Effect of Mg modification on the catalytic performance of Co/γ-Al2O3-TiO2 in the combustion of propane
-
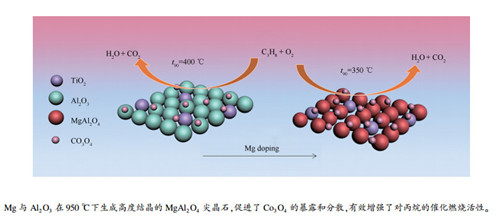
摘要: 采用分步浸渍法制备了系列Mg改性的Co/γ-Al2O3-TiO2催化剂,通过X射线衍射(XRD)、紫外可见漫反射光谱(DR-UV-vis)、N2吸附-脱附(BET)、X射线光电子能谱(XPS)和H2程序升温还原(H2-TPR)等技术对催化剂进行表征,并考察了其对丙烷燃烧的催化性能。结果表明,Co在原始γ-Al2O3-TiO2载体和Mg改性MgO/γ-Al2O3-TiO2载体上均以Co3O4的形式存在;Mg掺入后与Al2O3作用形成MgAl2O4尖晶石,改善了载体的织构性质,提升了Co3O4在催化剂载体表面的暴露数量和分散程度。此外,MgAl2O4与Co3O4相互作用提升了Co3O4颗粒表面Co3+/Co2+和Oads/Olatt的比例,并削弱了Co-O键键能,从而提升了其对丙烷的催化燃烧活性。当Mg负载量为15%(质量分数)时,在Co/MgO(15%)/γ-Al2O3-TiO2催化剂上进行丙烷燃烧,丙烷90%转化率的温度比无Mg掺杂的Co/γ-Al2O3-TiO2催化剂的降低了45℃,并且连续反应40 h其活性保持稳定。
-
关键词:
- Co3O4 /
- MgAl2O4尖晶石 /
- 催化燃烧 /
- 丙烷 /
- 相互作用
Abstract: A series of Mg-modified Co/MgO/γ-Al2O3-TiO2 catalysts were prepared by multi-step impregnation method and characterized by X-ray diffraction (XRD), diffuse reflectance visible ultra violet spectroscopy (DR-UV-vis), N2 adsorption-desorption, X-ray photoelectron spectroscopy (XPS) and H2 temperature-programmed reduction (H2-TPR); the effect of Mg modification on the catalytic performance of Co/MgO/γ-Al2O3-TiO2 in the combustion of propane was investigated. The results indicate that Co exists in the form of Co3O4 on both the pristine γ-Al2O3-TiO2 and Mg-modified MgO/γ-Al2O3-TiO2 supports; Mg added in the MgO/γ-Al2O3-TiO2 support interacts with Al2O3, forming MgAl2O4 spinel, which can improve the textural properties and promote the dispersion of Co3O4. Moreover, the interaction between MgAl2O4 and Co3O4 can increase the proportions of Co3+/Co2+ and Oads/Olatt on the catalyst surface, weaken the Co-O bond, and thereby enhance the activity of Co-based catalyst in propane combustion. For the propane combustion over the Co/MgO(15%)/γ-Al2O3-TiO2 catalyst with an Mg loading of 15%, the temperature to achieve a propane conversion of 90% is decreased by 45℃ in comparison with that over the Mg-free Co/γ-Al2O3-TiO2 catalyst; moreover, the Co/MgO(15%)/γ-Al2O3-TiO2 catalyst exhibits excellent stability and no decrease in the activity is observed in a 40 h continuous reaction test for propane combustion.-
Key words:
- Co3O4 /
- MgAl2O4 spinel /
- catalytic combustion /
- propane /
- interaction
-
图 1 不同催化剂催化燃烧丙烷活性; (b)lnr与1/RT关系图; (c) Co/Mg(15%)/AlTi在不同GHSV下催化燃烧丙烷活性; (d)Co/Mg(15%)/AlTi的稳定性评价
Figure 1 (a) Activity of various catalysts in propane combustion; (b) Arrhenius plots of lnr and 1/RT for the Co/AlTi and Co/Mg(15)/AlTi catalysts; (c) catalytic activity of Co/Mg(15%)/AlTi in propane combustion under different GHSVs; (d) long-term stability test of Co/Mg(15%)/AlTi. The feed consisted of 0.3% (volume ratio) propane and balanced air, with a gas hourly space velocity (GHSV) of 15000mL/(h·g) in the graphs of (a), (b) and (d)
表 1 催化剂与载体的织构性质
Table 1 Texture properties of catalysts and support
Sample Surface area A/(m2·g-1) Pore volume v/(cm3·g-1) Pore size d/nm AlTi 68.6 0.239 13.1 Mg(15%)/AlTi 73.3 0.225 8.4 Co/AlTi 61.6 0.216 13.1 Co/Mg(15%)/AlTi 69.3 0.229 8.3 表 2 催化剂的XPS参数
Table 2 XPS results of various catalysts
Catalyst Element contents wmol/% Binding energy E/eV Co2+ Co3+ Al Mg Co O Co3+/Co2+ Oads/Olatt Mg(15%)/AlTi 20.42 2.81 0.00 76.77 - - - 1.59 Co/AlTi 22.61 0.00 1.88 75.51 782.4 780.2 0.71 1.46 Co/Mg(15%)/AlTi 18.94 2.71 2.43 75.92 781.9 779.9 0.77 1.65 -
[1] HE C, CHENG J, ZHANG X, DOUTHWAITE M, PATTISSON S, HAO Z. Recent advances in the catalytic oxidation of volatile organic compounds:A review based on pollutant sorts and sources[J]. Chem Rev, 2019, 119(7):4471-4568. doi: 10.1021/acs.chemrev.8b00408 [2] KAMAL M S, RAZZAK S A, HOSSAIN M M. Catalytic oxidation of volatile organic compounds (VOCs) A review[J]. Atmos Environ, 2016, 140:117-134. https://www.sciencedirect.com/science/article/abs/pii/S1352231016303727 [3] LIU Y R, LI X, LIAO W M, JIA A P, WANG Y J, LUO M F, LU J Q. Highly active Pt/BN catalysts for propane combustion:The roles of support and reactant-induced evolution of active Sites[J]. ACS Catal, 2019, 9(2):1472-1481. doi: 10.1021/acscatal.8b03666 [4] RAO C, PENG C, PENG H, ZHANG L, LIU W, WANG X, ZHANG N, WU P. In situ embedded pseudo Pd-Sn solid solution in micropores silica with remarkable catalytic performance for CO and propane oxidation[J]. ACS Appl Mater Int, 2018, 10(11):9220-9224. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=bc4de0292cac38baf5451a8bcd341643 [5] LEDWA K A, PAWLYTA M, KĘPIHSKI L. RuxCe1-xO2-y nanoparticles deposited on functionalized γ-Al2O3 as a thermally stable oxidation catalyst[J]. Appl Catal B:Environ, 2018, 230:135-144. [6] HU Z, WANG Z, GUO Y, WANG L, GUO Y, ZHANG J, ZHAN W. Total oxidation of propane over a Ru/CeO2 catalyst at low temperature[J]. Environ Sci Technol, 2018, 52(16):9531-9541. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=090b21abd03915a81991d1ffdd69d1a7 [7] PIUMETTI M, FINO D, RUSSO N. Mesoporous manganese oxides prepared by solution combustion synthesis as catalysts for the total oxidation of VOCs[J]. Appl Catal B:Environ, 2015, 163:277-287. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=b599c4d8d73da95bd10d400deb80b03a [8] CHENG L, WANG J, ZHANG C, JIN B, MEN Y. Boosting acetone oxidation efficiency over MnO2 nanorods by tailoring crystal phases[J]. New J Chem, 2019, 43(48):19126-19136. https://pubs.rsc.org/en/content/articlelanding/2019/nj/c9nj04192c#! [9] ZHAO J, TANG Z, DONG F, ZHANG J. Controlled porous hollow Co3O4 polyhedral nanocages derived from metal-organic frameworks (MOFs) for toluene catalytic oxidation[J]. Mol Catal, 2019, 463:77-86. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=8eb98c90a445df03ab186178ffe94f31 [10] TANG W, XIAO W, WANG S, REN Z, DING J, GAO P-X. Boosting catalytic propane oxidation over PGM-free Co3O4 nanocrystal aggregates through chemical leaching:A comparative study with Pt and Pd based catalysts[J]. Appl Catal B:Environ, 2018, 226:585-595. [11] TSONCHEVA T, ISSA G, NIETO J M L, BLASCO T, CONCEPCION P, DIMITROV M, ATANASOVA G, KOVACHEVA D. Pore topology control of supported on mesoporous silicas copper and cerium oxide catalysts for ethyl acetate oxidation[J]. Microporous Mesoporous Mater, 2013, 180:156-161. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=b5f4e19472449ad568a90131cb95fdef [12] ZHANG X, JUNHUI Y, JING Y, TING C, BEI X, ZHE L, KUNFENG Z, LING Y, DANNONG H. Excellent low-temperature catalytic performance of nanosheet Co-Mn oxides for total benzene oxidation[J]. Appl Catal A:Gen, 2018, 566:104-112. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=4472d7e389d68272bd24a64d2d44aaf0 [13] CAI T, YUAN J, ZHANG L, YANG L, TONG Q, GE M, XIAO B, ZHANG X, ZHAO K, HE D. Ni-Co-O solid solution dispersed nanocrystalline Co3O4 as a highly active catalyst for low-temperature propane combustion[J]. Catal Sci Technol, 2018, 8(21):5416-5427. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2e638b85a8a920b4b99accc84bac1d66 [14] HU Z, QIU S, YOU Y, GUO Y, GUO Y, WANG L, ZHAN W, LU G. Hydrothermal synthesis of NiCeOx nanosheets and its application to the total oxidation of propane[J]. Appl Catal B:Environ, 2018, 225:110-120. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=1315ad9ad425763b68182a61d209a7a0 [15] BENJAMIN F F, ALPHONSE P. Co-Mn-oxide spinel catalysts for CO and propane oxidation at mild temperature[J]. Appl Catal B:Environ, 2016, 180:715-725. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=c27b5d3ea1972617250453ff4388780f [16] SOLSONA B, DAVIES T E, GARCIA T, V ZQUEZ I, DEJOZ A, TAYLOR S H. Total oxidation of propane using nanocrystalline cobalt oxide and supported cobalt oxide catalysts[J]. Appl Catal B:Environ, 2008, 84(1/2):176-184. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=838ae912f19e4fe439c4d7b0d219600b [17] HU W, LI G, CHEN J, HUANG F, GONG M, ZHONG L, CHEN Y. Enhancement of activity and hydrothermal stability of Pd/ZrO2-Al2O3 doped by Mg for methane combustion under lean conditions[J]. Fuel, 2017, 194:368-374. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2ae1502408dc9779d4f275fd563a3575 [18] 袁善良, 兰海, 薄其飞, 张彪, 肖熙, 蒋毅. TiO2掺杂CuMnCe/Al2O3催化剂对甲烷催化燃烧脱氧反应的影响[J].燃料化学学报, 2017, 45(2):243-248. http://manu60.magtech.com.cn/rlhxxb/fileup/HTML/20170215.htmlYUAN Shan-liang, LAN Hai, BO Qi-fei, ZHANG Biao, XIAO Xi, JIANG Yi. Effect of TiO2 doping on methane catalytic combustion deoxidation of CuMnCe/Al2O3 catalyst[J]. J Fuel Chem Technol, 2017, 45(2):243-248. http://manu60.magtech.com.cn/rlhxxb/fileup/HTML/20170215.html [19] XIE Y, GUO Y, GUO Y, WANG L, ZHAN W, WANG Y, GONG X-Q, LU G. A highly-efficient La-MnOx catalyst for propane combustion:The promotional role of La and the effect of the preparation method[J]. Catal Sci Technol, 2016, 6(23):8222-8233. https://pubs.rsc.org/-/content/articlelanding/2016/cy/c6cy01663d/unauth [20] 张磊, 周福勋, 赵建涛, 吴志伟, 王建国, 房倚天.铜基催化剂上甲烷催化燃烧反应动力学特性研究[J].燃料化学学报, 2014, 42(9):1140-1145. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18495.shtmlZHANG Lei, ZHOU Fu-xun, ZHAO Jian-tao, WU Zhi-wei, WANG Jian-guo, FANG Yi-tian. Reaction kinetics of methane combustion on copper-based catalyst[J]. J Fuel Chem Technol, 2014, 42(9):1140-1145. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18495.shtml [21] YU J, LIU Z, ZHANG H, HUANG T, HAN J, ZHANG Y, CHONG D. Synergistic effect of N-and F-codoping on the structure and photocatalytic performance of TiO2[J]. J Environ Sci (China), 2015, 28:148-156. https://www.sciencedirect.com/science/article/abs/pii/S100107421400271X [22] LOJACONO M, VERBEEK J L, SCHUIT G C A. Magnetic and spectroscopic investigations on cobalt-alumina and cobalt-molybdenum-alumina:Electron spin resonance of the oxidized, sulfided, and reduced catalysts[J]. J Catal, 1973, 29(3):463-474. https://www.sciencedirect.com/science/article/pii/0021951773902534 [23] BAI B, LI J. Positive effects of K+ ions on three-dimensional mesoporous Ag/Co3O4 catalyst for HCHO oxidation[J]. Acs Catal, 2014, 4(8):2753-2762. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f2d273a9da4d2a6097cf24522c445b6c [24] GARCIA T, AGOURAM S, SÁNCHEZ-ROYO J F, MURILLO R, MASTRAL A M, ARANDA A, VÁZQUEZ I, DEJOZ A, SOLSONA B. Deep oxidation of volatile organic compounds using ordered cobalt oxides prepared by a nanocasting route[J]. Appl Catal A:Gen, 2010, 386(1/2):16-27. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=8fa1c2da7f81c02ef80ed6187bb40ddf [25] MARCO J, GANCEDO J, GRACIA M, GAUTIER J, RIOS E, BERRY F. Characterization of the nickel cobaltite, NiCo2O4, prepared by several methods:An XRD, XANES, EXAFS, and XPS study[J]. J Solid State Chem, 2000, 153(1):74-81. https://www.sciencedirect.com/science/article/abs/pii/S0022459600987498 [26] HUANG J, DING T, MA K, CAI J, SUN Z, TIAN Y, JIANG Z, ZHANG J, ZHENG L, LI X. Modification of Cu/SiO2 catalysts by La2O3 to quantitatively tune Cu+-Cu0 dual sites with improved catalytic activities and stabilities for dimethyl ether steam reforming[J]. ChemCatChem, 2018, 10(17):3862-3871. https://www.researchgate.net/publication/325900895_Modification_of_CuSiO_Catalysts_by_LaO_to_Quantitatively_Tune_Cu-Cu_Dual_Sites_with_Improved_Catalytic_Activities_and_Stabilities_for_Dimethyl_Ether_Steam_Reforming [27] LI X, LI X, ZENG X, ZHU T. Correlation between the physicochemical properties and catalytic performances of micro/mesoporous CoCeO mixed oxides for propane combustion[J]. Appl Catal A:Gen, 2019, 572:61-70. https://www.onacademic.com/detail/journal_1000041581162399_074e.html [28] ROUSSEAU S, LORIDANT S, DELICHERE P, BOREAVE A, DELOUME J, VERNOUX P. La(1-x)SrxCo1-yFeyO3 perovskites prepared by sol-gel method:Characterization and relationships with catalytic properties for total oxidation of toluene[J]. Appl Catal B:Environ, 2009, 88(3/4):438-447. https://www.sciencedirect.com/science/article/abs/pii/S0926337308004025 [29] CHEN Z, WANG S, LIU W, GAO X, GAO D, WANG M, WANG S. Morphology-dependent performance of Co3O4 via facile and controllable synthesis for methane combustion[J]. Appl Catal A:Gen, 2016, 525:94-102. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=426bb4318551336a31370d383748d092 [30] 黄波, 鲁旖, 江志东.球磨法制备NiO/MgAl2O4催化剂用于低浓度甲烷催化燃烧[J].精细化工, 2020, 37(6):1220-1226+1248. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jxhg202006019HUANG Bo, LU Yi, JIANG Zhi-dong. Preparation of NiO/MgAl2O4 catalysts by ball-milling method for catalytic lean burn of high-diluted methane[J]. Fine Chem, 2020, 37(6):1220-1226+1248. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=jxhg202006019 [31] ZHANG W, WU F, LI J, YOU Z. Dispersion-precipitation synthesis of highly active nanosized Co3O4 for catalytic oxidation of carbon monoxide and propane[J]. Appl Surf Sci, 2017, 411:136-143. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=ce69fcef0071461027283ca223b1d538 [32] LIU Q, WANG L C, CHEN M, CAO Y, HE H Y, FAN K N. Dry citrate-precursor synthesized nanocrystalline cobalt oxide as highly active catalyst for total oxidation of propane[J]. J Catal, 2009, 263(1):104-113. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=180485a16fa72ff87700fed9b9e5eacf [33] 牛汝月, 刘鹏程, 李威, 王爽, 李晋平.柱状Co3O4催化剂的乏风催化燃烧性能[J].无机化学学报, 2018, 34(10):1929-1935. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=wjhxxb201810020NIU Ru-yue, LIU Peng-cheng, LI Wei, WANG Shuang, LI Jin-ping. Catalytic combustion of ventilation air methane over Co3O4 rectangular prism[J]. Chin J Inorg Chem, 2018, 34(10):1929-1935. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=wjhxxb201810020 -





 下载:
下载: