, 2017, 25:1784-1790. doi: 10.1016/j.cjche.2017.03.027 [32] NARANOV E R, SADOVNIKOV A A, MAXIMOV A L. Development of micro-mesoporous materials with lamellar structure as the support of NiW catalysts[J]. Microporous Mesoporous Mater, 2018, 263:150-157. doi: 10.1016/j.micromeso.2017.12.021 [33] SONG H, DAI M, SONG H, WAN X, XU X. A novel synthesis of Ni2P/MCM-41 catalysts by reducing a precursor of ammonium hypophosphite and nickel chloride at low temperature[J]. Appl Catal A:Gen, 2013, 462-463:247-255. doi: 10.1016/j.apcata.2013.05.015 [34] SONG H, WANG J, WANG Z, SONG H, LI F, JIN Z. Effect of titanium content on dibenzothiophene HDS performance over Ni2P/Ti-MCM-41 catalyst[J]. J Catal, 2014, 311:257-265. doi: 10.1016/j.jcat.2013.11.021 -





 下载:
下载: