Synthesis of Cu-Al spinels and its non-isothermal formation kinetics analysis
-
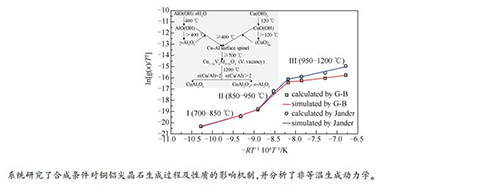
摘要: 以氢氧化铜和拟薄水铝石为原料,通过固相法合成了Cu-Al尖晶石,研究了合成温度、合成时间、Cu/Al物质的量比对尖晶石的生成、晶相组成和还原性能的影响。结果表明,配位缺陷的Cu-Al表面尖晶石在400℃已经生成,难还原尖晶石Cu2+和易还原尖晶石Cu2+分别在700和800℃已生成,尖晶石含量随合成温度升高而不断增多,生成Cu/Al物质的量比不等的富Al尖晶石固溶体,至1200℃生成计量比尖晶石CuAl2O4,因此,尖晶石还原性能随合成温度显著变化。Al适度过量时(Cu/Al(molar ratio)=1:3),在950℃生成难还原Cu2+物种含量较高(约为25.9%)的尖晶石固溶体;Cu过量时在1200℃生成CuAlO2,两者都比计量尖晶石CuAl2O4难还原。另外,延长合成时间也能促进尖晶石生成。非等温动力学分析表明,Cu-Al尖晶石的生成随温度表现出三个动力学区域,即700-850、850-950和950-1200℃,表观活化能分别为85.2、304.4和38.1 kJ/mol。当温度低于950℃时生成的产物层较薄,反应物通过产物层的扩散可认为是一维扩散;超过950℃后产物层变厚,反应物接近于三维发射扩散。Abstract: Cu-Al spinels were synthesized by a solid phase method using Cu(OH)2 and pseudo-boehmite as the raw materials. The effects of synthesis temperature, synthesis time and Cu/Al molar ratio on the formation and properties of Cu-Al spinels were fully investigated by the thermogravimetry(TG/DTG), X-ray diffraction(XRD), H2 temperature programmed reduction(H2-TPR). The non-isothermal kinetics of Cu-Al spinel formation process were analyzed using Coats-Redfern method and two diffusion-controlled kinetic models. Characterization results showed that the Cu-Al surface spinels with unsaturated coordination formed easily at the temperature as low as 400℃, and the content of these surface spinel decreased sharply with the synthesis temperature rising. The hardly-reducible spinel Cu2+ species and easily-reducible spinel Cu2+ species were identified at the synthesis temperature of 700 and 800℃, respectively. The spinel content increased gradually with the synthesis temperature increasing, leading to the formation of Al-rich spinel solid solutions with different Cu/Al molar ratios. At a higher temperature of 1200℃, however, the formation of stoichiometric CuAl2O4 spinel was observed. Hence, the spinel reducibility varied dramatically with the synthesis temperature as illustrated by the drastic change of the molar ratio of hardly-reducible spinel Cu2+ species and easily-reducible spinel Cu2+ species. An appropriate excess of Al3+(Cu/Al=1:3) could result in the formation of spinel solid solution with more hardly-reducible spinel Cu2+ species, while an excess of Cu2+ would lead to the formation of delafossite-type CuAlO2. Both samples owned low reducibility as compared to the stochiometric CuAl2O4 spinel. Besides, a longer synthesis time would favor the spinel formation as well but to a limited extent. Non-isothermal kinetics analysis showed that the formation process of Cu-Al spinel owned three kinetic regions in terms of synthesis temperature, namely 700-850, 850-950 and 950-1200℃, and the apparent activation energies were determined to be 85.2, 304.4 and 38.1 kJ/mol, respectively. The diffusion of reactants via product layer could be considered as an one-dimensional diffusion below 950℃, whereas it was more likely to be a three-dimensional diffusion above 950℃, indicating that the product layer became much thicker.
-
Key words:
- Cu-Al spinel /
- solid phase method /
- synthetic conditions /
- non-isothermal kinetics
-
表 1 CA2-t Cu-Al尖晶石催化剂的物化性质
Table 1 Physicochemical properties of the synthesized Cu-Al spinel catalysts CA2-t
CA2-700 CA2-800 CA2-850 CA2-900 CA2-950 CA2-1000 CA2-1100 CA2-1200 dCuO /nm[a] 20.7 23.5 26.1 26.3 30.6 33.8 35.4 - dspinel /nm[a] -[e] - 6.9 9.8 16.9 22.8 31.8 34.9 a /nm[b] - - 0.8007 0.8044 0.8062 0.8065 0.8071 0.8078 I(440)/I(311) - - 0.33 0.26 0.20 0.21 0.19 0.20 x[c] 0.2866 0.2563 0.2285 0.1351 0.0613 0.0428 0.0196 0.0010 Xspinel /%[d] 10.9 18.4 25.6 52.4 76.8 83.6 92.4 99.6 [a]: the crystallite sizes of CuO and Cu-Al spinel; [b]: the cell parameters of Cu-Al spinel; [c]: x value in Cu1-3xV xAl2+2xO4(V: vacancy); [d]: the molar ratio of spinel Cu to total Cu; [e]: the data cannot be obtained by XRD characterization results 表 2 CAx-tCu-Al尖晶石催化剂的物化性质
Table 2 Physicochemical properties of the synthesized Cu-Al spinel catalysts of CAx-t
CA-950 CA2-950 CA3-950 CA9-950 CA-1200 CA2-1200 CA3-1200 CA9-1200 a/nm[a] 0.8061 0.8063 0.8053 0.7980 - 0.8077 0.8076 0.8073 dspinel /nm[b] 17.5 16.9 12.7 6.0 - 34.9 34.5 30.9 XCuO /%[c] 59.4 13.4 5.2 0 5.89 0.0 0.0 0.0 XCu-O-Al /%[c] 4.4 9.8 13.4 44.7 - 0.4 0.2 1.5 Xspinel /%[c] 36.2 76.8 81.3 55.3 - 99.6 99.8 98.5 (Cu/Al)spinel[d] 0.362 0.384 0.271 0.061 - 0.498 0.499[e] 0.493[e] [a]: the cell parameters of Cu-Al spinel; [b]: the crystallite sizes of CuO and Cu-Al spinel; [c]: the molar ratio of different Cu2+ to total Cu; [d]: the Cu/Al molar ratio of Cu-Al spinel phase; [e]: the value was obtained by the assumption that all excessive Al3+ ions were participated out from spinel solid solution in the form of α-Al2O3, as illustrated in CA3-1200[11] 表 3 Cu-Al尖晶石的非等温合成动力学拟合
Table 3 Simulation results of Cu-Al spinel non-isothermal formation kinetics
t/℃ Ⅰ(700-850 ℃) Ⅱ(850-950 ℃) Ⅲ(950-1200 ℃) Model Jander G-B Jander G-B Jander G-B R2 0.986 0.986 0.991 0.986 0.969 0.992 E/(kJ·mol-1) 85.2 88.3 304.4 277.9 68.4 38.1 A/min-1 1.71 2.49 1.19×1011 5.98×109 1.98 4.34×10-2 -
[1] 熊文慧, 张文超, 俞春培, 沈瑞琪, 程佳, 叶家海, 秦志春.多孔纳米CoFe2O4的制备及其对高氯酸铵的热分解催化性能[J].物理化学学报, 2016, 32(8):2093-2100. http://d.old.wanfangdata.com.cn/Periodical/wlhxxb201608031XIONG Wen-hui, ZHANG Wen-chao, YU Chun-pei, SHEN Rui-qi, CHENG Jia, YE Jia-hai, QIN Zhi-chun. Preparation of nanoporous CoFe2O4 and its catalytic performance during the thermal decomposition of ammonium perchlorate[J]. Acta Phys Chim Sin, 2016, 32(8):2093-2100. http://d.old.wanfangdata.com.cn/Periodical/wlhxxb201608031 [2] MAITI S, DAS D, PAL K, LORCA J, SOLER L, COLUSSI S, TROVARELLI A, PRIOLKAR K R, SARODE P R, ASAKURA K, SEIKH M M, GAYEN A. Methanol steam reforming behavior of sol-gel synthesized nanodimensional CuxFe1-xAl2O4 hercynites[J]. Appl Catal A:Gen, 2019, 570:73-83. doi: 10.1016/j.apcata.2018.11.011 [3] HOU X N, QING S J, LIU Y J, LI L D, GAO Z X, QIN Y. Enhancing effect of MgO modification of Cu-Al spinel oxide catalyst for methanol steam reforming[J]. Int J Hydrogen Energy, 2019, 45(1):477-489. http://cn.bing.com/academic/profile?id=1fc1c742bbf31b1f48746c649d745a2f&encoded=0&v=paper_preview&mkt=zh-cn [4] LIU Y J, QING S J, HOU X N, QIN F J, WANG X, GAO Z X, XIANG H W. Cu-Ni-Al spinel oxide as an efficient durable catalyst for methanol steam reforming[J]. ChemCatChem, 2018, 10(24):5698-5706. doi: 10.1002/cctc.201801472 [5] WU J C, LI Y Z, YANG Y, ZHANG Q, YUN L, WU S W, ZHOU C Y, JIANG Z K, ZHAO X J. A heterogeneous single Cu catalyst of Cu atoms confined in the spinel lattice of MgAl2O4 with good catalytic activity and stability for NO reduction by CO[J]. J Mater Chem A, 2019, 7(12):7202-7212. doi: 10.1039/C8TA11528A [6] 杜诚, 高小惠, 陈卫.铜基非贵金属氧还原电催化剂的研究进展[J].催化学报, 2016, 37(7):1049-1061. http://d.old.wanfangdata.com.cn/Periodical/cuihuaxb201607010DU Cheng, GAO Xiao-hui, CHEN Wei. Recent developments in copper-based, non-noble metal electrocatalysts for the oxygen reduction reaction[J]. Chin J Catal, 2016, 37(7):1049-1061. http://d.old.wanfangdata.com.cn/Periodical/cuihuaxb201607010 [7] LIU Q, ZHANG X X, ZHANG B, LUO Y L, CUI G W, XIE F Y, SUN X P. Ambient N2 fixation to NH3 electrocatalyzed by a spinel Fe3O4 nanorod[J]. Nanoscale, 2018, 10(30):14386-14389. doi: 10.1039/C8NR04524K [8] 崔柏, 林红, 李建保, 赵晓冲, 李文迪. ZnCo2O4纳米粒子的可见光催化性能[J].物理化学学报, 2011, 27(10):2411-2415. doi: 10.3866/PKU.WHXB20110937CUI Bai, LIN Hong, LI Jian-bao, ZHAO Xiao-chong, LI Wen-di. Visible light induced photocatalytic activity of ZnCo2O4 nanoparticles[J]. Acta Phys Chim Sin, 2011, 27(10):2411-2415. doi: 10.3866/PKU.WHXB20110937 [9] CHANDRASEKARAN S, BOWEN C, ZHANG P X, LI Z L, YUAN Q H, REN X Z, DENG L B. Spinel photocatalysts for environmental remediation, hydrogen generation, CO2 reduction and photoelectrochemical water splitting[J]. J Mater Chem A, 2018, 6(24):11078-11104. doi: 10.1039/C8TA03669A [10] TATARCHUK T, AL-NAJAR, B, BOUOUDINA M, AHMED M A A. Catalytic and Photocatalytic Properties of Oxide Spinels:In Handbook of Ecomaterials[M]. 1nd ed. Cham:Springer International Publishing, 2018. [11] 庆绍军, 侯晓宁, 刘雅杰, 王磊, 李林东, 高志贤. Cu-Ni-Al尖晶石催化甲醇水蒸气重整制氢性能的研究[J].燃料化学学报, 2018, 46(10):1210-1217. doi: 10.3969/j.issn.0253-2409.2018.10.008QING Shao-jun, HOU Xiao-ning, LIU Ya-jie, WANG Lie, LI Lin-dong, GAO Zhi-xian. Catalytic performance of Cu-Ni-Al spinel for methanol steam reforming to hydrogen[J]. J Fuel Chem Technol, 2018, 46(10):1210-1217. doi: 10.3969/j.issn.0253-2409.2018.10.008 [12] XI H J, HOU X N, LIU Y J, QING S J, GAO Z X. Cu-Al spinel oxide as an efficient catalyst for methanol steam reforming[J]. Angew Chem Int Ed Eng, 2014, 53(44):11886-11889. doi: 10.1002/anie.201405213 [13] LI G J, GU C T, ZHU W B, WANG X F, YUAN X F, CUI Z J, WANG H L, GAO Z X. Hydrogen production from methanol decomposition using Cu-Al spinel catalysts[J]. J Clean Prod, 2018, 183:415-423. doi: 10.1016/j.jclepro.2018.02.088 [14] YAHIRO H, NAKAYA K, YAMAMOTO T, SAIKI K, YAMAURA H. Effect of calcination temperature on the catalytic activity of copper supported on γ-alumina for the water-gas-shift reaction[J]. Catal Commun, 2006, 7(4):228-231. doi: 10.1016/j.catcom.2005.11.004 [15] FAUNGNAWAKIJ K, KIKUCHI R, SHIMODA N, FUKUNAGA T, EGUCHI K. Effect of thermal treatment on activity and durability of CuFe2O4-Al2O3 composite catalysts for steam reforming of dimethyl ether[J]. Angew Chem Int Ed Eng, 2008, 47(48):9314-9317. doi: 10.1002/anie.200802809 [16] SHIMIZU K, MAESHIMA H, YOSHIDA H, SATSUMA A, HATTORI T. Spectroscopic characterisation of Cu-Al2O3 catalysts for selective catalytic reduction of NO with propene[J]. Phys Chem Chem Phys, 2000, 2(10):2435-2439. doi: 10.1039/b000943l [17] MATSUKATA M, UEMIYA S, KIKUCHI E. Copper-alumina spinel catalysts for steam reforming of methanol[J]. Chem Lett, 1988, 17(5):761-764. doi: 10.1246/cl.1988.761 [18] KIM T W, SONG M W, KOH H L, KIM K L. Surface properties and reactivity of Cu/γ-Al2O3 catalysts for NO reduction by C3H6:Influences of calcination temperatures and additives[J]. Appl Catal A:Gen, 2001, 210(1/2):35-44. https://www.sciencedirect.com/science/article/abs/pii/S0926860X00008012 [19] 李光俊, 郗宏娟, 张素红, 谷传涛, 庆绍军, 侯晓宁, 高志贤.尖晶石CuM2O4(M=A1、Fe、Cr)催化甲醇重整反应的特性[J].燃料化学学报, 2012, 40(12):1466-1471. doi: 10.3969/j.issn.0253-2409.2012.12.009LI Guang-jun, XI Hong-juan, ZHANG Su-hong, GU Chuan-tao, QING Shao-jun, HOU Xiao-ning, GAO Zhi-xian. Catalytic characteristics of spinel CuM2O4(M=A1、Fe、Cr) for the stream reforming of methanol[J]. J Fuel Chem Technol, 2012, 40(12):1466-1471. doi: 10.3969/j.issn.0253-2409.2012.12.009 [20] LIU Y J, QING S J, HOU X N, QIN F J, WANG X, GAO Z X, XIANG H W. Temperature dependence of Cu-Al spinel formation and its catalytic performance in methanol steam reforming[J]. Catal Sci Technol, 2017, 7(21):5069-5078. doi: 10.1039/C7CY01236E [21] LIU Y J, QINGS J, HOU X N, FENG G, ZHANG R B, WANG X, WANG S M, GAO Z X, XIANG H W. Synthesis and structural characterization of CuAl2O4 spinel with an unusual cation distribution[J]. J Mater Appl, 2018, 7(2):82-89. [22] VLAEV T, MARKOVSKA I G, LYUBCHEV L A. Non-isothermal kinetics of pyrolysis of rice husk[J]. Thermochim Acta, 2003, 406(1/2):1-7. http://cn.bing.com/academic/profile?id=70a0ab9589ac81506e546697437180e5&encoded=0&v=paper_preview&mkt=zh-cn [23] ÓRFĀO J J M, MARTINS F G. Kinetic analysis of thermogravimetric data obtained under linear temperature programming-a method based on calculations of the temperature integral by interpolation[J]. Thermochim Acta, 2002, 390(1/2):195-211. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c6ffa69d5c744ca227b5737102afd294 [24] STROHMEIER B R, LEYDEN D E, FIELD R S, HERCULES D M. Surface spectroscopic characterization of Cu/Al2O3 catalysts[J]. J Catal, 1985, 94(2):514-530. doi: 10.1016/0021-9517(85)90216-7 [25] FURUHASHI H, INAGAKI M, NAKA S. Determination of cation distribution in spinels by X-ray diffraction method[J]. J Inorg Nucl Chem, 1973, 35(8):3009-3014. doi: 10.1016/0022-1902(73)80531-7 [26] SEO J G, YOUN M H, CHUNG J S, SONG I K. Effect of calcination temperature of mesoporous nickel-alumina catalysts on their catalytic performance in hydrogen production by steam reforming of liquefied natural gas(LNG)[J]. J Ind Eng Chem, 2010, 16(5):795-799. doi: 10.1016/j.jiec.2010.05.010 [27] 张玉红, 熊国兴, 盛世善, 刘盛林, 杨维慎. NiO/γ-Al2O3催化剂中NiO与γ-Al2O3间的相互作用[J].物理化学学报, 1999, 15(8):735-741. doi: 10.3866/PKU.WHXB19990813ZHANG Yu-hong, XIONG Guo-xing, SHENG Shi-shan, LIU Sheng-lin, YANG Wei-shen. Interaction of NiO with γ-Al2O3 supporter of NiO/γ-Al2O3 catalysts[J]. Acta Phys-Chim Sin, 1999, 15(8):735-741. doi: 10.3866/PKU.WHXB19990813 [28] GHARAGOZLOU M. Synthesis, characterization and influence of calcination temperature on magnetic properties of nanocrystalline spinel Co-ferrite prepared by polymeric precursor method[J]. J Alloys Compd, 2009, 486(1/2):660-665. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d70b46c990cb4e8277798a05afc5cbfb [29] CABO M, PELLICER E, ROSSINYOL E, CASTELL O, SURIÑACH S, BARÓ M D. Mesoporous NiCo2O4 spinel:influence of calcination temperature over phase purity and thermal stability[J]. Cryst Growth Des, 2009, 9(11):4814-4821. doi: 10.1021/cg900648q [30] 熊礼龙, 徐友龙, 张成, 陶韬. Al3+对尖晶石型LiMn2O4正极材料的表面掺杂包覆改性[J].物理化学学报, 2012, 28(5):1177-1182. doi: 10.3866/PKU.WHXB201203092XIONG L L, XU Y L, ZHANG C, TAO T. Doping-coating surface modification of spinel LiMn2O4 cathode material with Al3+ for lithium-ion batteries[J]. Acta Phys Chim Sin, 2012, 28(5):1177-1182. doi: 10.3866/PKU.WHXB201203092 [31] QING S J, HOU X N, LIU Y J, LI L D, WANG X, GAO Z X, FAN W B. Strategic use of CuAlO2 as a sustained release catalyst for production of hydrogen from methanol steam reforming[J]. Chem Commun, 2018, 54(86):12242-12245. doi: 10.1039/C8CC06600K [32] JAMES T, PADMANABHAN M, WARRIER K G K, SUGUNAN S. CuAl2O4 formation and its effect on α-Al2O3 phase evolution on calcination of metal ion doped boehmite xerogels[J]. Mater Chem Phys, 2007, 103(2/3):248-254. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a03992c064a870c2eeeb25a194220fe9 [33] KHAWAM A, FLANAGAN D R. Solid-state kinetic models:Basics and mathematical fundamentals[J]. J Phys Chem B, 2006, 110(35):17315-17328. doi: 10.1021/jp062746a [34] CANIGLIA S, BARNA G L. Handbook of Industrial Refractories Technology: Pprinciples, Types, Properties and Applications[M]. William Andrew, 1992. -





 下载:
下载: