Oxidation of formic acid over palladium catalyst supported on activated carbon derived from polyaniline and modified lignosulfonate composite
-
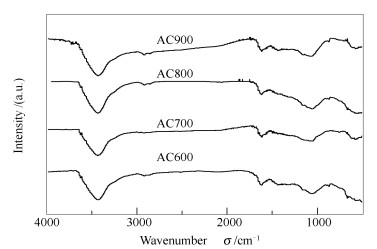
摘要: 将聚苯胺/改性木质素磺酸钠复合材料在不同炭化温度下进行处理得到活性炭材料,利用红外光谱、拉曼光谱、比表面积分析和扫描电镜等手段对其结构和表面性质进行了表征。通过液相还原方法将Pd纳米颗粒负载在所制备的活性炭材料上,获得Pd/C催化剂用于甲酸氧化,并采用X射线衍射、透射电镜和电化学测试等方法对该Pd/C催化剂进行表征。结果表明,以800℃下炭化得到的活性炭材料为载体所制备的Pd-AC800催化剂其催化性能最优;Pd粒径为5.4 nm,电化学活性面积为53.78 m2/g。由于在该催化剂上甲酸氧化通过直接途径进行,Pd-AC800可用作直接甲酸燃料电池的催化剂。Abstract: Activated carbons (AC) were obtained through carbonization of polyaniline and modified lignosulfonate composite (PAn-MLS) under different temperatures; they are characterized by fourier transform infrared spectroscopy (FT-IR), Raman spectroscopy, nitrogen sorption and scanning electron microscope (SEM). With these carbon materials as the support, a series of Pd-AC catalysts for the oxidation of formic acid were prepared by liquid phase reduction and characterized by X-ray diffraction (XRD), transmission electron microscope (TEM) and electrochemical analysis. The results show that with the activated carbon prepared at 800℃ (AC800) as the support, the Pd-AC800 catalyst obtained performs best in the oxidation of formic acid; the palladium particles have an average size of 5.4 nm and the electro-active surface area reaches 53.78 m2/g. As the oxidation of formic acid over Pd-AC800 is realized through direct pathway rather than CO pathway, Pd-AC800 may be considered as a potential electrode material in direct formic acid fuel cells (DFAFC).
-
Key words:
- polyaniline /
- modified lignosulfonate /
- activated carbon /
- Pd-AC /
- formic acid /
- oxidation /
- direct formic acid fuel cells
-
Table 1 Elemental composition of AC600, AC700, AC800 and AC900
Sample Content w/% C H N AC600 78.63 0.31 0.93 AC700 85.77 0.22 0.83 AC800 84.97 0.29 1.32 AC900 88.43 0.20 0.63 -
[1] REIS A, MERT S. Performance assessment of a direct formic acid fuel cell system through exergy analysis[J]. Int J Hydrogen Energ, 2015, 40: 12776-12783. doi: 10.1016/j.ijhydene.2015.07.131 [1] 刘波, 贺志佳, 金昊.风力发电现状与发展趋势[J].东北电力大学学报, 2016, 36(2):7-13. http://www.cqvip.com/QK/91366A/201305/47803838.htmlLIU Bo, HE Zhi-jia, JIN Hao. Wind power status and development trends[J]. J Northeast Dianli Univ, 2016, 36(2):7-13. http://www.cqvip.com/QK/91366A/201305/47803838.html [2] 胡鹏飞, 李勇.地热-太阳能联合有机朗肯循环发电技术研究[J].东北电力大学学报, 2015, 35(5):41-45. http://www.cnki.com.cn/Article/CJFDTotal-DBDL201505009.htmHU Peng-fei, LI Yong. Solar-geothermal hybrid cycle of Organic Rankine Cycle (ORC) analysis in power generation[J]. J Northeast Dianli Univ, 2015, 35(5):41-45. http://www.cnki.com.cn/Article/CJFDTotal-DBDL201505009.htm [3] REIS A, MERT S. Performance assessment of a direct formic acid fuel cell system through exergy analysis[J]. Int J Hydrogen Energy, 2015, 40(1):12776-12783. https://www.researchgate.net/publication/281052557_Performance_assessment_of_a_direct_formic_acid_fuel_cell_system_through_exergy_analysis [4] 贾若琨, 李艺花. Fe3O4-Au功能纳米复合材料的制备及催化性能[J].东北电力大学学报, 2016, 36(3):75-79.JIA Ruo-kun, LI Yi-hua. Preparation and catalytic properties of Fe3O4-Au functional nanocomposites[J]. J Northeast Dianli Univ, 2016, 36(3):75-79. [5] BAHARI A, HEKMATARA H, SEPAHVAND R, ADELI M. Carbon nanotube-graft-poly (citric acid) containing silver and palladium nanoparticles[J]. Nano, 2009, 4(4):217-223. doi: 10.1142/S179329200900168X [6] LESIAK B, MAZURKIEWICZ M, MALOLEPSZY A, STOBINSKI L, MIERZWA B, MIKOLAJCZUK-ZYCHORA A, JUCHNIEWICZ K, BORODZINSKI A, ZEMEK J, JIRICEK P. Effect of the Pd/MWCNTs anode catalysts preparation methods on their morphology and activity in a direct formic acid fuel cell[J]. Appl Surf Sci, 2016, 387(1):929-937. https://www.researchgate.net/publication/304577454_Effect_of_the_PdMWCNTs_anode_catalysts_preparation_methods_on_their_morphology_and_activity_in_a_direct_formic_acid_fuel_cell?_sg=L7XwybdX4OQwp_Yh_X7G91sWKtMJsRrgnU8Ooy_DMWnP4qmU8JBPpplbpvlVoeQynZIlfAG4IMoeJmAcMk08sw [7] 张瑛洁, 吕立杰, 史长东.多孔硅/炭复合电吸附电极材料的研究进展[J].东北电力大学学报, 2015, 35(5):17-22. http://www.sciencedirect.com/science/article/pii/S0013468616303760ZHANG Ying-jie, LV Li-jie, SHI Chang-dong. Study progress in porous silicon/carbon composite electrode materials of electric adsorption[J]. J Northeast Dianli Univ, 2015, 35(5):17-22. http://www.sciencedirect.com/science/article/pii/S0013468616303760 [8] GHARIBI H, GOLMOHAMMADI F, KHEIMAND M. Palladium/cobalt coated on multi-walled carbon nanotubes as an electro-catalyst for oxygen reduction reaction in passive direct methanol fuel cells[J]. Fuel Cells, 2013, 13(1):987-1004. doi: 10.1002/fuce.201200220/abstract [9] SAWANGPHRUK M, KRITTAYAVATHANANON A, CHINWIPAS N, SRIMUK P, VATANATHAM T, LIMTRAKUL S, FOORD J. Ultraporous palladium supported on graphene-coated carbon fiber paper as a highly active catalyst electrode for the oxidation of methanol[J]. Fuel Cells, 2013, 13(1):881-888. https://www.researchgate.net/publication/264260428_Ultraporous_Palladium_Supported_on_Graphene-Coated_Carbon_Fiber_Paper_as_a_Highly_Active_Catalyst_Electrode_for_the_Oxidation_of_Methanol [10] WANG L, ZHANG Y, LOU Y, GUO Y, LU G, GUO Y. Pd catalyst supported on activated carbon honeycomb monolith for CO oxidation and the application in air purification of vehicular tunnel[J]. Fuel Process Technol, 2014, 122(1):23-29. http://or.nsfc.gov.cn/handle/00001903-5/210952 [11] CHEN T, LI D, JIANG H, XIONG C. High-performance Pd nanoalloy on functionalized activated carbon for the hydrogenation of nitroaromatic compounds[J]. Chem Eng J, 2015, 259(1):161-169. http://www.sciencedirect.com/science/article/pii/S1385894714009449 [12] NSIB M F, SAAFI S, RAYES A, MOURSSA N, HOUAS A. Enhanced photocatalytic performance of Ni-ZnO/polyaniline composite for the visible-light driven hydrogen generation[J]. J Energy Inst, 2016, 89(4):694-703. doi: 10.1016/j.joei.2015.05.001 [13] TAKPIRE S R, WAGHULEY S A. Investigating the optical and structural properties of PANi/Ti polymer composites for photovoltaic application[J]. J Energy Inst, 2017, 90(1):44-50. doi: 10.1016/j.joei.2015.11.001 [14] YUAN D, ZHOU T, ZHOU S, ZOU W, MO S, XIA N. Nitrogen-enriched carbon nanowires from the direct carbonization of polyaniline nanowires and its electrochemical properties[J]. Electrochem Commun, 2011, 13(3):242-246. doi: 10.1016/j.elecom.2010.12.023 [15] JIA Y, JIANG J, SUN K. Pyrolysis of polyaniline-poly (styrene sulfonate) hydrogels to prepare activated carbons for the adsorption of vitamin B12[J]. J Anal Appl Pyrolysis, 2015, 111(1):247-253. https://www.researchgate.net/publication/268821787_Pyrolysis_of_polyaniline-polystyrene_sulfonate_hydrogels_to_prepare_activated_carbons_for_the_adsorption_of_vitamin_B12 [16] LV Q, HE Z, ZHANG J, LIN Q. Preparation and properties of nitrogen-containing hollow carbon nanospheres by pyrolysis of polyaniline-lignosulfonate composites[J]. J Anal Appl Pyrolysis, 2011, 92(1):152-157. doi: 10.1016/j.jaap.2011.05.009 [17] DAI T, JIA Y. Supramolecular hydrogels of polyaniline-poly (styrene sulfonate) prepared in concentrated solutions[J]. Polymer, 2011, 52(12):2550-2558. doi: 10.1016/j.polymer.2011.04.006 [18] JIA Y, JIANG J, SUN K. Electrocatalytic performance of Pt supported on polyaniline-poly (styrene sulfonate) hydrogel[J]. J Appl Polym Sci, 2012, 125(1):3702-3707. doi: 10.1002/app.36712/abstract?globalMessage=0 [19] JIA Y, JIANG J, SUN K, DAI T. Enhancement of capacitance performance of activated carbon-Polyaniline composites by introducing methyl orange[J]. Electrochim Acta, 2012, 71(1):213-218. https://www.researchgate.net/publication/256697084_Enhancement_of_capacitance_performance_of_activated_carbon-Polyaniline_composites_by_introducing_methyl_orange [20] TAYLOR K K, COLE C V, SOORA R, DILDAY J C, HILL A M, BERRY B, VISWANTHAN T. The use of lignosulfonic acid in the synthesis of water-dispersible polyaniline[J]. J Appl Polym Sci, 2008, 108(1):1496-1500. http://www.academia.edu/7984129/The_use_of_lignosulfonic_acid_in_the_synthesis_of_water-dispersible_polyaniline [21] TRCHOVÁ M, KONYUSHENKO E, STEJSKAL J, KOVÁROVÁ J, CIRIC-MARJANOVIC G. The conversion of polyaniline nanotubes to nitrogen-containing carbon nanotubes and their comparison with multi-walled carbon nanotubes[J]. Polym Degrad Stabil, 2009, 94(6):929-938. doi: 10.1016/j.polymdegradstab.2009.03.001 [22] ROZLIVKOVA Z, TRCHOVA M, EXNEROVA M, STEJSKAL J. The carbonization of granular polyaniline to produce nitrogen-containing carbon[J]. Synth Met, 2011, 161(11/12):1122-1129. https://www.researchgate.net/publication/238502365_The_carbonization_of_granular_polyaniline_to_produce_nitrogen-containing_carbon [23] 曹剑瑜, 汤佳丽, 宋玲政, 许娟, 王文昌, 陈智栋. EDTA对活性炭的功能化处理及其对炭载Pd催化剂电催化性能的影响[J].物理化学学报, 2013, 29(1):144-150. http://www.whxb.pku.edu.cn/EN/abstract/abstract28248.shtmlCAO Jian-yu, TANG Jia-li, SONG Ling-zheng, XU Juan, WANG Wen-chang, CHEN Zhi-dong. Functionalization of activated carbon with EDTA and its effect on electrocatalytic performance of carbon supported Pd catalysts[J]. Acta Phys-Chim Sin, 2013, 29(1):144-150. http://www.whxb.pku.edu.cn/EN/abstract/abstract28248.shtml [24] WANG K, ZHAO J, LI H, ZHANG X, SHI H. Removal of cadmium (Ⅱ) from aqueous solution by granular activated carbon supported magnesium hydroxide[J].[QX (Y12#]J Taiwan Inst Chem Eng, 2016, 61(1):287-291. [25] HUANG X, JIA K, LIU X. Nano titanium dioxide/multi-walled carbon nanotube heterostructure containing single one carbon nanotube and its electromagnetic properties[J]. Nano, 2015, 10(7):20151550102-1-1550102-7. doi: 10.1142/S1793292015501027 [26] WANG H, KONG W, ZHU W, WANG L, YANG S, LIU F. One-step synthesis of Pd nanoparticles functionalized crystalline nanoporous CeO2 and their application for solvent-free and aerobic oxidation of alcohols[J]. Catal Commun, 2014, 50(1):87-91. https://www.researchgate.net/publication/260800091_One-step_synthesis_of_Pd_nanoparticles_functionalized_crystalline_nanoporous_CeO2_and_their_application_for_solvent-free_and_aerobic_oxidation_of_alcohols [27] LIU B, ZHENG L, LIAO S, ZENG J. Volume production of high loading Pt/C catalyst with high performance via a microwave-assisted organic colloid route[J]. J Power Sources, 2012, 210(1):54-59. http://adsabs.harvard.edu/abs/2012JPS...210...54L [28] HA S, LARSEN R, ZHU Y, MASEL R. Direct formic acid fuel cells with 600mA·cm-2 at 0.4V and 22℃[J]. Fuel Cells, 2004, 4(1):337-343. [29] HA S, LAREN R, MASEL R. Performance characterization of Pd/C nanocatalyst for direct formic acid fuel cells[J]. J Power Sources, 2005, 144(1):28-34. doi: 10.1016/j.jpowsour.2004.12.031 [30] FATHIRAD F, AFZALI D, MOSTAFAVI A. Bimetallic Pd-Zn nanoalloys supported on Vulcan XC-72R carbon as anode catalysts for oxidation process in formic acid fuel cell[J]. Int J Hydrogen Energy, 2016, 41(30):13220-13226. doi: 10.1016/j.ijhydene.2016.05.098 -





 下载:
下载: