Effect of reduction temperature on the performance of Ni2P/Ti-MCM-41 catalyst in hydrodesulfurization
-
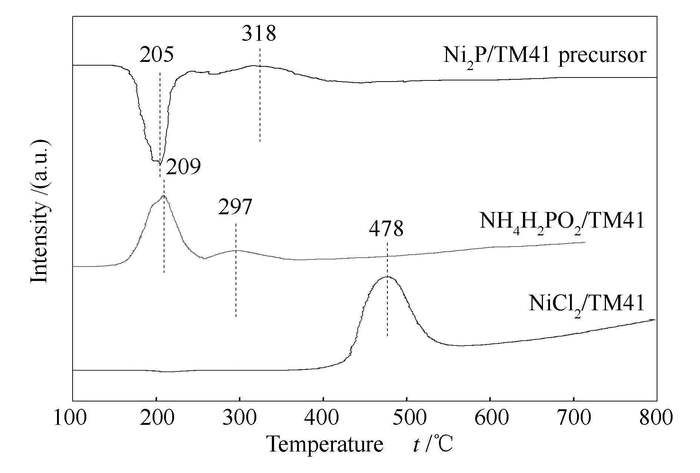
摘要: 以氯化镍(NiCl2·6H2O)为镍源、次磷酸铵(NH4H2PO2)为磷源、Ti-MCM-41为载体,通过程序升温还原法制备了Ni2P/Ti-MCM-41催化剂,并采用H2-TPR、XRD、BET、XPS、TEM等手段对其结构和性质进行了表征。以二苯并噻吩(DBT)为模型化合物,考察了还原温度对Ni2P/Ti-MCM-41催化剂的加氢脱硫(HDS)性能的影响。结果表明,程序升温还原法制备的Ni2P/Ti-MCM-41催化剂前驱体的还原温度为318℃,比传统程序升温还原制备的Ni2P低200℃。在350-500℃下还原得到的催化剂活性相为单一的Ni2P相,较低的还原温度有利于形成更小粒径的磷化镍晶粒。还原温度为400℃时,制得的Ni2P/Ti-MCM-41催化剂比表面积高、分散性最好、表面P富集少,具有最高的HDS活性;在340℃、3.0MPa、H2/油体积比500、质量空速(WHSV)为2.0h-1的条件下,二苯并噻吩HDS转化率达到99.4%。Abstract: The supported Ni2P/Ti-MCM-41 catalyst is prepared by temperature-programmed reduction method with nickel chloride (NiCl2·6H2O) as the nickel precursor, ammonium hypophosphite (NH4H2PO2) as the phosphorus precursor and Ti-MCM-41 as the support. The Ni2P/Ti-MCM-41 catalyst was characterized by H2-TPR, XRD, BET, XPS, and TEM; the effect of reduction temperature on its catalytic performance in hydrodesulfurization (HDS) was investigated by using dibenzothiophene (DBT) as a model compound. The results show that the precursors on the catalyst prepared in this way can be reduced at 318℃, which is at least 200℃ lower than that prepared by traditional methods. Pure Ni2P phase can be obtained by reduction at 350-500℃; the low reduction temperature is in favor of forming small Ni2 Pcrystallite size. The Ni2P/Ti-MCM-41 catalyst obtained at a reduction temperature of 400℃ exhibits the highest surface area, the best dispersion of Ni2P crystallite size, the lowest surface phosphorus content and the highest HDS activity; under 340℃, 3.0MPa, a H2/oil ratio of 500 (volume ratio) and a weight hourly space velocity (WHSV) of 2.0h-1, the conversion of DBT for HDS reaches 99.4%.
-
Key words:
- hydrodesulfurization /
- nickel phosphide /
- reduction temperature /
- Ti-MCM-41 /
- dibenzothiophene
-
表 1 载体和Ni2P/TM41-x催化剂的表面结构
Table 1 Textural properties of the MCM-41 support and Ni2P/Ti-MCM-41-x catalysts
Sample ABET/(m2·g-1) vp/(cm3·g-1) dp/nm Dc/nma CO uptake /(μmol·g-1) MCM-41 1 012 0.816 3.2 - - Ti-MCM-41 918 0.674 2.9 - - Ni2P/TM41-350 511 0.299 2.1 7 9 Ni2P/TM41-400 546 0.335 2.2 8 12 Ni2P/TM41-450 539 0.296 2.2 10 11 Ni2P/TM41-500 524 0.275 2.1 12 8 a:calculated from the Scherrer equation based on the Ni2P {111} face 表 2 不同温度还原的Ni2P/TM41-x的XPS光谱学参数
Table 2 Spectral parameters of the Ni2P/TM41-x catalysts from XPS analysis
Sample Binding energy E/eV Superficial atomic ratio Ni 2p3/2 P 2p Ni/P Ni/Si Ni2+ Niδ+ PO43- H2PO3- Pδ- Ni2P/TM41-350 856.5 852.6 134.8 133.5 129.5 0.332 0.048 Ni2P/TM41-400 856.7 853.1 134.8 133.5 129.7 0.426 0.054 Ni2P/TM41-450 856.8 852.4 135.0 133.5 129.6 0.368 0.047 Ni2P/TM41-500 856.5 852.5 134.9 133.4 129.4 0.341 0.042 -
[1] TIAN F P, SHEN Q C, FU Z K, WU Y H, JIA C Y.Enhanced adsorption desulfurization performance over hierarchically structured zeolite Y[J].Fuel Process Technol, 2014, 128:176-182. doi: 10.1016/j.fuproc.2014.07.018 [2] 宋华, 代敏, 宋华林.Ni2P加氢脱硫催化剂[J].化学进展, 2012, 24(5):43-47.SONG Hua, DAI Min, SONG Hua-lin.Ni2P catalyst for hydrodesulfurization[J].Prog Chem, 2012, 24(5):43-47. [3] ZHAO H Y, OYAMA S T, FREUND H J, WLODARCZYK R, SIERKA M.Nature of active sites in Ni2P hydrotreating catalysts as probed by iron substitution[J].Appl Catal B:Environ, 2015, 164:204-216. doi: 10.1016/j.apcatb.2014.09.010 [4] SONG H, DAI M, SONG H L, WAN X, XU X W, ZHANG C Y, WANG H Y.Synthesis of a Ni2P catalyst supported on anatase-TiO2 whiskers with high hydrodesulfurization activity, based on triphenylphosphine[J].Catal Commun, 2014, 43:151-154. doi: 10.1016/j.catcom.2013.10.002 [5] SONG H, WANG J, WANG Z D, SONG H L, LI F, JIN Z S.Effect of titanium content on dibenzothiophene HDS performance over Ni2P/Ti-MCM-41 catalyst[J].J Catal, 2014, 311:257-265. doi: 10.1016/j.jcat.2013.11.021 [6] SONG H, DAI M, GUO Y T, ZHANG Y J.Preparation of composite TiO2-Al2O3 supported nickel phosphide hydrotreating catalysts and catalytic activity for hydrodesulfurization of dibenzothiophene[J].Fuel Process Technol, 2012, 96:228-236. doi: 10.1016/j.fuproc.2012.01.001 [7] SONG L M, ZHANG S J, WEI Q W.A new route for synthesizing nickel phosphide catalysts with high hydrodesulfurization activity based on sodium dihydrogenphosphite[J].Catal Commun, 2011, 12:1157-1160. doi: 10.1016/j.catcom.2011.03.038 [8] MELENDEZ-ORTIZ H I, GARCIA-CERDA L A, OLIVARES-MALDONADO Y, CASTRUITA G, MERCADO-SILVA J A, PERERA-MERCADO Y A.Preparation of spherical MCM-41 molecular sieve at room temperature:Influence of the synthesis conditions in the structural properties[J].Ceram Int, 2012, 38(8):6353-6358. doi: 10.1016/j.ceramint.2012.05.007 [9] 宋华, 张福勇, 徐晓伟, 宋华林, 李峰.镨和铈对低温还原法制备的Ni2P催化剂加氢脱硫活性的影响[J].燃料化学学报, 2015, 43(9):1128-1133. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18699.shtmlSONG Hua, ZHANG Fu-yong, XU Xiao-wei, SONG Hua-lin, LI Feng.Effect of Pr and Ce on the activity of Ni2P catalyst prepared by low temperature reduction in hydrodesulphurization[J].J Fuel Chem Technol, 2015, 43(9):1128-1133. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18699.shtml [10] 宋华, 王紫东, 代敏, 宋华林, 万霞, 李锋.Ni2P催化剂的低温还原法合成及其HDS性能[J].燃料化学学报, 2014, 42(6):733-737. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18436.shtmlSONG Hua, WANG Zi-dong, DAI Min, SONG Hua-lin, WAN Xia, LI Feng.Preparation of Ni2P catalysts at low reduction temperature and its HDS performance[J].J Fuel Chem Technol, 2014, 42(6):733-737. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract18436.shtml [11] WANG X, CLARK P, OYAMA S T.Synthesis, characterization, and hydrotreating activity of several iron group transition metal phosphides[J].J Catal, 2002, 208(2):321-331. doi: 10.1006/jcat.2002.3604 [12] OYAMA S T, WANG X Q, LEE Y K, CHUN W J.Active phase of Ni2P/SiO2 in hydroprocessing reactions[J].J Catal, 2004, 221(2):263-273. doi: 10.1016/S0021-9517(03)00017-4 [13] KOSSLICK H, LISCHKE G, LANDMESSER H, PARLITZ B, STOREK W, FRICKE R.Acidity and catalytic behavior of substituted MCM-48[J].J Catal, 1998, 176:102-114. doi: 10.1006/jcat.1998.2015 [14] CHEN T, YANG B L, LI S S, WANG K L, JIANG X D, ZHANG Y, HE G W.Ni2P catalysts supported on titania-modified alumina for the hydrodesulfurization of dibenzothiophene[J].Ind Eng Chem Res, 2011, 50:11043-11048. doi: 10.1021/ie201188v [15] ELICHE-QUESADA D, MERIDA-ROBLES J, MAIRELES-TORRES P, RODRIGUEZ-CASTELLON E, BUSCA G, FINOCCHIO E, JIMENEZ-LOPEZ A.Effects of preparation method and sulfur poisoning on the hydrogenation and ring opening of tetralin on NiW/zirconium-doped mesoporous silica catalysts[J].J Catal, 2003, 220(2):457-467. doi: 10.1016/S0021-9517(03)00271-9 [16] KUHN J N, LAKSHMINARAYANAN N, OZKAN U S.Effect of hydrogen sulfide on the catalytic activity of Ni-YSZ cermets[J].J Mol Catal A:Chem, 2008, 282(1/2):9-21. [17] KORANYI T I.Phosphorus promotion of Ni (Co)-containing Mo-free catalysts in thiophene hydrodesulfurization[J].Appl Catal A:Gen, 2003, 239(1/2):253-267. [18] CECILIA J A, INFANTES-MOLINA A, RODRIGUEZ-CASTELLON E, JIMENEZ-LOPEZ A.A novel method for preparing an active nickel phosphide catalyst for HDS of dibenzothiophene[J].J Catal, 2009, 263(1):4-15. doi: 10.1016/j.jcat.2009.02.013 [19] LAYMAN K A, BUSSEL M E.Infrared spectroscopic investigation of CO adsorption on silica-supported nickel phosphide catalysts[J].J Phys Chem B, 2004, 108:10930-10941. doi: 10.1021/jp037101e [20] OYAMA S T, WANG X, LEE Y K, BANDO K, REQUEJO F G.Effect of phosphorus content in nickel phosphide catalysts studied by XAFS and other techniques[J].J Catal, 2002, 210(1):207-217. doi: 10.1006/jcat.2002.3681 [21] SONG H, DAI M, SONG H L, WAN X, XU X W.A novel synthesis of Ni2P/MCM-41 catalysts by reducing a precursor of ammonium hypophosphite and nickel chloride at low temperature[J].Appl Catal A:Gen, 2013, 462-463:247-255. doi: 10.1016/j.apcata.2013.05.015 [22] SAWHILL S J, LAYMAN K A, VAN WYK D R, ENGELHARD M H, WANG C, BUSSELL M E.Thiophene hydrodesulfurization over nickel phosphide catalysts:Effect of the precursor composition and support[J].J Catal, 2005, 231(2):300-313. doi: 10.1016/j.jcat.2005.01.020 -





 下载:
下载: