Preparation of nano-scale nickel-tungsten catalysts by pH value control and application in hydrogenolysis of cellulose to polyols
-
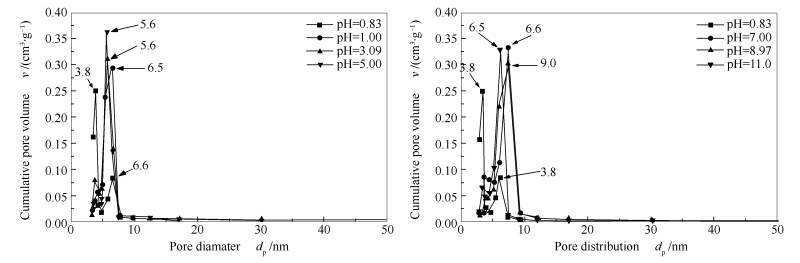
摘要: 采用等体积浸渍法制备了双金属Ni-W催化剂,并研究了制备过程中前驱体溶液pH值对活性的影响。柠檬酸和氢氧化钾分别作为酸碱调节剂将前驱体溶液pH值调节至0.83、1.00、3.09、5.00、7.03、8.97和11.0。纤维素氢解反应结果表明,当前驱体溶液的pH值为1.00时,在氢压为5.0 MPa、温度为518 K的反应条件下,得到低碳(C2,3)多元醇的总收率为74.5%。此外,通过BET和SEM表征了催化剂的物理性质。其比表面积为330-450 m2/g,且在高温煅烧下SBA-15仍保持良好的水热稳定性。TEM和SEM-EDX结果显示,催化剂粒子在载体表面分散性较好,但也有少量的聚集现象。XRD表征表明,NiO等物种的还原程度明显受到pH值的影响,且镍、钨物种的相态也存在着显著不同。Abstract: The Ni-W/SBA-15 catalysts prepared by incipient-wetness impregnation method and controlled by the pH value were developed. Potassium hydroxide and citric acid regarded as the pH value regulators were added to the solution before impregnation to provide an acidic or basic condition at different pH values (0.83, 1.00, 3.09, 5.00, 7.03, 8.97 and 11.0). The 74.5% yield of total low carbon (C2, 3) polyols including ethylene glycol (EG), 1, 2-propylene glycol (1, 2-PG) and glycerol (Gly) was obtained over 10%Ni-20%W/SBA-15 catalyst prepared at pH value of 1.00 and 518 K under H2 pressure of 5.0 MPa. Furthermore, the physical properties of this series of nickel-tungsten catalysts were characterized by BET and SEM. The results demonstrated that the catalysts showed excellent thermal stability and the surface area was mainly in range of 330-450 m2/g. The particles had a good dispersion on the surface of SBA-15 but some aggregations were existed which could be characterized by TEM and SEM-EDX. However, the reduction of metallic oxides especially for NiO was obviously influenced by pH value control and some metallic species characterized by XRD. According to XRD results, the impregnation pH value influenced the reduction of NiO and the phase states of nickel and tungsten species.
-
Key words:
- cellulose /
- hydrogenolysis /
- pH value control /
- nickel-tungsten catalysts /
- polyols
-
Table 1 Physical properties of 10%Ni-20%W/SBA-15 catalysts prepared in diffirent pH values

Table 2 Liquid production for cellulose hydrogenolysis over Ni-W/SBA-15 catalystsa

-
[1] JUBEN N C, GEORGE W H, JAMES A D. Liquid-phase catalytic processing of biomass-derived oxygenated hydrocarbon to fuels and chemicals[J]. Angew Chem Int Ed, 2007, 46(38):7164-7183. doi: 10.1002/(ISSN)1521-3773 [2] YABUSHITA M, KOBAYASHI H, FUKUOKA A. Catalytic transformation into platform chemicals[J]. Appl Catal B:Environ, 2014, 145(1):1-9. http://www.sciencedirect.com/science/article/pii/S0926337313000726 [3] SCHWARTZ T J, O'NEILL B J, SHANKS B H, DUMESIC J A. Bridging the chemical and biological catalysis gap:Challenges and outlook for producing sustainable chemicals[J]. ChemInform, 2014, 45(31):2060-2069. doi: 10.1021/cs500364y?src=recsys&journalCode=accacs [4] WANGA Q, ZHANG T. One-pot conversion of cellulose to ethylene glycol with multi-functional tungsten-based catalysts[J]. Accounts Chem Res, 2013, 46(7):1377-1386. doi: 10.1021/ar3002156 [5] JI N, ZHENG M Y, WANG A Q, ZHANG T, CHEN J G. Ni-promoted tungsten carbide for cellulose conversion:Effect of preparation methods[J]. ChemSusChem, 2012, 5(5):939-944. doi: 10.1002/cssc.201100575 [6] PANG J F, ZHENG M Y, WANG A Q, ZHANG T. Catalytic hydrogenation of corn stalk to ethylene glycol and 1, 2-propylene glycol[J]. Ind Eng Chem Res, 2011, 50(11):6601-6608. doi: 10.1021/ie102505y [7] ABDEL-AZIM S M, ABOUL-GHEIT A K, AHMED S M, EL-DSOUKI D S, ABDEL-MOTTALEB M S. Preparation and application of mesoporous nanotitania photocatalysts using different templates and pH media[J]. Int J Photoenergy, 2014, 2014(5):1-6. https://www.hindawi.com/journals/ijp/2014/687597/ [8] BAE J W, LEE Y J, PARK J Y, JUN K W. Influence of pH of the impregnation solution on the catalytic properties of Co/γ-alumina for Fischer-Tropsch synthesis[J]. Energy Fuels, 2008, 22(5):2885-2891. doi: 10.1021/ef800155v [9] HUANG Y P, DONG X Q, YU Y Z. The influence of surface xygen and hydroxyl groups on the dehydrogenation of ethylene, acetic acid and hydrogenated vinyl acetate on pure Pd(100):A DET study[J]. Appl Surf Sci, 2016, 388(12):455-460. http://www.sciencedirect.com/science/article/pii/S0169433215029311 [10] LI G N, PIDKO E A, HENSEN J M. A periodic DFT study of glucose to fructose isomerization on tungstite (WO3 center dot H2O):Influence of group Ⅳ-Ⅳ dopants and cooperativity with hydroxyl groups[J]. ACS Catal, 2016, 6(7):4162-4169. doi: 10.1021/acscatal.6b00869 [11] ENGELBREKT C, MALCHO P, ANDERSEN J, ZHANG L J, STAHL K, LI B, HU J, ZHANG J D. Selective synthesis of clinoatacammite Cu2(OH)3Cl and tenorite CuO nanoparticle by pH control[J]. J Nanopart Res, 2014, 16(8):2562-2574. doi: 10.1007/s11051-014-2562-4 [12] LIU Q Y, LIAO Y H, WANG T J, CAI C L, ZHANG Q, TSUBAKI N, MA L L. One-pot transformation of cellulose to sugar alcohols over acidic metal phosphates combined with Ru/C[J]. Ind Eng Chem Res, 2014, 53(32):12655-12664. doi: 10.1021/ie5016238 [13] SUN R Y, ZHENG M Y, PANG J F, LIU X, WANG J H, PAN X L, WANG A Q, WANG X D, ZHANG T. Selectivity-switchable conversion of cellulose to glycols over Ni-Sn catalysts[J]. ACS Catal, 2016, 6(1):191-201. doi: 10.1021/acscatal.5b01807 [14] XIAO Z Q, GE Q W, XING C, JIANG C J, FANG S, JI J B, MAO J W. Self-reducing bi-functional Ni-W/SBA-15 catalyst for cellulose hydrogenolysis to low carbon polyols[J]. J Energy Chem, 2015, 25(3):434-444. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=trqz201603014&dbname=CJFD&dbcode=CJFQ [15] KOSMULSKI M. The pH-dependent surface charging and points of zero charge[J]. J Colloid Interf Sci, 2011, 353(1):1-5. doi: 10.1016/j.jcis.2010.08.023 [16] NGUYEN VAN T D, SUFIAN S, MANSOR N, YAHYA N. Characterization of carbon nanofibers treated with thermal nitrogen as a catalyst support using point of zero charge analysis[J]. J Nanomater, 2014, 2014(12):1-6. http://dl.acm.org/citation.cfm?id=2696444.2696445 [17] LI H, FANG Z, LUO J, YANG S. Direct conversion of biomass components to the bio-fuel methyl levulinate catalyzed by acid-base bi-functional zirconia-zeolites[J]. Appl Catal B:Environ, 2017, 200:182-191. doi: 10.1016/j.apcatb.2016.07.007 [18] ZHU S H, GAO X Q, ZHU Y L, ZHU Y F, ZHENG H Y, LI Y G. Promoting effect of boron oxide on Cu/SiO2 catalyst for glycerol hydrogenolysis to 1, 2-peopanediol[J]. J Catal, 2013, 303(7):70-79. http://www.sciencedirect.com/science/article/pii/S0021951713001036 [19] FABICOVICOVA K, LUCAS M, CLAUS P. From microcrystalline cellulose to hard-and softwood-based feedstocks:Their hydrogenolysis to polyols over a highly efficient ruthenium-tungsten catalyst[J]. Green Chem, 2015, 17(5):3075-3083. doi: 10.1039/C5GC00421G [20] PAN G Y, MA Y L, MA X X, SUN Y G, LV J M, ZHANG J L. Catalytic hydrogenation of corn stalk into polyols over Ni-W/MCM-41 catalyst[J]. Chem Eng J, 2016, 299(1):386-392. http://www.sciencedirect.com/science/article/pii/S1385894716305241 [21] SUAREZ-TORILLO V A, SANTOLALLA-VARGAS C E, DE LOS REYES J A, VAZQUEZ-ZAVALA A, VRINAT M, GEANTET C. Influence of the solution pH in impregnation with citric acid and activity of Ni/W/Al2O3 catalysts[J]. J Mol Catal A:Chem, 2015, 404-405(s):36-46. http://www.sciencedirect.com/science/article/pii/S1381116915001442 [22] LI Y P, LIAO Y H, CAO X F, WANG T J, MA L L, LONG J X, LIU Q Y, XUA Y. Advances in hexitol and ethylene glycol production by one-pot hydrolytic hydrogenation and hydrogenolysis of cellulose[J]. Bio Bioenergy, 2015, 74(1):148-161. http://www.cabdirect.org/abstracts/20153145540.html [23] LEE S, ZHANG Z T, WANG X M, PFEFFERLE L D, HALLER G L. Characterization of multi-walled carbon nanotubes catalyst supports by point of zero charge[J]. Catal Today, 2011, 164(1):68-73. doi: 10.1016/j.cattod.2010.10.031 [24] CAO Y L, WANG J W, KANG M Q, ZHU Y L. Efficient synthesis of ethylene glycol from cellulose over Ni-WO3/SBA-15 catalyst[J]. J Mol Catal A Chem, 2014, 381:46-53. doi: 10.1016/j.molcata.2013.10.002 [25] ZHENG M Y, WANG A Q, JI N, PANG J F, WANG X D, ZHANG T. Transition metal-tungsten bimetallic catalysts for the conversion of cellulose into ethylene glycol[J]. ChemSusChem, 2010, 3(1):63-66. doi: 10.1002/cssc.v3:1 [26] SUN R Y, ZHENG M Y, PANG J F, LIU X, WANG J H, PAN X L, WANG A Q, WANG X D, ZHANG T. Selectivity-switchable conversion of cellulose yo glycols over Ni-Sn catalysts[J]. ACS Catal, 2016, 6(1):191-201. doi: 10.1021/acscatal.5b01807 -





 下载:
下载: