Reaction mechanism of hydrocyanic acid with calcium oxide in sludge pyrolysis: A density functional theory study
-
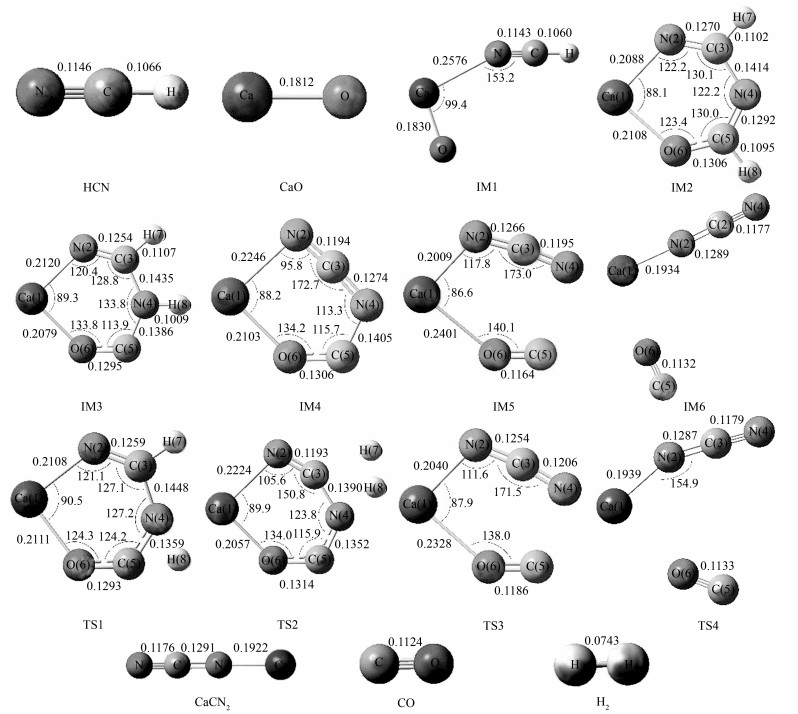
摘要: 采用密度泛函理论对污泥热解中CaO与HCN在低温段的反应进行了研究。在B3LYP/6-311++(3df,2p)水平上计算得到了反应路径上各驻点的几何构型与频率,并在此构型上使用CCSD(T)/cc-pVQZ进行单点能计算。结果表明,两个HCN分子吸附于CaO后,质子发生转移时出现反应路径中最大能垒(310.33 kJ/mol)。使用经典过渡态理论拟合了反应中各步骤的阿累尼乌斯公式,计算了三种典型温度下各步骤的反应速率,发现质子转移为该反应的决速步骤,且温度越高CaO对HCN的作用效果越好。Abstract: The reaction mechanism of CaO with HCN during low temperature sludge pyrolysis was investigated by density function theory. The geometric optimization and frequency calculations of reactants, products, intermediates and transition state in the reaction pathway were performed at B3LYP/6-311++(3df, 2p) level; single point energy calculation was performed at CCSD (T)/cc-pVQZ level and the total energy was corrected by zero-point energy at B3LYP/6-311++(3df, 2p) level. The results indicate that largest energy barrier (310.33 kJ/mol) appears in proton transition process after 2 HCN molecules are adsorbed on CaO. Arrhenius equation for each step was fitted by classical transition state theory and the reaction rate was calculated at 3 typical temperatures. The results suggest that proton transition is the rate-determining step; moreover, the promoting effect of CaO on HCN is enhanced with the increase of temperature.
-
Key words:
- calcium oxide /
- hydrocyanic acid /
- density functional theory /
- transition state theory
-
表 1 反应物与产物的特征键键长
Table 1 Bond lengths of the reactants and products
表 2 反应各驻点的总能量E、相对能量Erel及过渡态的振动虚频
Table 2 Energies and relative energies of various compounds and the imaginary frequency of transition states
Species B3LYP/6-311++G (3df, 2p) CCSD (T)/cc-pVQZ ZPE/(a.u.) E/(a.u.) Erel/(kJ·mol-1) ν/cm-1 E/(a.u.) Erel/(kJ·mol-1) CaO 0.001 8 -752.828 0 -751.888 HCN 0.016 3 -93.444 1 -93.265 6 IM1 0.019 2 -846.290 2 245 289.963 0 -845.172 3 244 819.736 0 IM2 0.044 2 -939.831 6 -302.982 7 -938.516 8 -256.248 8 IM3 0.043 4 -939.787 6 -187.460 7 -938.467 -125.498 9 IM4 0.019 4 -938.891 8 2 164.462 2 -937.290 4 2 963.664 4 IM5 0.018 4 -938.578 9 2 985.981 2 -937.295 9 2 949.224 2 IM6 0.018 0 -938.575 8 2 994.120 2 -937.296 8 2 946.861 2 TS1 0.037 1 -939.721 4 -13.652 6 -1 912.48 -938.398 6 54.085 3 TS2 0.031 0 -939.679 8 95.568 2 -2 043.50 -938.363 5 146.240 4 TS3 0.018 2 -938.578 9 2 985.981 2 -134.99 -937.293 6 2 955.262 8 TS4 0.018 0 -938.574 9 2 996.483 2 -42.07 -937.295 6 2 950.011 8 CaCN2 0.012 2 -825.216 1 -824.125 2 CO 0.005 0 -113.351 7 -113.164 4 H2 0.010 1 -1.169 9 -1.163 7 表 3 各中间反应的阿累尼乌斯公式参数
Table 3 Parameters of Arrhenius equation in the intermediate reaction
Step A ΔE/(kJ·mol-1) IM2→IM3 5.17×1013 318.65 IM3→IM4 9.56×1013 279.83 IM5→IM6 7.45×1013 6.58 the remaining steps could occur spontaneously 表 4 典型温度下的反应速率常数
Table 4 Reaction rate constants at typical temperature
Step 298 K 523 K 723 K IM2→IM3 7.21×10-43 7.72×10-19 4.92×10-10 IM3→IM4 8.48×10-36 1.07×10-14 5.79×10-7 IM5→IM6 5.24×1012 1.64×1013 2.49×1013 -
[1] KIM Y, PARKER W. A technical and economic evaluation of the pyrolysis of sewage sludge for the production of bio-oil[J]. Bioresour Technol, 2008, 99(5):1409-1416. doi: 10.1016/j.biortech.2007.01.056 [2] HANSSON K M, SAMUELSSON J, TULLIN C, AMANG L E. Formation of HNCO, HCN, and NH3 from the pyrolysis of bark and nitrogen-containing model compounds[J]. Combust Flame, 2004, 137(3):265-277. doi: 10.1016/j.combustflame.2004.01.005 [3] 郭明山, 金晶, 林郁郁, 王永贞, 侯封校.城市污泥慢速热解过程中氮的转化规律[J].化工进展, 2016, 35(1):302-307. http://www.cnki.com.cn/Article/CJFDTOTAL-HGJZ201601064.htmGUO Ming-shan, JIN Jing, LIN Yu-yu, WANG Yong-zhen, HOU Feng-xiao. Transformation mechanism of nitrogen of municipal sewage sludge in the slow pyrolysis process[J]. Chem Ind Eng Prog, 2016, 35(1):302-307. http://www.cnki.com.cn/Article/CJFDTOTAL-HGJZ201601064.htm [4] 张军.微波热解污水污泥过程中氮转化途径及调控策略[D].哈尔滨:哈尔滨工业大学, 2013. http://www.oalib.com/references/18338133ZHANG Jun. Nitrogen conversion and control of nitrogen-containing compounds during microwave pyrolysis of sewage sludge[D]. Harbin:Harbin Institute of Technology, 2013. http://www.oalib.com/references/18338133 [5] JOUNI P, HÄMÄLÄINEN, MARTTI J A. Effect of fuel composition on the conversion of volatile solid fueI-N to N2O and NO[J]. Fuel, 1994, 74(12):1922-1924. https://www.researchgate.net/publication/263601144_Effect_of_fuel_composition_on_the_conversion_of_volatile_solid_fuel-N_to_N2O_and_NO [6] 沈洪浩, 金晶, 林郁郁, 郭明山, 侯封校, 李尚. CaO对大豆蛋白热解特性及NH3等含氮化合物释放的影响[J].化工进展, 2016, 35(7):2263-2267.SHEN Hong-hao, JIN Jing, LIN Yu-yu, GUO Ming-shan, HOU Feng-xiao, LI Shang. Influence of CaO on soybean protein pyrolysis characteristics and NH3 and other nitrogenous compounds release[J]. Chem Ind Eng Prog, 2016, 35(7):2263-2267. [7] 魏砾宏, 姜秀民, 杨天华, 李延吉, 王雷.矿物成分对超细化煤粉燃烧过程中氮转化的影响[J].环境科学学报, 2006, 26(11):1780-1784. http://www.cnki.com.cn/Article/CJFDTOTAL-HJXX200611005.htmWEI Li-hong, JIANG Xiu-min, YANG Tian-hua, LI Yan-ji, WANG Lei. Influence of mineral matter on nitrogen conversion in coal during combustion[J]. Acta Energ Sin, 2006, 26(11):1780-1784. http://www.cnki.com.cn/Article/CJFDTOTAL-HJXX200611005.htm [8] WU Z, SUGIMOTO Y, KAWASHIMA H. Effect of demineralization and catalyst addition on N2 formation during coal pyrolysis and on char gasification[J]. Fuel, 2003, 82(s15-17):2057-2064. https://www.researchgate.net/publication/244067518_Effect_of_demineralization_and_catalyst_addition_on_N_2_formation_during_coal_pyrolysis_and_on_char_gasification [9] LIU X. Effects of minerals on the release of nitrogen species from anthracite[J]. Energ Source Part A, 2007, 29(4):313-327. doi: 10.1080/009083190948603 [10] 崔燕妮, 张军, 田禹.矿物质对污水污泥微波热解过程中NOx前驱物的影响研究[J].环境工程, 2012, (S2):481-485. http://www.cnki.com.cn/Article/CJFDTotal-hjgc2012s2133.htmCUI Yan-ni, ZHANG Jun, TIAN Yu. The effect of mineral matter on the formation of NOx precursors during microwave-induced pyrolysis of sewdge sludge[J]. Environ Eng, 2012, (S2):481-485. http://www.cnki.com.cn/Article/CJFDTotal-hjgc2012s2133.htm [11] TAN H, WANG X, WANG C, XU T. Characteristics of HCN removal using CaO at high temperatures[J]. Energy Fuels, 2009, 23(1):1545-1550. https://www.researchgate.net/profile/Xuebin_Wang2/publication/238176951_Characteristics_of_HCN_Removal_Using_CaO_at_High_Temperatures/links/0046352d9f68b0c177000000.pdf?inViewer=true&pdfJsDownload=true&disableCoverPage=true&origin=publication_detail [12] CAO J P, XIN H, ZHAO X Y, WEI X Y, TAKARADA T. Nitrogen transformation during gasification of livestock compost over transition metal and Ca-based catalysts[J]. Fuel, 2015, 140(3):477-483. https://www.researchgate.net/publication/267159635_Nitrogen_transformation_during_gasification_of_livestock_compost_over_transition_metal_and_Ca-based_catalysts [13] 周浩生, 陆继东, 周琥.燃煤流化床中CaO催化还原N2O机理研究[J].东南大学学报:自然科学版, 2004, 30(2):111-115.ZHOU Hao-sheng, LU Ji-dong, ZHOU Hu. Reduction of N2O over CaO under fluidized bed combustion of coal[J]. J Southeast Univ:Nat Sci Ed, 2004, 30(2):111-115. [14] GUO X, WANG L, ZHANG L, LI S, HAO J. Nitrogenous emissions from the catalytic pyrolysis of waste rigid polyurethane foam[J]. J Anal Appl Pyrolysis, 2014, 108(7):143-150. http://www.docin.com/p-1355874799.html [15] LIU H, ZHANG Q, HU H, LIU P, HU X, LI A, YAO H. Catalytic role of conditioner CaO in nitrogen transformation during sewage sludge pyrolysis[J]. Proc Combust Inst, 2014, 35(3):135-139. https://www.researchgate.net/publication/263665581_Catalytic_role_of_conditioner_CaO_in_nitrogen_transformation_during_sewage_sludge_pyrolysis [16] ZHANG Q, LIU H, LU G, YI L, HU H, CHI H, YAO H. Mechanism of conditioner CaO on NOx, precursors evolution during sludge steam gasification[J]. Proc Combust Inst, 2016, in press. https://www.researchgate.net/publication/309230273_Mechanism_of_conditioner_CaO_on_NOx_precursors_evolution_during_sludge_steam_gasification [17] 袁帅, 李军, 周志杰, 王辅臣.吡啶型氮快速热解中HCN和NH3生成机理研究[J].燃料化学学报, 2011, 39(6):413-418. http://rlhxxb.sxicc.ac.cn/CN/Y2011/V39/I06/413YUAN Shuai, LI Jun, ZHOU Zhi-jie, WANG Fu-chen. Mechanisms of HCN and NH3 formation during rapid pyrolysis of pyridinic nitrogen containing substances[J]. J Fuel Chem Technol, 2011, 39(6):413-418. http://rlhxxb.sxicc.ac.cn/CN/Y2011/V39/I06/413 [18] 袁帅, 李军, 陈雪莉, 代正华, 周志杰, 王辅臣.吡咯型氮快速热解中NH3和HCN生成机理研究[J].燃料化学学报, 2011, 39(11):801-805. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17825.shtmlYUAN Shuai, LI Jun, CHEN Xue-li, DAI Zheng-hua, ZHOU Zhi-jie, WANG Fu-chen. Study on NH3 and HCN formation mechanisms during rapid pyrolysis of pyrrolic nitrogen[J]. J Fuel Chem Technol, 2011, 39(11):801-805. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract17825.shtml [19] 刘海明, 张军营, 郑楚光, 王春梅.煤中吡啶型氮热解机理的量子化学研究[J].煤炭转化, 2004, 27(2):19-22. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH200402003.htmLIU Hai-ming, ZHANG Jun-yin, ZHANG Chu-guang, WANG Chun-mei. Quantum chemical study on the pyrolysis of pyridinic nitreogen functionalities in coal[J]. Coal Convers, 2004, 27(2):19-22. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH200402003.htm [20] 朱恒毅, 孙保民, 信晶, 尹书剑, 肖海平.含氮煤焦边缘模型氧化生成NO途径研究[J].煤炭转化, 2015, 38(1):48-52. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH201501010.htmZHU Heng-yi, SUN Bao-min, XIN Jing, YIN Shu-ping, XIAO Hai-ping. Comparision of two pathways on no desorption reaction by the edge of nitrogen-containing char[J]. Coal Convers, 2015, 38(1):48-52. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH201501010.htm [21] ZHANG X, ZHOU Z, ZHOU J, LIU J, CEN K. Density functional study of NO desorption from oxidation of nitrogen containing char by O2[J]. Combust Sci Technol, 2012, 184(4):445-455. doi: 10.1080/00102202.2011.648031 [22] 温正城, 王智化, 周俊虎, 周志军, 刘建忠, 岑可法.金属钙对煤焦异相还原NO催化机理的量子化学研究[J].燃烧科学与技术, 2009, 15(6):505-510. http://www.cnki.com.cn/Article/CJFDTOTAL-RSKX200906006.htmWEN Zheng-cheng, WANG Zhi-hua, ZHOU Jun-hu, ZHOU Zhi-jun, LIU Jian-zhong, CEN Ke-fa. Quantum chemistry study on catalytic mechanism of Ca on NO-Char heterogeneous reaction[J]. J Combust Sci Technol, 2009, 15(6):505-510. http://www.cnki.com.cn/Article/CJFDTOTAL-RSKX200906006.htm [23] LIU L, JIN J, LIN Y, HOU F, LI S. The effect of calcium on nitric oxide heterogeneous adsorption on carbon:A first-principles study[J]. Energy, 2016, 106:212-220. doi: 10.1016/j.energy.2016.02.148 [24] BOZOVIC A D, ZHAO X, BOHME D K. Exploration of the catalytic oxidation of ethylene with N2O mediated by atomic alkaline-earth metal cations[J]. Int J Mass Spectrom, 2006, 254(3):155-162. doi: 10.1016/j.ijms.2006.05.002 [25] VALENTIN C D, FIGINI A, PACCHIONI G. Adsorption of NO and NO2 on terrace and step sites and on oxygen vacancies of the CaO (100) surface[J]. Surf Sci, 2004, 556(s2/3):145-158. https://www.researchgate.net/publication/256857602_Adsorption_of_NO_and_NO2_on_terrace_and_step_sites_and_on_oxygen_vacancies_of_the_CaO1_0_0_surface [26] PISKORZ W, ZASADA F, STELMACHOWSKI P, KOTARBA A, SOJKA Z. DFT modeling of reaction mechanism and ab Initio microkinetics of catalytic N2O decomposition over alkaline earth oxides:from molecular orbital picture account to simulation of transient and stationary rate profiles[J]. J Phys Chem C, 2013, 117(36):18488-18501. doi: 10.1021/jp405459g [27] NOWIAK G, SKURSKI P, ANUSIEWICZ L. Attaching an alkali metal atom to an alkaline earth metal oxide (BeO, MgO or CaO) yields a triatomic metal oxide with reduced ionization potential and redirected polarity[J]. J Mol Model, 2016, 22(4):1-8. https://www.researchgate.net/profile/Iwona_Anusiewicz/publication/298897702_Attaching_an_alkali_metal_atom_to_an_alkaline_earth_metal_oxide_BeO_MgO_or_CaO_yields_a_triatomic_metal_oxide_with_reduced_ionization_potential_and_redirected_polarity/links/56f1186a08ae0dcdafd6e0be.pdf?origin=publication_detail [28] BUTLER G B, BERLIN K D. Fundamentals of Organic Chemistry:Theory and Application[M]. NewYork:Ronald Press Co, 1972. [29] YANG X, ZHAO B, ZHUO Y, GAO Y, CHEN C, XU X. DRIFTS study of ammonia activation over CaO and sulfated CaO for NO reduction by NH3[J]. Environ Sci Technol, 2011, 45(3):1147-51. doi: 10.1021/es103075p [30] FU S L, SONG Q, TANG J S, YAO Q. Effect of CaO on the selective non-catalytic reduction deNOx, process:Experimental and kinetic study[J]. Chem Eng J, 2014, 249(4):252-259. https://www.researchgate.net/publication/261764920_Effect_of_CaO_on_the_selective_non-catalytic_reduction_deNOx_process_Experimental_and_kinetic_study [31] 傅献彩, 沈文霞, 姚天扬, 侯文华.物理化学[M].北京:高等教育出版社, 2006.FU Cai-xia, SHEN Wen-xia, YAO Tian-yang, HOU Wen-hua. Physical Chemistry[M]. Beijing:Higher Education Press, 2006. -





 下载:
下载: