Low-cost preparation of carbon-supported cobalt catalysts from MOFs and their performance in CO hydrogenation
-
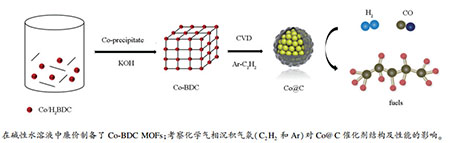
摘要: 以对苯二甲酸(H2BDC)为配体、乙酸钴为Co源、水作溶剂,通过共沉淀法合成了金属有机框架材料(Co-BDC MOFs);以其为前驱体分别在乙炔和氩气氛下采用化学气相沉积法制备了核壳结构Co@C催化剂。结合XRD、氮吸附、SEM、TEM、XPS、TGA和Raman光谱等手段对Co@C催化剂的结构和组成进行了表征,考察了该催化剂在费托合成反应中的活性及稳定性。结果表明,炭化气氛对炭层结构的石墨化程度有较大影响,而对金属Co核的物相结构和粒径影响较小;乙炔气氛有助于形成多孔的石墨炭壳,从而促进烃链的生长,Co@C-C2H2催化剂上的C5+烃产物选择性高达82.66%,反应过程中催化剂物相由单相金属Co转变为金属Co与Co2C的混合相,且无失活现象发生,表明Co2C具有较高的费托反应催化活性。Abstract: With terephthalic acid (H2BDC) as ligand and cobalt acetate as Co source, metal-organic frameworks (Co-BDC MOFs) were synthesized in water by co-precipitation; after that, core-shell Co@C catalysts were prepared by chemical vapor deposition (CVD) of Co-BDC MOFs in acetylene and Ar atmosphere. The structure, composition and properties of Co@C catalysts were characterized by XRD, nitrogen physisorption, SEM, TEM, XPS, TGA and Raman spectroscopy and their catalytic performance in Fischer-Tropsch synthesis (FTS) were investigated in a fixed-bed tubular reactor. The results demonstrated that the carbonization atmosphere has an important influence on the graphitization degree of carbon shell, whereas has little effect on the phase and size of Co core. The pore of graphite shell is significantly improved by CVD in acetylene, which can enhance the selectivity to heavier hydrocarbons (C5+) for CO hydrogenation; in particular, the Co@C-C2H2 catalyst shows a high selectivity of 82.66% to the C5+ hydrocarbons. As the carbon shell can effectively inhibit the cobalt nanoparticles from migration and agglomeration during the FTS reaction, the Co species were well distributed in both the fresh and spent catalysts and no significant sintering and deactivation are observed for the Co@C catalysts upon the FTS tests. During the FTS reaction, the active phase changes from metallic Co to a mixture of metallic Co and Co2C, whilst the catalytic activity of Co@C-C2H2 keeps almost unchanged, suggesting that Co2C may also be an active phase for the Fischer-Tropsch synthesis.
-
Key words:
- Co-based catalyst /
- metal-organic frameworks /
- Fischer-Tropsch synthesis /
- carbon support /
- Co@C /
- Co2C
1) 本文的英文电子版由Elsevier出版社在ScienceDirect上出版(http://www.sciencedirect.com/science/journal/18725813). -
图 3 不同pH值(a)、不同反应物配比(b)、不同温度(c)下合成样品的XRD谱图
Figure 3 XRD patterns of various Co-BDC MOFs samples synthesized under different pH values
((a)other synthesis condition: 70 ℃, H2BDC :Co = 2), synthesis mixture compositions ((b)other synthesis condition: 70 ℃, pH = 6), and temperatures ((c)other synthesis condition: pH = 6, H2BDC :Co =2)
表 1 不同条件合成的Co-BDC MOFs的织构性质
Table 1 Textural properties of Co-BDC MOFs synthesized under different conditions
Reaction condition ABET /(m2·g-1) vtotal/(cm3·g-1) Pore size d/nm pH=5a 83 0.37 17.7 pH=6a 17 0.09 20.3 pH=7a 27 0.11 16.2 70 ℃b 83 0.37 17.7 80 ℃ b 90 0.37 16.3 90 ℃b 48 0.21 18.6 H2BDC:Coc=3:1 44 0.22 19.7 H2BDC:Coc=2:1 17 0.09 20.3 H2BDC:Coc=1:1 66 0.42 25.4 H2BDC:Coc=1:2 42 0.23 21.6 H2BDC:Coc=1:3 18 0.07 15.3 a: other synthesis condition: 70 ℃, H2BDC :Co=2 :1(molar ratio);
b: other synthesis condition: pH=6, H2BDC :Co=2 :1(molar ratio);
c: other synthesis condition: 70 ℃, pH=6表 2 Co-BDC MOFs和Co@C-Ar、Co@C-C2H2催化剂的性质
Table 2 Textural properties and surface elemental content of the Co-BDC MOFs and Co@C catalysts
Sample Surface elemental contenta w/% ABET/(m2·g-1) Pore size d/nm vtol/(cm3·g-1) Co C O Co-BDC 10.29 54.72 34.99 64 12.9 0.20 Co@C-C2H2 1.16 83.15 15.70 137 13.3 0.25 Co@C-Ar 1.65 75.78 22.57 86 11.0 0.23 a: analysed by XPS -
[1] 戴德立. 2018BP世界能源统计年鉴[Z]. http://www.bp.com/papercopies. 2018-6.DUDLEY B. BP Statistical Review of World Energy[Z]. http://www.bp.com/papercopies. 2018-6. [2] DRY M E. The Fischer-Tropsch process:1950-2000[J]. Catal Today, 2002, 71(3):227-241. doi: 10.1016-S0920-5861(01)00460-6/ [3] VAN DER LAAN G P, BEENACKERS A A C M. Kinetics and selectivity of the Fischer-Tropsch synthesis:A literature review[J]. Catal Rev, 1999, 41(3/4):255-318. doi: 10.1081-CR-100101170/ [4] ZHANG Q H, DENG W P, WANG Y. Recent advances in understanding the key catalyst factors for Fischer-Tropsch synthesis[J]. J Energy Chem, 2013, 22(1):27-38. doi: 10.1016/S2095-4956(13)60003-0 [5] DAVIS B H. Fischer-Tropsch synthesis:Comparison of performances of iron and cobalt catalysts[J]. Chem Eng Prog, 2007, 46(26):8938-8945. http://cn.bing.com/academic/profile?id=c69b60cc0d01abd799cb935c1d44cfa2&encoded=0&v=paper_preview&mkt=zh-cn [6] YU Z X, BORG Y, CHEN D, ENGER C B, FRØSETH V, RYTTER E, WIGUM H, HOLMEN A. Carbon nanofiber supported cobalt catalysts for Fischer-Tropsch synthesis with high activity and selectivity[J]. Catal Lett, 2006, 109(1/2):43-47. doi: 10.1007-s10562-006-0054-6/ [7] GIRARDON J S, QUINET E, GRIBOVAL-CONSTAANT A, CHERNAVSKⅡ P A, GENGEMBRE L, KHODAKOV A Y. Cobalt dispersion, reducibility, and surface sites in promoted silica-supported Fischer-Tropsch catalysts[J]. J Catal, 2007, 248(2):143-157. doi: 10.1016/j.jcat.2007.03.002 [8] BERGE P J V, LOOSDRECHT J V D, BARRADAS S. Oxidation of cobalt based Fischer-Tropsch catalysts as a deactivation mechanism[J]. Catal Today, 2000, 58(4):321-334. doi: 10.1016/S0920-5861(00)00265-0 [9] RYTTER E, HOLMEN A. Deactivation and regeneration of commercial type Fischer-Tropsch Co-catalysts-A mini-review[J]. Catalysts, 2015, 5(2):478-499. doi: 10.3390/catal5020478 [10] TAVASOLI A, KARIMI S, TAGHAVI S, ZOLFAGHARI Z, AMIRFIROUZKOUHI H. Comparing the deactivation behaviour of Co/CNT and Co/γ-Al2O3 nano catalysts in Fischer-Tropsch synthesis[J]. J Nat Gas Chem, 2012, 21(5):605-613. doi: 10.1016/S1003-9953(11)60409-X [11] LOOSDRECHT J V D, BALZHINIMAEV B, DALMON J A, NIEMANTSVERDRIET J W, TSYBULYA S V, SAIB A M, BERGE P J V, VISAGIE J L. Cobalt Fischer-Tropsch synthesis:Deactivation by oxidation?[J]. Catal Today, 2007, 123(1/4):293-302. http://cn.bing.com/academic/profile?id=1ff471a689ea5f0cfffede647cd5d32d&encoded=0&v=paper_preview&mkt=zh-cn [12] TAVASOLI A, TRÉPANIER M, DALAI A K, ABATZOGLOU N. Effects of confinement in carbon nanotubes on the activity, selectivity, and lifetime of Fischer-Tropsch Co/carbon nanotube catalysts[J]. J Chem Eng Data, 2010, 55(8):2757-2763. doi: 10.1021/je900984c [13] TRÉPANIER M, DALAI A K, ABATZOGLOU N. Synthesis of CNT-supported cobalt nanoparticle catalysts using a microemulsion technique:Role of nanoparticle size on reducibility, activity and selectivity in Fischer-Tropsch reactions[J]. Appl Catal A:Gen, 2010, 374(1/2):79-86. http://cn.bing.com/academic/profile?id=2e37922d9383565eb7efc0b067b67307&encoded=0&v=paper_preview&mkt=zh-cn [14] CHAIKITTISILP W, ARIGA K, YAMAUCHI Y. A new family of carbon materials:Synthesis of MOF-derived nanoporous carbons and their promising applications[J]. J Mater Chem A, 2013, 1(1):14-19. [15] DONG W H, ZHANG L, WANG C H, CHENG F, SHANG N Z, GAO S T, WANG C. Palladium nanoparticles embedded in metal-organic framework derived porous carbon:Synthesis and application for efficient Suzuki-Miyaura coupling reactions[J]. RSC Adv, 2016, 6(43):37118-37123. doi: 10.1039/C6RA00378H [16] EI Y P, LI Z, LI Y W. Highly active and selective Co-based Fischer-Tropsch catalysts derived from metal-organic frameworks[J]. AIChE J, 2017, 63(7):2935-2944. doi: 10.1002/aic.15677 [17] QIU B, YANG C, GUO W H, XU Y, LIANG Z B, MA D, ZOU R Q. Highly dispersed Co-based Fischer-Tropsch synthesis catalysts from metal-organic frameworks[J]. J Mater Chem A, 2017, 5(17):8081-8086. doi: 10.1039/C7TA02128C [18] SUN X H, OLIVOS-SUAREZ A I, OAR-ARTETA L, ROZHKO E, OSADCHⅡ D, BAVYKINA A, KAPTEIJN F, GASCON J. Metal-organic framework mediated cobalt/nitrogen-doped carbon hybrids as efficient and chemoselective catalysts for the hydrogenation of nitroarenes[J]. Chem Cat Chem, 2017, 9(10):1854-1862. http://cn.bing.com/academic/profile?id=c330a139d978dde13a60d5dde7b14d41&encoded=0&v=paper_preview&mkt=zh-cn [19] XIA W, MAHMOOD A, ZOU R Q, XU Q. Metal-organic frameworks and their derived nanostructures for electrochemical energy storage and conversion[J]. Energy Environ Sci, 2015, 8(7):1837-1866. doi: 10.1039/C5EE00762C [20] ZHANG C H, GUO X X, YUAN Q C, ZHANG R L, CHANG Q, LI K, XIAO B, LIU S Y, MA C P, LIU X, XU Y Q, WEN X D, YANG Y, LI Y W. Ethyne-reducing metal-organic frameworks to control fabrications of core/shell nanoparticles as catalysts[J]. ACS Catal, 2018, 8(8):7120-7130. doi: 10.1021/acscatal.8b01691 [21] CHO H S, DENG H X, MIYASAKA K, DONG Z Y, CHO M, NEIMARK A V, KANG J K, YAGHI O M, TERASAKI O. Extra adsorption and adsorbate superlattice formation in metal-organic frameworks[J]. Nature, 2015, 527(7579):503-7. doi: 10.1038/nature15734 [22] LI H L, EDDAOUDI M, KEEFFE M O, YAGHI O M. Design and synthesis of an exceptionally stable and highly porous metal-organic framework[J]. Nature, 1999, 402(6759):276-279. doi: 10.1038/46248 [23] SAMBANDAM B, SOUNDHARRAJAN V, MATHEW V, SONG J J, KIM S J, JO J, DUONG P T, KIM S, MATHEW V, KIM J. Metal-organic framework-combustion:A new, cost-effective and one-pot technique to a porous Co3V2O8 microspheres anode for high energy Lithium-ion batteries[J]. J Mater Chem A, 2016, (4):14605-14613. [24] CENDROWSKI K, ZENDEROWSKA A, BIEGANSKA A, MIJOWSKA E. Graphene nanoflakes functionalized with cobalt/cobalt oxides formation during cobalt organic framework carbonization[J]. Dalton T, 2017, 46(24):7722-7732. doi: 10.1039/C7DT01048F [25] 贺永林.碱减量废水中对苯二甲酸的回收与提纯[D].无锡: 江南大学, 2013. http://cdmd.cnki.com.cn/Article/CDMD-10295-1014148962.htmHE Yong-lin. Recycing and purifing terephthalic acid from wastewater of alkali deweighting finishing of polyester[D]. Wuxi: Jiangnan University, 2013. http://cdmd.cnki.com.cn/Article/CDMD-10295-1014148962.htm [26] 解谷声.碱减量加工废液中对苯二甲酸的利用[J].辽宁丝绸, 2001, (2):38-40. doi: 10.3969/j.issn.1671-3389.2001.02.014XIE Gu-sheng. Utilization of terephthalic acid in waste liquor of alkali reduction processing[J]. Liaoning Tussah Silk, 2001, (2):38-40. doi: 10.3969/j.issn.1671-3389.2001.02.014 [27] 省聪聪.基于MOFs合成Co-空心/核壳结构@石墨烯复合材料、表征及性能研究[D].郑州: 郑州大学, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10459-1017150611.htmSHENG Cong-cong. Based on MOFs to Co-hollow/core-shell@graphene composite materials: Preparation, characterization and properties[D]. Zhengzhou: Zhengzhou University, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10459-1017150611.htm [28] WELLER S, HOFER L J E, ANDERSON R B. The role of bulk cobalt carbide in the Fischer-Tropsch synthesis[J]. J Am Chem Soc, 1948, (2):799-801. [29] XIONG J M, DING Y J, WANG T, YAN L, CHEN W M, ZHU H J, LU Y. The formation of Co2C species in activated carbon supported cobalt-based catalysts and its impact on Fischer-Tropsch reaction[J]. Catal Lett, 2005, 102(3/4):265-269. http://cn.bing.com/academic/profile?id=6fcfef4809e0e9e4dbfda805b7930339&encoded=0&v=paper_preview&mkt=zh-cn [30] ZHONG L, YU F, AN Y, ZHAO Y, SUN Y, LI Z, LIN T, LIN Y, QI X, DAI Y, GU L, HU J, JIN S, SHEN Q, WANG H. Cobalt carbide nanoprisms for direct production of lower olefins from syngas[J]. Nature, 2016, 538(7623):84-87. doi: 10.1038/nature19786 [31] ZHANG R G, WEN G X, ADIDHARMA H, RUSSELL A G, WANG B J, RADOSZ M, FAN M. C2 oxygenates synthesis via Fischer-Tropsch synthesis on Co2C and Co/Co2C interface catalysts:How to control the catalyst crystal facet for optimal selectivity[J]. ACS Catal, 2017, 7(12):8285-8295. doi: 10.1021/acscatal.7b02800 -





 下载:
下载: