Effect of mineral matter on emissions of SO2 and NOx during combustion of lean coal in O2/CO2 atmosphere
-
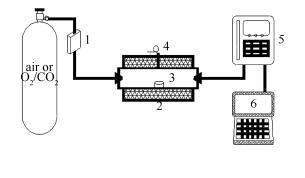
摘要: 利用管式炉反应器在550-1 000℃对长治贫煤和脱矿物质煤分别在空气和O2/CO2气氛进行了燃烧实验。利用XRF、XRD等分析手段,对煤样的基本性能进行了分析表征,并采用热分析仪(TG-DTG)以及傅里叶红外气体分析仪(Antaris-IGS)对贫煤燃烧过程中的燃烧特性和SO2和NOx释放规律进行了研究。结果表明,与原煤相比,脱矿物质煤的着火温度和燃烬温度有所降低;O2/CO2气氛下,原煤和脱矿物质煤的着火温度和燃烬温度都升高,说明当O2浓度为20%时,空气气氛比O2/CO2气氛更易于着火和燃烬。此外,与长治原煤相比,脱矿物质煤在相同条件下燃烧时SO2的释放量明显提高,而NOx的释放量却有所降低。O2/CO2气氛下原煤燃烧时SO2浓度比空气气氛下的要高,而脱矿物质煤燃烧时释放的SO2浓度明显比空气气氛下的低;O2/CO2气氛下原煤和脱矿物质煤燃烧时释放的NOx浓度比空气气氛下燃烧时释放的NOx浓度要低。Abstract: Combustion experiments of Changzhi coal at 550-1 000℃ were conducted in a tube furnace reactor in air and O2/CO2, respectively. The XRF, XRD etc. were employed to study the characteristics of raw coal and its demineralized coal. Effects of mineral matter in coal on combustion characteristics and release of SO2 and NOx during combustion were investigated using TG-DTG and Antaris-IGS. The results show that ignition and burnout temperature of the demineralized coal are lower than those of raw coal. In O2/CO2 the ignition and burnout temperature of raw coal and the demineralized coal are all higher. This shows that when oxygen concentration is 20%, the coals are easier to ignite and burnout in air. Besides that, SO2 emission of the demineralized coal is higher than that of raw coal; while NOx emission is opposite. For raw coal, SO2 emission is higher when combusted in O2/CO2; while for the demineralized coal, it is opposite. During combustion in O2/CO2, NOx emission is always lower whether for the raw or the demineralized coal.
-
Key words:
- lean coal /
- oxy-fuel combustion /
- sulfur /
- nitrogen /
- emission characteristics
-
表 1 煤的工业分析和元素分析
Table 1 Proximate and ultimate analysis of coal samples
Sample Proximate analysis wad/% Ultimate analysis wad/% M A V FC C H N O* St CZ lean coal 0.58 32.48 12.91 54.03 58.36 2.93 0.84 2.87 1.94 CZ coal dem 1.28 2.12 13.32 83.28 86.47 3.85 1.04 2.44 2.80 *: by difference 表 2 硫的形态分析
Table 2 Forms of sulfur
Sample Content wad /% St Ss Sp So CZ lean coal 1.94 0.01 0.75 1.18 CZ coal dem 2.80 0.05 0.99 1.76 表 3 煤的灰成分分析
Table 3 Chemical composition of the coal ashes
Sample Content wad /% SiO2 Al2O3 Fe2O3 CaO MgO K2O Na2O P2O5 TiO2 CZ lean coal 58.10 34.80 3.63 4.25 0.64 1.43 0.46 <0.10 1.21 CZ coal dem 0.27 0.23 5.51 0.11 - - - - 0.75 -: not detecte 表 4 脱矿前后煤样的比表面积
Table 4 BET surface area of the coal before and after demineralization
Sample Pore volume v/(cm3·g-1·10-3) BET surface area A/(m2·g-1) Average pore diameter d/nm CZ lean coal 0.007 234 4.2 6.8 CZ coal dem 0.007 248 4.5 6.5 -
[1] 付兴民, 张玉秀, 郭战英, 刘海兵, 柳树成, 贾晋炜, 舒新前.炼焦煤尾煤热解特性及动力学研究[J].煤炭学报, 2013, 38(2):320-325. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201302026.htmFU Xing-min, ZHANG Yu-xiu, GUO Zhang-ying, LIU Hai-bing, LIU Shu-cheng, JIA Jin-wei, SHU Xin-qian. Characteristics and kinetics of the pyrolysis of coking coal tailings[J]. J China Coal Soc, 2013, 38(2):320-325. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201302026.htm [2] 陈昭睿, 煤热解过程中热解气停留时间对热解产物的影响[D]. 浙江: 浙江大学, 2015.CHEN Zhao-rui. Influence of Gas Residence Time on Product Distribution Of Coal Pyrolysis[D]. Zhejiang:Zhejiang University, 2015. [3] 陈皓侃, 李保庆, 张碧江.矿物质对煤热解和加氢热解含硫气体生成的影响[J].燃料化学学报, 1999, 27(S1):6-11. http://www.cnki.com.cn/Article/CJFDTOTAL-RLHX1999S1001.htmCHEN Hao-kan, LI Bao-qing, ZHANG bi-jiang. Effects of mineral matter on evolution of sulfur-containing gases in pyrolysis and hydropyrolysis[J]. J Fuel Chem Technol, 1999, 27(S1):6-11. http://www.cnki.com.cn/Article/CJFDTOTAL-RLHX1999S1001.htm [4] 刘艳华. 煤中氮/硫的赋存形态及其变迁规律研究[D]. 西安: 西安交通大学, 2002.LIU Yan-hua. An Investigation on the Forms and the Fates of Coal Nitrogen/Sulfur Functionalities[D]. Xi'an:Xi'an Jiaotong University, 2002. [5] 赵宗彬, 李文, 李保庆.矿物质对煤焦燃烧过程中NO释放规律的影响[J].化工学报, 2003, 54(1):100-106. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ200301027.htmZHAO Zong-bin, LI Wen, LI Bao-qing. Effect of mineral matter on release of no during coal char combustion[J]. J Chem Ind Eng, 2003, 54(1):100-106. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ200301027.htm [6] 杨志忠.煤的矿物质特征分析技术[J].电站系统工程, 1996, (2):39-41. http://www.cnki.com.cn/Article/CJFDTOTAL-DZXT199602007.htmYANG Zhi-zhong. An analysis technology of mineral substance character in coal[J]. Power Syst Eng, 1996, (2):39-41. http://www.cnki.com.cn/Article/CJFDTOTAL-DZXT199602007.htm [7] 晏蓉, 周燕陵.煤中矿物质成分影响燃烧性能的实验研究[J].热力发电, 1996, (3):33-37. http://www.cnki.com.cn/Article/CJFDTOTAL-RLFD603.006.htmYAN Rong, ZHOU Yan-ling. Effect of mineral matters on coal combustion properties[J]. Therm Power Gener, 1996, (3):33-37. http://www.cnki.com.cn/Article/CJFDTOTAL-RLFD603.006.htm [8] KOZLOWSKI M, MAES I I, WACHOWSKA H, YPERMAN J, FRANCO D V, MULLENS J, VAN POUCKE L C. Reduction of high-sulphur coal in the potassium-liquid ammonia system[J]. Fuel, 1999, 78(78):769-774. [9] SHIMIZU K, IWAMI Y, SUGANUMA A, SAITO I. Behaviour of sulfur in high-sulfur coal in a superacidic medium without gaseous hydrogen[J]. Fuel, 1997, 76(10):939-943. doi: 10.1016/S0016-2361(97)00097-5 [10] 刘艳华, 车得福, 徐通模.煤中矿物质对燃煤污染物排放特性的影响[J].燃料化学学报, 2005, 33(1):18-23. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract16508.shtmlLIU Yan-hua, CHE De-fu, XU Tong-mo. Effects of minerals on pollutant emission during coal combustion[J]. J Fuel Chem Technol, 2005, 33(1):18-23. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract16508.shtml [11] 黄瀛华, 王曾辉, 杭月珍.煤化学及工艺学实验[M].上海:华东化工学院出版社, 1988.HUANG Ying-hua, WANG Zeng-hui, HANG Yue-zhen. Experiment of Coal Chemistry and Technology[M]. Shanghai:East China School of Chemical Engineering Press, 1988. [12] 马志斌, 白进, 程芳琴. 高温弱还原气氛下煤灰中矿物质的定量研究[J]. 燃料化学学报, 2016, 44(6): 641-647.MA Zhi-bin, BAI Jin, LI Wen, CHENG Fang-qin.Quantitative analysis of mineral matters in coal ash under reducing atmosphere at high temperature[J]. J Fuel Chem Technol, 2016, 44(6):641-647. [13] 郑庆荣, 曾凡桂, 张世同.中变质煤结构演化的FT-IR分析[J].煤炭学报, 2011, 36(3):481-486. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201103025.htmZHEN Qing-rong, ZENG Fan-gui, ZHANG Shi-tong. FT-IR study on structure evolution of middle maturate coals[J]. J China Coal Soc, 2011, 36(3):481-486. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201103025.htm [14] 周莉, 于亨达, 杨海涛, 韩朝龙.兴安矿深部软岩的扫描电镜-X射线能谱分析[J].黑龙江科技大学学报, 2012, (2):148-153. http://www.cnki.com.cn/Article/CJFDTOTAL-HLJI201202012.htmZHOU Li, YU Heng-da, YANG Hai-tao, HAN Chao-long. SEM-EDS analysis of deep soft rock for Xing'an mine[J]. J Heilongjiang Univ Sci Technol, 2012, (2):148-153. http://www.cnki.com.cn/Article/CJFDTOTAL-HLJI201202012.htm [15] 蒋松, 动力煤配混特性及其矿物质对配煤燃烧特性的影响[D]. 江苏: 中国矿业大学, 2014.JIANG Song. Properties of Power Coal Blending and the Influence of Minerals on Its Combustion Characteristics[D]. Jiangsu:China University of Mining and Technology, 2014. [16] NORMANN F, ANDERSSON K, LECKNER B, JOHNSSON F. Emission control of nitrogen oxides in the oxy-fuel process[J]. Prog Energy Combust Sci, 2009, 35(5):385-397 doi: 10.1016/j.pecs.2009.04.002 [17] 刘粉荣, 董雪松, 李文, 马青兰, 胡瑞生, 苏梅全, 李保庆.热重法研究煤的燃烧行为及其动力学模型[J].煤炭转化, 2011, 34(2):8-12. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH201102004.htmLIU Fen-rong, DONG Xue-song, LI Wen, MA Qing-lan, HU Rui-sheng, SU Mei-quan, LI Bao-qing. Combustion behavior of coals and its kinetic model studied by a thermogravimetric analysis[J]. Coal Conv, 2011, 34(2):8-12. http://www.cnki.com.cn/Article/CJFDTOTAL-MTZH201102004.htm [18] SHIMP N F, KUHN J K, HELFINSTINE R J. Determination of Forms of Sulfur in Coal[J]. Energy Sources, 1977, 32(2):93-109. https://web.anl.gov/PCS/acsfuel/preprint%20archive/Files/20_2_PHILADELPHIA_04-75_0099.pdf [19] TAN Y W, CROISET E, DOUGLAS M A, THAMBIMUTHU K V. Combustion characteristics of coal in a mixture of oxygen and recycled flue gas[J]. Fuel, 2006, 85(4):507-512. doi: 10.1016/j.fuel.2005.08.010 [20] YANG Z Z, ZHANG Y Y, LIU L L, WANG X D, ZHANG Z T. Environmental investigation on co-combustion of sewage sludge and coal gangue:SO2, NOx and trace elements emissions.[J]. Waste Manag, 2016, 50:213-221. doi: 10.1016/j.wasman.2015.11.011 -





 下载:
下载: