-
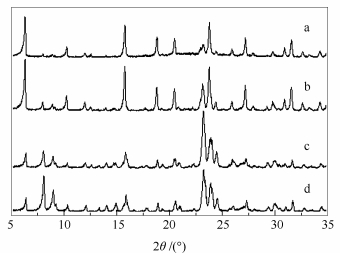
摘要: 采用水热晶化法制备了HUSY@MFI核壳结构复合分子筛。通过XRD、SEM、N2吸附-脱附、NH3-TPD及吡啶吸附红外等手段表征催化剂的结构和性质。结果表明,HUSY@MFI晶粒在形貌上呈椭球状,表面是鳞片状结构的MFI型分子筛,里面是光滑的HUSY型分子筛,焙烧模板剂前几乎没有或只有很少量的N2能进入其孔道结构,致密的壳层MFI覆盖在HUSY型分子筛表面,形成了新的弱酸位,而中强酸强度和酸类型并没有受到影响,复合分子筛的表面酸量及总酸量减少。将所制备的HUSY@MFI复合分子筛催化剂应用于以离子液体1-乙基-3-甲基咪唑氯盐([Emim]Cl)为溶剂的纤维素水解反应中,与HUSY催化的纤维素水解相比,HUSY@MFI复合分子筛催化纤维素水解反应的速率较慢,葡萄糖收率由30.9%提高到41.3%。Abstract: In this paper, core-shell composite zeolites (HUSY@MFI) were prepared by hydrothermal method. The composite zeolites were characterized by XRD, SEM, N2-adsorption, NH3-TPD and Py-FTIR. The results indicated that HUSY@MFI has both HUSY and MFI structure. Scanning Electron Microscope (SEM) reflects a core-shell morphology of HUSY@MFI. The particles displayed an elliptical sphere structure with scale-like surface. The growth of MFI shell results in a decrease of external acid density and the total acid sites. When the HUSY@MFI was used as catalyst instead of HUSY in hydrolysis of cellulose to glucose in 1-ethyl-3-methylimidazolium chloride ([Emim]Cl), the glucose yield could be significantly improved from 30.9% to 41.3%.
-
Key words:
- HUSY@MFI /
- HUSY /
- cellulose /
- hydrolysis /
- glucose
-
表 1 样品的孔参数
Table 1 Pore parameters of the samples
Sample Surface area ABET /(m2·g-1) Pore volume v/(cm3·g-1) micropore mesopore total micropore mesopore total HUSY 679 134 813 0.27 0.23 0.50 MFI 239 134 373 0.10 0.11 0.21 HUSY@MFI(calcined) 334 150 484 0.14 0.12 0.26 HUSY@MFI(non-calcined) 29 24 53 0.01 0.03 0.04 表 2 样品的红外酸度分析
Table 2 Brønsted acid sites and Lewis acid sites of the catalysts
Sample Total acid/
(mmol·g-1)Acid amount/
(mmol·g-1)B/L brønsted lewis HUSY 0.843 0.543 0.300 1.8 HUSY@MFI 0.486 0.286 0.200 1.4 表 3 不同催化剂的最佳反应效果
Table 3 Best results of the reactions with different catalysts
Catalyst Time t/h Yield w/% TRS oligosaccharide glucose 5-HMF HUSY@MFI 3.5 88 5.5 41.29 3.97 HUSY 2 65 3.12 30.91 7.82 -
[1] PEREGO C, BOSETTI A B. Biomass to fuels:The role of zeolite and mesoporous materials[J]. Microporous Mesoporous Mater, 2011, 144:28-39. doi: 10.1016/j.micromeso.2010.11.034 [2] GOYAL H B, SEAL Diptendu, SAXENA R C. Bio-fuels from thermochemical conversion of renewable resources:A review[J]. Renewable Sustainable Energy Rev, 2008, 12:504-517. doi: 10.1016/j.rser.2006.07.014 [3] GUO H X, LIAN Y F, YAN L L, QI X H, SMITH R. Cellulose-derived superparamagnetic carbonaceous solid acid catalyst for cellulose hydrolysis in an ionic liquid or aqueous reaction system[J].Green Chem, 2013, 15:2167-2174. doi: 10.1039/c3gc40433a [4] CHUNDAWAT S P S, BECKHAM G T, HIMMEL M E, DALE B E, Deconstruction of lignocellulosic biomass to fuels and chemicals[J]. Annu Rev Chem Biomol Eng, 2011, 2:121-145. doi: 10.1146/annurev-chembioeng-061010-114205 [5] GEBOERS J, VYVER S V, OOMS R, BEECK B, JACOBS P, SELS B F. Chemocatalytic conversion of cellulose:Opportunities, advances and pitfalls[J]. Catal Sci Technol, 2011, 1:1714-1726. http://pubs.rsc.org/en/content/articlehtml/2011/cy/c1cy00093d [6] CHANDRAKANT P, BISARIA V S. Simultaneous bioconversion of cellulose and hemicellulose to ethanol[J]. Crit Rev Biotechnol, 1998, 18(4):295-331. doi: 10.1080/0738-859891224185 [7] DENG W P, ZHANG Q H, WANG Y. Catalytic transformations of cellulose and its derived carbohydrates into 5-hydroxymethylfurfural, levulinic acid, and lactic acid[J]. Sci China Chem, 2015, 58(1):29-46. doi: 10.1007/s11426-014-5283-8 [8] TAN M X, ZHAO L, ZHANG Y G. Production of 5-hydroxymethyl furfural from cellulose in CrCl2/Zeolite/BMIMCl system[J]. biomass and bioenergy, 2011, 35:1367-1370. doi: 10.1016/j.biombioe.2010.12.006 [9] RINALDI R, SCHÜTH F. Acid hydrolysis of cellulose as the entry point into biorefinery schemes[J]. Chemsuschem, 2009, 21:1096-1107. doi: 10.1002/cssc.200990045/abstract [10] TORGET R W, KIM J, LEE Y Y. Fundamental aspects of dilute acid hydrolysis/fractionation kinetics of hardwood carbohydrates. 1. Cellulose hydrolysis[J]. Ind Eng Chem Res, 2000, 39:2817-2825. doi: 10.1021/ie990915q [11] 赵博, 胡尚连, 龚道勇, 李会萍.固体酸催化纤维素水解转化葡萄糖的研究进展[J].化工进展, 2017, 36(2):555-567.. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgjz201411021ZHAO Bo, HU Shang-lian, GONG Dao-yong, LI Hui-ping. New advances on hydrolysis of cellulose to glucose by solid acid[J]. Chem Ind Eng Prog, 2017, 36(2):555-567. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hgjz201411021 [12] 申曙光, 李焕梅, 王涛, 蔡蓓, 秦海峰, 王春艳.煤化程度对煤基固体酸结构及其水解纤维素性能的影响[J].燃料化学学报, 2013, 41(12):1466-1472.. http://rlhxxb.sxicc.ac.cn/CN/Y2013/V41/I12/1466SHEN Shu-guang, LI Huan-mei, WANG Tao, CAI Bei, QIN Hai-feng, WANG Chun-yan. Effect of coal rank on structure of coal-based solid acids and their catalytic performance in cellulose hydrolysis[J]. J Fuel Chem Technol, 2013, 41(12):1466-1472. http://rlhxxb.sxicc.ac.cn/CN/Y2013/V41/I12/1466 [13] CAI H L, LI C Z, WANG A Q, XU G L, ZHANG T. Zeolite-promoted hydrolysis of cellulose in ionic liquid, insight into the mutual behavior of zeolite, cellulose and ionic liquid[J]. Appl Catal B:Environ, 2012, 123/124:333-338. doi: 10.1016/j.apcatb.2012.04.041 [14] LAI D M, DENG L, GUO Q X, FU Y. Hydrolysis of biomass by magnetic solid acid[J]. Energy Environ Sci, 2011, 4:3552-3557. doi: 10.1039/c1ee01526e [15] RINALDI R, PALKOVITS R, SCHUTH F. Depolymerization of cellulose using solid catalysts in ionic liquids[J]. Angew Chem Int Ed, 2008, 47:8047-8050. doi: 10.1002/anie.v47:42 [16] GUO H X, QI X H, LI L Y, SMITH R L. Hydrolysis of cellulose over functionalized glucose-derived carbon catalyst in ionic liquid[J]. Bioresour Technol, 2012, 116:355-359. doi: 10.1016/j.biortech.2012.03.098 [17] SATOSHI S, MASAAKI K, DAIZO Y, KIYOTAKA N, HIDEKI K, SHIGENOBU H, MICHIKAZU H. Hydrolysis of cellulose by amorphous carbon bearing SO3H, COOH, and OH groups[J]. J Am Chem Soc, 2008, 130(38):12787-12793. doi: 10.1021/ja803983h [18] ZHOU L P, LIU Z, SHI M T, DU S S, SUA Y L, YANG X M, XU J. Sulfonated hierarchical H-USY zeolite for efficient hydrolysis of hemicellulose/cellulose[J]. Carbohydrate Polymers, 2013, 98:146-151. doi: 10.1016/j.carbpol.2013.05.074 [19] ONDA A, OCHI T, YANAGISAWA K. Selective hydrolysis of cellulose into glucose over solid acid catalysts[J]. Green Chem, 2008, 10(10):1033-1037. doi: 10.1039/b808471h [20] SWATLOSKI R P, SPEAR S K, HOLBREY J D, ROGERS R D. Dissolution of cellose with ionic liquids[J]. J Am Chem Soc, 2002, 124(18):4974-4975. doi: 10.1021/ja025790m [21] LOPES A M D C, JOÃO K G, RUBIK D F, BOGE-ŁUKASIK E, DUARTE L, ANDREAUS J, BOGEL-ŁUKASIK R. Pre-treatment of lignocellulosic biomass using ionic liquids:Wheat straw fractionation[J]. Bioresour Technol, 2013, 142:198-208. doi: 10.1016/j.biortech.2013.05.032 [22] LOPES A M D C, BOGEL-ŁUKASIK R. Acidic ionic liquids as sustainable approach of cellulose and lignocellulosic biomass conversion without additional catalysts[J]. ChemSusChem, 2015, 8:947-965. doi: 10.1002/cssc.v8.6 [23] TAO F R, SONG H L, CHOU L J. Hydrolysis of cellulose in SO3H-functionalized ionic liquids[J]. Bioresour Technol, 2011, 102:9000-9006. doi: 10.1016/j.biortech.2011.06.067 [24] WANG J Y, ZHOU M D, YUAN Y G, ZHANG Q, FANG X C, ZANG S L. Hydrolysis of cellulose catalyzed by quaternary ammonium perrhenates in 1-allyl-3-methylimidazolium chloride[J]. Bioresour Technol, 2015, 197:42-47. doi: 10.1016/j.biortech.2015.07.110 [25] YUAN Y G, WANG J Y, FU N H, ZANG S L. Hydrolysis of cellulose in 1-allyl-3-methylimidazolium chloride catalyzed by methyltrioxorhenium[J]. Catal Commun, 2016, 76:46-49. doi: 10.1016/j.catcom.2015.12.024 [26] ABOU-YOUSEF H, HASSAN E B. A novel approach to enhance the activity of H-form zeolite catalyst for production of hydroxymethylfurfural from cellulose[J]. J Ind Eng Chem, 2014, 20:1952-1957. doi: 10.1016/j.jiec.2013.09.016 [27] ZHANG Z H, ZHAO Z B. Solid acid and microwave-assisted hydrolysis of cellulose in ionic liquid[J]. Carbohydrate Res, 2009, 344:2069-2072. doi: 10.1016/j.carres.2009.07.011 [28] ZHOU L P, LIU Z, BAI Y Q, LU T L, YANG X M, XU J. Hydrolysis of cellobiose catalyzed by zeolites-the role of acidity and micropore structure[J]. J Energy Chem, 2016, 25:141-145. doi: 10.1016/j.jechem.2015.11.010 [29] ZHENG J J, SUN X B. Structural features of core-shell zeolite-zeolite composite and its performance for methanol conversion into gasoline and diesel[J]. J Mater Res, 2016, 31:2302-2316. doi: 10.1557/jmr.2016.208 [30] MILLER G L. Use of dinitrosaiicyiic acid reagent for determination of reducing sugar[J]. Anal Chem, 1959, 31:426-428. doi: 10.1021/ac60147a030 [31] MAO R, RAMSARAN A, XIAO S Y, YAO J H, SEMMER V. PH of the sodium carbonate solution used for the desilication of zeolite materials[J]. J Materials Chem, 1995, 5(3):533-535. doi: 10.1039/jm9950500533 [32] GROEN J C, MOULIJN J A, PÉREZ-RAMÍREZ J. Desilication:On the controlled generation of mesoporosity in MFI zeolites[J]. J Mater Chem, 2006, 16(22):2121-2131. doi: 10.1039/B517510K [33] 郭大雷, 郑家军, 易玉明, 张球, 潘梦, 李瑞丰. FMZ双沸石复合物的合成及其表征[J].石油学报(石油加工), 2013, 29(4):591-596.. doi: 10.3969/j.issn.1001-8719.2013.04.007GUO Da-lei, ZHENG Jia-jun, YI Yu-ming, ZHANG Qiu, PAN Meng, LI Rui-feng. Synthesis and characterization of FMZ bi-phases composite zeolites[J]. Acta Pet Sin(Pet Process Sect), 2013, 29(4):591-596. doi: 10.3969/j.issn.1001-8719.2013.04.007 [34] QI X H, WATANABE M, AIDA T M, SMITH R L. Catalytic conversion of cellulose into 5-hydroxymethylfurfural in high yields via a two-step process[J]. Cellulose, 2011, 18:1327-1333. doi: 10.1007/s10570-011-9568-1 -





 下载:
下载: