Stability, rheology and displacement performance of nano-SiO2/HPAM/NaCl dispersion systems
-
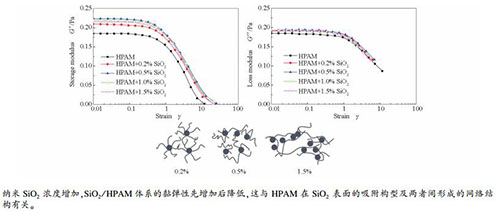
摘要: 利用纳米粒度及Zeta电位分析仪、流变仪和悬滴法对纳米SiO2/HPAM/NaCl体系60 ℃的稳定性、流变性及油水界面张力进行了研究。结果表明,HPAM的加入使SiO2悬浮液的Zeta电位更负、粒径明显增加,静置10 d无明显浑浊现象。加入纳米SiO2后,HPAM溶液的黏度增加,耐温、耐盐和耐剪切性能得到改善。对于质量分数为0.18%的HPAM溶液,SiO2质量分数小于0.5%时,随SiO2质量分数的增加,体系的黏度、储能模量和损耗模量增加,临界线性应变减小,蠕变回复能力增强;SiO2质量分数大于0.5%时,出现了相反的现象;这是因为SiO2质量分数不同时,HPAM在SiO2表面的吸附量、吸附构型及两者之间形成的网状结构不同。纳米SiO2的加入同时强化了HPAM降低油水界面张力的性能,加入质量分数为0.2%和0.5%的SiO2后,HPAM的采收率分别提高了4.5%和6.0%。Abstract: The stability, rheological properties and oil/water interfacial tension of Nano-SiO2/HPAM/NaCl systems at 60 ℃ were studied by Zetasizer, rheometer and spin-drop method, respectively. The results indicated that the zeta potential value of nano-SiO2 became more negative and the particle size was significantly increased with addition of HPAM. Meanwhile, there was no obvious turbidity phenomenon after 10 d. The nano-SiO2/HPAM suspensions had higher viscosity and the viscosity retention was improved in the presence of salt at high temperature and shear rate as compared to HPAM solution. In this work, the nano-SiO2 threshold for 0.18%(mass ratio) HPAM solution was 0.5% (mass ratio). When the mass ratio of nano-SiO2 was less than 0.5%, the viscosity, storage modulus, loss modulus and creeping recovery properties were enhanced as well as the critical strain was decreased with the increase of nano-SiO2 mass fraction. However, the opposite phenomenon was investigated when the mass ratio of nano-SiO2 was more than 0.5%. The reason for this result was that the polymer amounts, polymer conformation onto the nano-SiO2 surface and the network structure between nano-SiO2 and HPAM were different when the nano-SiO2 mass fraction was different. Oil/water interfacial tension values of nano-SiO2/HPAM suspensions were lower than that of HPAM solution, and thus with addition of 0.2% and 0.5% (mass ratio) nano-SiO2, the nano-SiO2/HPAM suspensions had ultimate oil recoveries of 4.5% and 6.0% higher than polymer flooding.
-
Key words:
- nano-SiO2 /
- HPAM /
- viscoelasticity /
- stability /
- enhanced oil recovery
-
表 1 分散体系不同静置时间的Zeta电位
Table 1 eta potentials of dispersion systems at different time intervals
w(SiO2)/% Zeta potential/mV HPAM+SiO2 SiO2 initial 10 d initial 10 d 0 -65.5 -52.3 - - 0.2 -32.8 -29.2 -8.8 -7.9 0.5 -31.5 -27.9 -15.4 -12.9 1.0 -30.5 -26.6 -17.6 -14.8 1.5 -27.4 -24.9 -19.3 -16.9 表 2 不同体系提高稠油采收率实验
Table 2 Experimental data of different systems for enhanced heavy oil recovery
Chemical formula Viscosity/(mPa·s) Permeability/10-3μm2 Slug size/PV Water flood recovery/% Final recovery/% Tertiary recovery/% HPAM 41.5 1329.0 0.5 42.0 64.7 22.7 HPAM+0.2%SiO2 51.0 1355.0 0.5 42.8 70.0 27.2 HPAM+0.5%SiO2 53.1 1340.0 0.5 42.5 71.2 28.7 -
[1] WANG J, DONG M. Optimum effective viscosity of polymer solution for improving heavy oil recovery[J]. J Pet Sci Eng, 2009, 67(3):155-158. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d9d5edfd5335d64c85f731cc257008e5 [2] 李美蓉, 柳智, 宋新旺, 马宝东, 张文.金属阳离子对聚丙烯酰胺溶液黏度的影响及其降黏机理研究[J].燃料化学学报, 2012, 40(1):43-47. doi: 10.3969/j.issn.0253-2409.2012.01.007LI Mei-rong, LIU Zhi, SONG Xin-wang, MA Bao-dong, ZHANG Wen. Effect of metal ions on the viscosity of polyacrylamide solution and the mechanism of viscosity degradation[J]. J Fuel Chem Technol, 2012, 40(1):43-47. doi: 10.3969/j.issn.0253-2409.2012.01.007 [3] PEI H H, ZHANG G C, GE J J, ZHANG L, WANG H. Effect of polymer on the interaction of alkali with heavy oil and its use in improving oil recovery[J]. Colloid Surface A, 2014, 446(5):57-64. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6deb24cb15bce2afd38c7117044998d7 [4] 李美蓉, 黄漫, 曲彩霞, 曹绪龙, 张继超, 刘坤.剪切作用对功能聚合物微观结构性能的影响研究[J].燃料化学学报, 2013, 41(4):449-455. doi: 10.3969/j.issn.0253-2409.2013.04.010LI Mei-rong, HUANG Man, QU Cai-xia, CAO Xu-long, ZHANG Ji-chao, LIU Kun. Effect of shear action on the microcosmic structure and performance of functional polymer used in oil displacement[J]. J Fuel Chem Technol, 2013, 41(4):449-455. doi: 10.3969/j.issn.0253-2409.2013.04.010 [5] CORREDOR R L M, SARAPARDEH A H, HUSEIN M M, DONG P M. Rheological behavior of surface modified silica nanoparticles dispersed in partially hydrolyzed polyacrylamide and xanthan gum solutions:Experimental measurements, mechanistic understanding, and model development[J]. Energy Fuels, 2018, 32(10):10628-10638. doi: 10.1021/acs.energyfuels.8b02658 [6] CAO J, SONG T, WANG X J, ZHU Y J, WANG S S, ZHAO M T, MIAO Y J, ZHANG J. Studies on the rheological properties of amphiphilic nanosilica and a partially hydrolyzed polyacrylamide hybrid for enhanced oil recovery[J]. Chem Eng Sci, 2019, 206:146-155. doi: 10.1016/j.ces.2019.05.034 [7] 郑超.表面改性纳米二氧化硅的制备及其对HPAM驱油性能的影响[D].开封: 河南大学, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10475-1017227144.htmZHENG Chao. Preparation of surface modified nanosilica and its impact on enhanced oil recovery of HPAM solution[D]. Kaifeng: Henan University, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10475-1017227144.htm [8] 覃孝平.改性纳米SiO2/AA/AM共聚物提高波及效率的可行性研究[D].成都: 西南石油大学, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10615-1014419507.htmQIN Xiao-ping. The feasible research of improving sweep efficiency using the modified nano-SiO2/AA/AM copolymer[D]. Chengdu: Southwest Petroleum University, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10615-1014419507.htm [9] MOHAMMED B A, KOUROSH R, RADZUAN J, ALI E B. Appraising the impact of metal-oxide nanoparticles on rheological properties of HPAM in different electrolyte solutions for enhanced oil recovery[J]. J Pet Sci Eng, 2019, 172:1057-1068. doi: 10.1016/j.petrol.2018.09.013 [10] KANG W L, CAO C X, GUO S J, TANG X C, ZEESHAN A L, GAO Y B, ZHANG X F, MUHAMMAD W I, YANG H B. Mechanism of silica nanoparticles better-thickening effect on amphiphilic polymers in high salinity condition[J]. J Mol Liq, 2019, 277:254-260. doi: 10.1016/j.molliq.2018.12.092 [11] NEETISH K M, AJAY M. Studies on behavior of suspension of silica nanoparticle in aqueous polyacrylamide solution for application in enhanced oil recovery[J]. Pet Sci Technol, 2016, 34(5):429-436. doi: 10.1080/10916466.2016.1145693 [12] MAGHZI A, KHARRAT R, MOHEBBI A, GHAZANFARI M H. The impact of silica nanoparticles on the performance of polymer solution in presence of salts in polymer flooding for heavy oil recovery[J]. Fuel, 2014, 123:123-132. doi: 10.1016/j.fuel.2014.01.017 [13] KHALILINEZHAD S S, CHERAGHIAN G, ROAYAEI E, TABATABAEE H, KARAMBEIGI M S. Improving heavy oil recovery in the polymer flooding process by utilizing hydrophilic silica nanoparticles[J]. Energy Sources Part A, 2017, 12:1-10. doi: 10.1080/15567249.2014.881931 [14] SAEED J D S, LESLEY A J, ZHANG, Y H. Insight into the stability of hydrophilic silica nanoparticles in seawater for Enhanced oil recovery implications[J]. Fuel, 2018, 216:559-571. doi: 10.1016/j.fuel.2017.11.091 [15] METIN C O, LAKEA L W, MIRANDA C R, NGUYEN Q P. Stability of aqueous silica nanoparticle dispersions[J]. J Nanopart Res, 2010, 13(2):839-850. doi: 10.1007/s11051-010-0085-1 [16] 王婷. pH诱导HPAM及HPAM/HEC的水相自组装[D].无锡: 江南大学, 2009. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=Y1584865WANG Ting. pH-induced self-assembly of HPAM and HPAM/HEC in aqueous solution[D]. Wuxi: Jiangnan University, 2009. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=Y1584865 [17] 林迪.细菌胞外聚合物在土壤矿物和纳米颗粒表面的吸附及其驱动团聚机制[D].武汉: 华中农业大学, 2018. http://d.old.wanfangdata.com.cn/Thesis/Y3396505LIN Di. Adsorption and aggregation mechanisms between bacterial extracellular polymeric substrances and soil minerals and nanoparticles[D]. Wuhan: Huazhong Agricultural University, 2018. http://d.old.wanfangdata.com.cn/Thesis/Y3396505 [18] GIRALDO L J, GIRALDO M A, LLANOS S, MAYA G, ZABALA R D, NASSAR N N, FRANCO C A, ALVARADO V, CORTES F B. The effects of SiO2 nanoparticles on the thermal stability and rheological behavior of hydrolyzed polyacrylamide based polymeric solutions[J]. J Pet Sci Eng, 2017, 159:841-852. doi: 10.1016/j.petrol.2017.10.009 [19] BARANY S. Polymer adsorption and electrokinetic potential of dispersed particles in weak and strong electric fields[J]. Adv Colloid Interface, 2015, 222:58-69. doi: 10.1016/j.cis.2014.09.009 [20] 刘付胜聪, 肖汉宁, 李玉平.聚丙烯酸在纳米TiO2表面吸附行为的研究[J].高等学校化学学报, 2005, 26(4):742-746. doi: 10.3321/j.issn:0251-0790.2005.04.028LIU FU Sheng-cong, XIAO Han-ning, LI Yu-ping. Adsorption behavior of poly(acrylic acid) on the surface of nanoparticulate titanium dioxide[J]. Chem J Chin Univ, 2005, 26(4):742-746. doi: 10.3321/j.issn:0251-0790.2005.04.028 [21] GREGORY J, BARANY S. Adsorption and flocculation by polymers and polymer mixtures[J]. Adv Colloid Interface Sci, 2011, 169(1):1-12. doi: 10.1016/j.cis.2011.06.004 [22] 孙秀芝.注聚过程中HPAM水溶液剪切稳定性研究[D].青岛: 中国石油大学(华东), 2009. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=Y1542839SUN Xiu-zhi. Study on shearing stability of HPAM solution in flooding[D]. Qingdao: China University of Petroleum(East China), 2009. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=Y1542839 [23] CHEN W H, CHEN J. Crystallization behaviors of biodiesel in relation to its rheological properties[J]. Fuel, 2016, 171:178-185. doi: 10.1016/j.fuel.2015.12.049 [24] 蒲万芬, 彭彩珍, 扬清彦, 吴晓玲, 黄爱斌.聚合物溶液在孔隙介质中的蠕变回复和驱油效率[J].西南石油学院学报, 2000, 22(2):62-67. doi: 10.3863/j.issn.1674-5086.2000.02.019PU Wan-fen, PENG Cai-zhen, YANG Qing-yan, WU Xiao-ling, HUANG Ai-bin. Effect of creeping reversion of polymer solution in porous medium on the displacement efficiency[J]. J Southwest Pet Inst, 2000, 22(2):62-67. doi: 10.3863/j.issn.1674-5086.2000.02.019 [25] BINKS B P, RODRIGUES J A, FRITH W J. Synergistic interaction in emulsions stabilized by a mixture of silica nanoparticles and cationic surfactant[J]. Langmuir, 2007, 23(14):3626-3636. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6a1d23b02d38902303096e6876a206f6 -





 下载:
下载: