Catalytic performances of Al-ITQ-13 zeolites with different SiO2/Al2O3 ratios in the conversion of methanol to propene
-
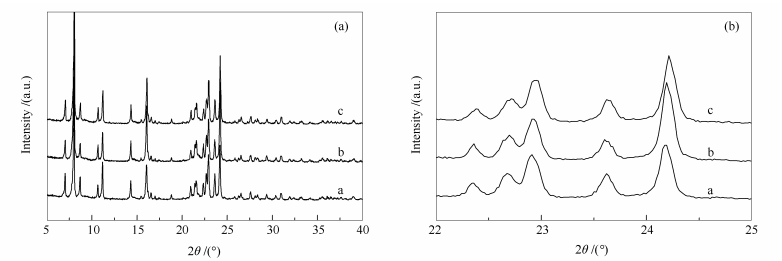
摘要: 采用晶种法直接合成了硅铝比(SiO2/A12O3物质的量比)为137、224和309的三种Al-ITQ-13分子筛,并采用粉末X射线衍射(XRD)、扫描电镜(SEM)、N2吸附-脱附、固体核磁共振(MAS NMR)和NH3-程序升温脱附(NH3-TPD)等分析方法对不同硅铝比分子筛进行了表征,并在固定床微型反应评价装置上,考察了硅铝比对甲醇转化制丙烯反应性能的影响。结果表明,不同硅铝比Al-ITQ-13分子筛呈现出相似的织构性质,酸量及酸强度随着硅铝比的升高逐渐下降。硅铝比对甲醇转化反应的产物分布存在较大的影响;随着硅铝比的升高,氢转移反应和芳构化反应活性降低,使得乙烯选择性下降,而丙烯和丁烯的选择性升高。硅铝比由137提高到309,丙烯的选择性(质量分数)由46.04%增加到55.52%,而丙烯/乙烯比由3.39提高到6.57。Abstract: Al-ITQ-13 zeolites with different SiO2/Al2O3 molar ratios were synthesized by using seeds in the gel and characterized by XRD, SEM, N2 physisorption, MAS NMR and NH3-TPD. The effect of SiO2/Al2O3 molar ratio on the catalytic performance of Al-ITQ-13 in the conversion of methanol to propene (MTP) was investigated in a fixed-bed micro-reactor. The results showed that the Al-ITQ-13 zeolites with different SiO2/Al2O3 molar ratios are similar in their textural properties; however, the amount and strength of acid sites decrease with the increase of SiO2/Al2O3 molar ratio. Moreover, the SiO2/Al2O3 molar ratio has a significant influence on the catalytic behavior of Al-ITQ-13 in MTP. As the hydrogen transfer and aromatization reactions were suppressed over the Al-ITQ-13 zeolite with high SiO2/Al2O3 molar ratio, with the increase of SiO2/Al2O3 molar ratio, the selectivity to propene and butene is increased at the expense of the selectivity to propene; that is, with the increase of SiO2/Al2O3 molar ratio from 137 to 309, the selectivity to propene is increased from 46.04% to 55.52% and meanwhile, the propene/ethene ratio is increased from 3.39 to 6.57.
-
Key words:
- methanol to propene (MTP) /
- Al-ITQ-13 zeolite /
- methanol /
- propene /
- SiO2/Al2O3 molar ratio /
- acidity
-
表 1 不同硅铝比Al-ITQ-13分子筛样品的织构性质
Table 1 Textural properties of various Al-ITQ-13 zeolites
Sample A B C SiO2/Al2O3 in gel 150 250 350 Relatively crystallinity /% 96 93 96 SiO2/Al2O3 in product 137 224 309 ABET /(m2·g-1) 313 310 308 Amicro/(m2·g-1) 290 289 290 vpore /(cm3·g-1) 0.160 0.159 0.159 vmicro /(cm3·g-1) 0.140 0.137 0.137 note: the SiO2/Al2O3 molar ratios in Al-ITQ-13 product were determined by XRF method; BET surface area, micropore surface area and micropore volume were determined by t-plot method; the total pore volume was obtained at p/p0 = 0.99 表 2 不同硅铝比Al-ITQ-13分子筛催化剂上MTP反应产物的分布
Table 2 Product distribution for MTP over the Al-ITQ-13 zeolites with different SiO2/Al2O3 molar ratios
Catalyst Conv. x /% Product selectivity w/% C3H6/C2H4 HTC s(C5+)/s(C2=+ C3=) CH4 C2H6 C2H4 C3H8 C3H6 C4H10 C4H8 C5+ C2H4+C3H6 C2H4 +C3H6+C4H8 A 99.9 3.72 0.77 13.60 2.09 46.04 3.44 23.45 6.88 59.64 83.09 3.39 0.03 0.12 B 100 2.29 0.17 10.52 1.10 49.07 2.15 28.50 6.20 59.59 88.09 4.66 0.01 0.10 C 99.9 1.45 0.39 8.44 1.08 55.52 1.35 29.99 1.78 63.96 93.95 6.57 0.01 0.03 note: the reactions were carried out at atmospheric pressure and 450 ℃, with a WHSV of 1.5 h-1, mCH3OH:mH2O=1:1, time on stream (TOS) of 7 h -
[1] 周伙胜. 2012年丙烯采购策略及方案探讨[J].云南化工, 2012, 39(2):60-64. http://d.wanfangdata.com.cn/Periodical/ynhg201202017ZHOU Huo-sheng. Purchasing strategy and scheme of propylene in 2012[J]. Yunnan Chem Technol, 2012, 39(2):60-64. http://d.wanfangdata.com.cn/Periodical/ynhg201202017 [2] 黄慧文, 孟晓静, 陈晨, 张敏秀, 李春义, 崔秋凯.不同硅铝比HZSM-5分子筛催化剂甲醇制丙烯反应性能研究[J].石化技术与应用, 2016, 34(4):284-289. http://d.wanfangdata.com.cn/Periodical/shjsyyy201604005HUANG Hui-wei, MENG Xiao-jing, CHEN Chen, ZHANG Min-xiu, LI Chun-yi, CUI Qiu-kai. Study on SiO2/Al2O3 molar ratio of HZSM-5 catalyst in methanol to propylene reaction performance[J]. Petrochem Technol Appl, 2016, 34(4):284-289. http://d.wanfangdata.com.cn/Periodical/shjsyyy201604005 [3] 魏入朝, 李春义, 杨朝合, 山红红.氟硅酸铵改性纳米HZSM-5分子筛的表征及催化甲醇制低碳烯烃[J].工业催化, 2011, 19(3):40-44. http://d.wanfangdata.com.cn/Periodical/gych201103009WEI Ru-chao, LI Chun-yi, YANG Chao-he, SHAN Hong-hong. Characterization and catalytic performance of (NH4)2SiF6-modified nanosized HZSM-5 catalyst for conversion of methanol to olefins[J]. Ind Catal, 2011, 19(3):40-44. http://d.wanfangdata.com.cn/Periodical/gych201103009 [4] 郭强胜, 毛东森, 劳嫣萍, 卢冠忠.氟改性对纳米HZSM-5分子筛催化甲醇制丙烯的影响[J].催化学报, 2009, 30(12):1248-1254. doi: 10.3321/j.issn:0253-9837.2009.12.012GUO Qiang-sheng, MAO Dong-sen, LAO Yan-ping, LU Guan-zhong. The effect of fluorine modification on catalytic performance of nanosized HZSM-5 zeolite for conversion of methanol to propene[J]. Chin J Catal, 2009, 30(12):1248-1254. doi: 10.3321/j.issn:0253-9837.2009.12.012 [5] BOIX T, PUCHE M, CAMBLOR M, CORMA A. Synthetic porous crystalline material ITQ-13, its synthesis and use:US6471941[P]. 2002-10-29. http://www.freepatentsonline.com/6471941.html [6] CORMA A, PUCHE M, REY F, SANKAR G, TEAT S J. A zeolite structure (ITQ-13) with three sets of medium-pore crossing channels formed by 9-and 10-rings[J]. Angew Chem Int Ed, 2003, 42:1156-1159. doi: 10.1002/anie.200390304 [7] SKISTAD W, TEKETEL S, BLEKEN F L, BEATO P, BORDIGA S, NILSEN M H, OLSBYE U, SVELLE S, LILLERUD K P. Methanol conversion to hydrocarbons (MTH) over H-ITQ-13(ITH) zeolite[J]. Top Catal, 2014, 57:143-158. doi: 10.1007/s11244-013-0170-7 [8] CASTANEDA R, CORMA A, FORNES V, MARTINEZ-TRIGUERO J, VALENCIA S. Direct synthesis of a 9×10 member ring zeolite (Al-ITQ-13):A highly shape-selective catalyst for catalytic cracking[J]. J Catal, 2006, 238:79-87. doi: 10.1016/j.jcat.2005.11.038 [9] SASTRE G, PULIDO A, CASTANEDA R, CORMA A. Effect of the germanium incorporation in the synthesis of EU-1, ITQ-13, ITQ-22, and ITQ-24 zeolites[J]. J Phys Chem B, 2004, 108:8830-8835. doi: 10.1021/jp0378438 [10] LLOPIS F, SASTRE G, CORMA A. Isomerization and disproportionation of m-xylene in a zeolite with 9-and 10-membered ring pores:Molecular dynamics and catalytic studies[J]. J Catal, 2006, 242:195-206. doi: 10.1016/j.jcat.2006.05.034 [11] CORMA A, VALENCIA. Catalytic cracking process:US 6709572[P]. 2004-3-23. http://www.freepatentsonline.com/6709572.html [12] EMEIS C A. Determination of integrated molar extinction coefficients for infrared absorption bands of pyridine adsorbed on solid acid catalysts[J]. J Catal, 1993, 141:347-354. doi: 10.1006/jcat.1993.1145 [13] ZENG P H, LIANG Y, JI S F, SHEN B J, LIU H H, WANG B J, ZHAO H J, LI M F. Preparation of phosphorus-modified PITQ-13 catalysts and their performance in 1-butene catalytic cracking[J]. J Energy Chem, 2014, 23:193-200. doi: 10.1016/S2095-4956(14)60135-2 [14] 曾鹏晖, 季生福, 梁云, 申宝剑, 付丽娜, 赵晓争. Al-ITQ-13分子筛的碱改性及其催化裂化性能[J].石油化工, 2014, 43(4):405-411. http://d.wanfangdata.com.cn/Periodical/syhg201404009ZENG Peng-hui, JI Sheng-fu, LIANG Yun, SHEN Bao-jian, FU Li-na, ZHAO Xiao-zheng. Alkali modification of Al-ITQ-13 zeolite and its performances for catalytic cracking[J]. Petrochem Technol, 2014, 43(4):405-411. http://d.wanfangdata.com.cn/Periodical/syhg201404009 [15] ZENG P H, GUO X Z, ZHU X C, GUO Q X, WANG Y D, REN S Y, SHEN B J. On the synthesis and catalytic cracking properties of Al-ITQ-13 zeolites[J]. Microporous Mesoporous Mater, 2017, 246:186-192. doi: 10.1016/j.micromeso.2017.03.033 [16] SKISTAD W, TEKETEL S, BLEKEN F L, BEATO P, BORDIGA S, NILSEN M H, OLSBYE U, SVELLE S, LILLERUD K P. Methanol conversion to hydrocarbons (MTH) over H-ITQ-13(ITH) zeolite[J]. Top Catal, 2014, 57:143-158. doi: 10.1007/s11244-013-0170-7 [17] SASTRE G, PULIDO A, CASTANEDA R, CORORM A. Effect of the germanium incorporation in the synthesis of EU-1, ITQ-13, ITQ-22, and ITQ-24 zeolites[J]. J Phys Chem B, 2004, 108(26):8830-8835. doi: 10.1021/jp0378438 [18] CORMA A, REY F, VALENCIA S. A zeolite with interconnected 8-, 10-and 12-ring pores and its unique catalytic selectivity[J]. Nat Mater, 2003, 2:493-497. doi: 10.1038/nmat921 [19] 焦永东, 朱斌, 林民, 朱华元. ITQ-13分子筛的制备、表征与轻烃芳构化催化性能研究[J].石油学报(石油加工), 2006, (增刊):184-187. http://d.wanfangdata.com.cn/Conference_6201064.aspxJIAO Yong-dong, ZHU Bin, LIN Ming, ZHU Hua-yuan. Synthesis, characterization and catalytic light olefin aromatization performance of zeolite ITQ-13[J]. Acta Pet Sin (Pet Process Sect), 2006, (Supplement):184-187. http://d.wanfangdata.com.cn/Conference_6201064.aspx [20] XU G, ZHU X, LIU S, XIE S, XU L. Synthesis of pure silica ITQ-13 zeolite using fumed silica as silica source[J]. Microporous Mesoporous Mater, 2010, 129:278-284. doi: 10.1016/j.micromeso.2009.10.005 [21] XU C, GUAN J. Guan, WU S, KAN Q. Effect of silica source on the hydrothermal synthesis of ITQ-13 zeolite[J]. Acta Phys Chim Sin, 2009, 25:2275-2278. http://en.cnki.com.cn/Article_en/CJFDTOTAL-WLHX200911019.htm [22] REN X, LIU J, LI Y, YU J, XU R. Hydrothermal synthesis of an ITH-type germanosilicate zeolite in a non-concentrated gel system[J] J Porous Mater, 2013, 20:975-981. doi: 10.1007/s10934-013-9676-4 [23] 魏入朝. ZSM-5分子筛的合成、表征及MTO反应性能研究[D]. 青岛: 中国石油大学(华东), 2011. http://cdmd.cnki.com.cn/Article/CDMD-10425-1011287236.htmWEI Ru-chao. Synthesis, characterization of ZSM-5 zeolite and studies on its catalytic performance for the reaction of MTO[D]. Qingdao:China University of Petroleum (Huadong), 2011. http://cdmd.cnki.com.cn/Article/CDMD-10425-1011287236.htm [24] ZHAO T S, TAKEMOTO T, YONEYAMA Y. Selective conversion of dimethyl ether to propylene and light olefins over modified H-ZSM-5[J]. Chem Lett, 2005, 34(7):970-971. doi: 10.1246/cl.2005.970 -





 下载:
下载: