NO removal using oxidation of free radicals produced from Fe2+ and heat synergic activation of oxone
-
摘要: 在气液撞击流反应器中,研究了Fe2+协同热活化过一硫酸氢钾盐诱导自由基脱除模拟烟气中的NO。考察了主要工艺参数(溶液温度、Fe2+浓度、过一硫酸氢钾盐浓度、溶液pH值、NO入口浓度)对NO脱除效率的影响。分析检测了反应产物和自由基。基于不同系统的对比研究、反应产物检测和活性自由基的捕获,揭示了NO脱除过程的机制和反应路径。结果表明,提高溶液温度、Fe2+浓度和过一硫酸氢钾盐浓度均提高了NO的脱除效率,而提高溶液pH值和NO入口浓度均降低了NO的脱除效率。Fe2+和热对活化过一硫酸氢钾盐产生自由基有显著的协同效应。自由基氧化是NO脱除的主要路径,而过一硫酸氢钾盐直接氧化是次要的脱除路径。Fe2+和热的协同活化体系具有比其他体系高得多的NO脱除率。Abstract: The NO removal using oxidation of free radicals produced from Fe2+ and heat synergic activation of oxone in a gas-liquid impinging stream reactor was investigated. The effects of several main process parameters (solution temperature, Fe2+ concentration, oxone concentration, solution pH value, NO inlet concentrations) on NO removal were examined. The reaction products and free radicals were also detected and analyzed. Based on the comparative study of different systems, detection of reaction products and capture of active free radicals, the mechanism and reaction pathways of NO removal process were revealed. The results indicate that the increase in oxone concentration, solution temperature or Fe2+ concentration elevates the NO removal efficiency, but the increase in the solution pH value or NO inlet concentration reduces the NO removal efficiency. A synergistic effect between Fe2+ and heat, which activates the oxone to generate sulfate radicals and hydroxyl radicals, was observed. It reveals that the sulfate radicals and hydroxyl radicals are the primary reactive oxidants, and oxone is the complementary oxidant for NO removal. The synergistic activation system of Fe2+ and heat has much higher NO removal efficiency than other systems.
-
Key words:
- oxone /
- Fe2+ activation /
- heat activation /
- NO /
- free radicals oxidation
-
图 1 实验装置示意图
Figure 1 Schematic diagram of experimental apparatus
1-4: cylinder gases (N2/O2/SO2/NO); 5-8: rotameters; 9: gas blender; 10: gas valves; 11: thermometer; 12: constant temperature device; 13: impinging stream reactor; 14: accelerating tube; 15: atomizing nozzles; 16: solution; 17: circulating pump; 18: flue gas analyzer; 19: scrubber bottle; 20: nozzles distribution
图 2 溶液温度, Fe2+浓度, oxone浓度, 溶液pH值, NO入口质量浓度(e)对NO脱除效率的影响
Figure 2 Effects of solution temperature (a), Fe2+ concentration (b), oxone concentration (c), solution pH value (d) and NO concentration (b) on NO removal efficiency
experimental conditions: Fe2+ concentration, 0.03 mol/L; oxone concentration, 0.35 mol/L; solution temperature, 338 K; solution pH value, 2.09; Liquid-gas ratio, 10.0; O2 concentration, 6.0%; NO concentration, 401 mg/m3; SO2 concentration, 2286 mg/m3
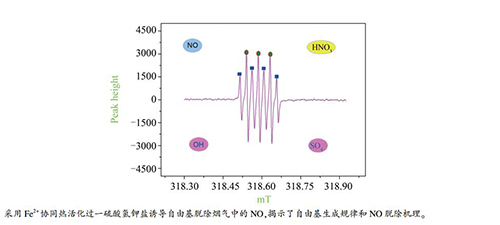
图 3 电子自旋共振(ESR)捕获技术捕获的羟基和硫酸根自由基
Figure 3 Capture of SO4-· and ·OH using ESR spectrometer
(basic experimental conditions are same as those under Figure 2 title)
图 4 不同脱除系统中NO脱除效率的对照
Figure 4 Comparison of NO removal efficiency in different removal systems
(basic experimental conditions are same as those in Figure 2 title)
A: oxone+298 K; B: oxone+338K; C: oxone-298 K+0.03 mol/L Fe2+; D: oxone-338 K+0.03 mol/L Fe2+; E: 338 K+0.03 mol/L Fe2+表 1 反应产物检测和氮元素的质量平衡
Table 1 Measurement of reaction products and mass balance for N
10 min SO42- SO32- NO3- NO2- NO2 Measured anion concentration /(mg·L-1) 4.3× 104 0 5.4 0 0 Calculated anion concentration /(mg·L-1) - - 6.2 - - Relative error /% - - 12.9 - - 20 min SO42- SO32- NO3- NO2- NO2 Measured anion concentration /(mg·L-1) 4.8 × 104 0 11.4 0 0 Calculated anion concentration /(mg·L-1) - - 12.3 - - Relative error /% - - 7.3 - - -
[1] SU C Y, RAN X, HU J L, SHAO C L. Photocatalytic process of simultaneous desulfurization and denitrification of flue gas by TiO2-polyacrylonitrile nanofibers[J]. Environ Sci Technol, 2013, 47(20):11562-11568. doi: 10.1021/es4025595 [2] WEI Z S, NIU H J, JI Y F. Simultaneous removal of SO2 and NOx by microwave with potassium permanganate over zeolite[J]. Fuel Process Technol, 2009, 90(2):324-329. doi: 10.1016/j.fuproc.2008.09.005 [3] LIU Y X, ZHNAG J, SHENG C D. Simultaneous removal of NO and SO2 from coal-fired flue gas by UV/H2O2 advanced oxidation process[J]. Chem Eng J, 2010, 162(3):1006-1011. doi: 10.1016/j.cej.2010.07.009 [4] LIU Y X, ZHNAG J, PAN J F, TANG A K. Investigation on the removal of NO from SO2 containing simulated flue gas by an ultraviolet/fenton-like reaction[J]. Energy Fuels, 2012, 26(9):5430-5436. doi: 10.1021/ef3008568 [5] ADEWUYI Y G, KHAN N E. Modeling the ultrasonic cavitation-enhanced removal of nitrogen oxide in a bubble column reactor[J]. AIChE J, 2012, 58(8):2397-2411. doi: 10.1002/aic.12751 [6] GUO R T, PAN W G, ZHANG X B, REN J X. Removal of NO by using Fenton reagent solution in a lab-scale bubbling reactor[J]. Fuel, 2011, 90(11):3295-3298. doi: 10.1016/j.fuel.2011.06.030 [7] ZHAO Y, WEN X Y, GUO T X, ZHOU J H. Desulfurization and denitrogenation from flue gas using Fenton reagent[J]. Fuel Process Technol, 2014, 128(10):54-60. http://www.sciencedirect.com/science/article/pii/S0378382014002884 [8] ZHAO Y, YUAN B, SHEN Y, HAO R, YANG S. Simultaneous removal of NO and SO2 from flue gas using vaporized H2O2 catalyzed by nanoscale zero-valent iron[J]. Environ Sci Pollut R, 2018, 25(25):1-12. doi: 10.1007/s11356-018-2628-4 [9] LIU Y X, WANG Y. Elemental mercury removal from flue gas using heat and Co2+/Fe2+ coactivated oxone oxidation system[J]. Chem Eng J, 2018, 348(15):464-475. [10] XU W, LIU Y X, WANG Q, ZHANG J, PAN J F. Removal of nitric oxide from flue gas using sulfate/hydroxyl radicals from activation of oxone with cobalt and high temperature[J]. Environ Prog Sustainable Energy, 2017, 36(4):1013-1021. doi: 10.1002/ep.v36.4 [11] LIU Y X, WANG Q. Removal of elemental mercury from flue gas by thermally activated ammonium persulfate in a bubble column reactor[J]. Environ Sci Technol, 2014, 48(20):12181-12189. doi: 10.1021/es501966h [12] ADEWUYI Y G, OWUSU S O. Aqueous absorption and oxidation of nitric oxide with oxone for the treatment of tail gases:Process feasibility, stoichiometry, reaction pathways, and absorption rate[J]. Ind Eng Chem Res, 2003, 42(17):4084-4100. doi: 10.1021/ie020709+ [13] WU Y. Impinging Streams:Fundamentals, Properties, and Applications[M]. Amsterdam:Elsevier, 2007. [14] LIU Y X, WANG Y, YIN Y S, PAN J F, ZHANG J. Oxidation removal of nitric oxide from flue gas using ultraviolet light (UV) and heat coactivated oxone system[J]. Energy Fuels, 2018, 32(2):1999-2008. doi: 10.1021/acs.energyfuels.7b03165 [15] LIU Y X, WANG Y, YANG W, PAN Z H, WANG Q. Simultaneous oxidation-absorption of nitric oxide and sulfur dioxide using aqueous ammonium persulfate synergistically activated by UV-light and heat[J]. Chem Eng Res Des, 2018, 130(2):321-333. http://www.sciencedirect.com/science/article/pii/S0263876217307177 [16] ADEWUYI Y G, SAKYI N Y. Removal of nitric oxide by aqueous sodium persulfate simultaneously activated by temperature and Fe2+ in a lab-scale bubble reactor[J]. Ind Eng Chem Res, 2013, 52(41):14687-14697. doi: 10.1021/ie4025177 [17] ADEWUYI Y G, KHAN M A, SAKYI N Y. Kinetics and modeling of the removal of nitric oxide by aqueous sodium persulfate simultaneously activated by temperature and Fe2+[J]. Ind Eng Chem Res, 2014, 53(2):828-839. doi: 10.1021/ie402801b [18] LIU Y X, WANG Y, WANG Q, PAN J F, ZHANG J. Simultaneous removal of NO and SO2 using vacuum ultraviolet light (VUV)/heat/peroxymonosulfate (PMS)[J]. Chemosphere, 2018, 190(1):431-441. http://www.ncbi.nlm.nih.gov/pubmed/29024887 [19] LIU Y X, XU W, PAN J F, WANG Q. Oxidative removal of NO from flue gas using ultrasound, Mn2+/Fe2+ and heat coactivation of oxone in an ultrasonic bubble reactor[J]. Chem Eng J, 2017, 326(10):1166-1176. http://www.sciencedirect.com/science/article/pii/S1385894717309774 -





 下载:
下载: