-
摘要: 通过液相离子交换法制备了Cu(Ⅰ)Y、NiY、CeY分子筛,以XRD、低温N2吸附-脱附、NH3-TPD、吡啶红外Py-FTIR等方法对其进行物性表征。利用固定床技术、WK-2D微库伦仪及硫化学发光检测GC-SCD色谱评价了改性分子筛对于硫含量300 μg/g模拟油(含硫化合物二丙硫醚、环己硫醇和1-庚硫醇与壬烷配制)及HDS汽油的吸附脱硫性能。结果表明,吸附剂对模拟油和HDS燃料油品中硫醇硫醚具有吸附效果,且改性后的分子筛在吸附脱硫过程中,强的B酸对于吸附脱硫有负作用,会使油品中硫醇硫醚发生催化反应,聚合的大分子硫化物遮盖及阻塞吸附活性位点,从而使吸附剂不能够完全地吸附硫化物,造成吸附硫容较小,而弱L酸无催化活性对吸附脱硫有正面影响。Abstract: Samples of Cu (Ⅰ) Y, NiY and CeY were prepared by modifying a NaY type zeolite with a liquid ion-exchange method and characterized by means of XRD, ICP-MS, pyridine Py-FTIR, NH3-TPD and low temperature N2 adsorption-desorption.Use of micro coulombmeter WK-2D, sulfide luminescence detecting chromatography GC-SCD and fixed bed technology to evaluate the desulfurization performance of the samples for a 300 μg/g sulfur content model oil (dipropyl sulfide, cyclohexanol mercaptan and 1-heptyl mercaptan are prepared with nonane, etc.) and a hydrodesulfurization (HDS) gasoline. Results show that Cu (Ⅰ) Y has good adsorption desulfurization performance on those different oils. In the liquid ion-exchange process, the modified Y zeolite's surface acidity has greatly changed. The strong surface Bronsted (B) acidity of modified Y zeolite has negative effects on adsorption desulfurization through catalyzing mercaptan and thioether to form bigger molecules, which cover the surface of Y zeolite and block the adsorption activity sites, resulting in lower sulfur adsorption capacity of the catalyst. The weak surface Lewis (L) acidity has positive effects on adsorption desulfurization.
-
Key words:
- Y zeolite /
- mercaptan /
- sulfide /
- adsorption desulfurization /
- catalyze /
- surface acidity
-
图 4 HDS油经吸附剂处理前后的GC-SCD-1谱图
Figure 4 GC-SCD-1 chromatograms of HDS oil treated by different adsorbents
A: C1-3-thiol; B: thiophene(TP); C: 2-methylthiophene(2-MTP); D: 3-methylthiophene(3-MTP); E: C5-thiol; F: 2, 5-dimethylthiophene(2, 5-DMTP); G: 2, 3-dimethylthiophene(2, 3-DMTP); H: di-n-propylsulfide; I: cyclohexanethiol; J: 2-ethyl-5-methylthiophene(2-E-5-MDMTP); K: heptanethiol; L: C4-thiophene
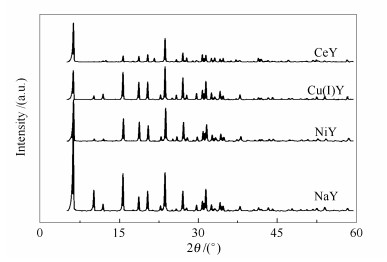
表 1 改性Y型分子筛的结构参数
Table 1 Textural parameters of the samples
Sorbent Exchange degree of Na+ vp/(cm3·g-1) A/(m2·g-1) Average pore diameter d/nm NaY - 0.360 874 0.70 CeY 91 0.307 607 0.68 NiY 74 0.333 654 0.69 Cu(Ⅰ)Y 68 0.336 721 0.69 表 2 改性金属Y对不同模拟油的静态脱除
Table 2 Desulfurization capacity of the modified Y zeolites for different model oils
Sorbent η/(mg·g-1) di-n-propyl sulfide cyclohexanethiol 1-heptanethiol NaY 2.95 2.92 2.94 CeY 13.617 10.85 15.86 NiY 15.72 17.86 25.34 Cu(Ⅰ)Y 46.783 27.38 39.07 表 3 HDS燃料油中主要硫化物的含量及脱除率
Table 3 Contents and desulfurization rate of main sulfur compounds in the HDS gasoline
Retention time t/min Sulfur compound Sulfur compound Desulfurization rate η/% NaY NiY Cu(Ⅰ)Y CeY 3.778-4.562 C1-3 sulfide 3.9 55.7 78.4 100 100 5.60 TP 2.0 22.5 16.6 63.4 42.7 9.16 C5-sulfide 3.1 21.9 98.2 100 99.1 9.76 2-MTHF 0.7 55.9 100 100 100 10.77 2, 5-DMTP 3.9 13.7 14.1 46.2 43.1 11.21 di-n-propylsulfide 1.0 14.6 67.3 97.6 100 14.10 cyclohexanethiol 1.1 12.7 80.1 97.2 100 14.70 2-E-5-MDMTP 5.3 16.7 15.6 18.8 20.2 18.02 heptanethiol 2.3 10.2 98.1 100 100 表 4 改性分子筛动态穿透硫容
Table 4 Adsorptive sulfur capacity of the modified Y zeolites at the breakthrough point of 1 μg/g
Sorbent Cs/(mg·g-1) thiophene di-n-propyl sulfide cyclohexanethiol 1-heptanethiol NaY 2.15 2.41 2.34 2.44 CeY 3.66 11.15 8.67 12.49 NiY 7.66 13.22 7.09 21.07 Cu(Ⅰ)Y 15.43 25.05 13.54 22.66 -
[1] 张玉芬, 申宝剑.硫醇转化催化剂及硫醇氧化反应机理研究进展[J].工业催化, 2009, 17(10):7-11. doi: 10.3969/j.issn.1008-1143.2009.10.002ZHANG Yu-fen, SHEN Bao-jian. Latests study on mercaptan conversion cataiysts and mechenism for mercaptan oxidan[J]. Ind Catal, 2009, 17(10):7-11. doi: 10.3969/j.issn.1008-1143.2009.10.002 [2] 高立达, 薛青松, 路勇, 何鸣元.汽油中正戊硫醇的催化空气氧氧化脱除I.CuZnAI复合氧化物催化剂的催化活性[J].催化学报, 2008, 29(7):633-637. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=chua200807010&dbname=CJFD&dbcode=CJFQGAO Li-da, XUE Qing-song, LU Yong, HE Ming-yuan. Aerobic oxidation removal of 1-pentanethiol from gasoline1. Catalytic activity of CuZnAl composite oxide[J].Chin J Catal, 2008, 29(7):633-637. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=chua200807010&dbname=CJFD&dbcode=CJFQ [3] KHARASCH M S, NUDENBERG W, MANTELL G J. Reactions of atoms and free radicals in solution. XXY. The reactions of olefins with mercaptans in the presence of osygen[J]. J Org Chem, 1951, 16(4):524-532. doi: 10.1021/jo01144a005 [4] 田高友, 吕志凤, 战风涛, 夏道宏.硫醇的氧化行为及其对催化裂化柴油安定性的影响[J].石油学报(石油加工), 2002, 18(6):67-72. http://industry.wanfangdata.com.cn/dl/Detail/Periodical?id=...TIAN Gao-you, LU Zhi-feng, ZHAN Feng-tao, XIA Dao-hong. The oxidation of thiols and its effects on the stability of fludized catalytic cracked diesel[J]. Acta Pet Sin(Pet Proce Sect), 2002, 18(6):67-72. http://industry.wanfangdata.com.cn/dl/Detail/Periodical?id=... [5] SHIH S S, OWEN S P J. Mobils OCTGAINTM process:FCC gasoline desulfurization reaches a new performance level//1999 NPRA AnnuaI meeting[C]. San Antonio, Texas, 1999:AM-9-30. [6] ITOE, VAN VEEN RJ A. On novel processes for removing sulphur from refinery streams[J]. Catal Today, 2006, 116(4):446-460. doi: 10.1016/j.cattod.2006.06.040 [7] 于宏超, 张士国, 卞贺, 夏道宏. FCC汽油中己硫醇的生成机理[J].石油学报(石油加工), 2011, 27(3):381-386. http://d.wanfangdata.com.cn/Periodical/syxb-syjg201103009YU Hong-chao, ZHANG Shi-guo, BIAN He, XIA Dao-hong. Theoretical study on the formation of hexanethiol in FCC gasoline[J]. Acta Pet Sin(Pet Proce Sect), 2011, 27(3):381-386. http://d.wanfangdata.com.cn/Periodical/syxb-syjg201103009 [8] 申志兵, 柯明, 宋昭峥, 蒋庆哲.硫醚化脱除FCC汽油中硫醇和二烯烃研究进展[J].化学工业与工程, 2010, 27(6):544-550. http://www.wenkuxiazai.com/doc/5f6eedd7941ea76e58fa04c3-2.htmlSHEN Zhi-bing, KE Ming, SONG Zhao-zheng, JIANG Qing-zhe. Review for thioetherification in reducing mercaptan and diene of FCC gasoline[J]. Chem Ind Eng, 2010, 27(6):544-550. http://www.wenkuxiazai.com/doc/5f6eedd7941ea76e58fa04c3-2.html [9] MA X L, SUBRAMANI V, JAE H K, SONG C S. Deep desulfurjzation of gasoline by selective adsorption over solid adsorbents and impact of analytical methods on ppm-evel sulfur quantification for fuel cell applications[J]. Appl Catal B:Environ, 2005, 56(1/2):l37-147. https://ar.scribd.com/document/36647107/gulfpub-hp-201009 [10] SHIRAISHI Y, NALTO T, HIRA I T. Vanadosilicate molecular sieve as a catalyst for oxidative desulfurization of light oil[J]. Ind Eng Chem Res, 2003, 42(15):6034-6039. https://www.researchgate.net/publication/231371258_Vanadosilicate... [11] SHOREY S W. Exploiting synergy between FCC and feed pretreating units to improve refinery margins and produce low-sulfur fuels//1999 NPRA Annual Meeting[C]. San Antonio, Texas, 1999:AM-99-55. [12] 唐克, 宋丽娟, 段林海, 李秀奇, 桂建舟, 孙兆林.杂原子Y分子筛的二次合成及其吸附脱硫性能[J].物理化学学报, 2006, 22(9):1116-1120. http://www.cqvip.com/qk/92644X/200609/23046291.htmlTANG Ke, SONG Li-juan, DUAN Lin-hai, LI Xiu-qi, GUI Jian-zhou, SUN Zhao-lin. Deep desulfurization by selective adsorption on heteroatom zeolite prepared by secondary synthesis[J]. Acta Phys-Chim Sin, 2006, 22(9):1116-1120. http://www.cqvip.com/qk/92644X/200609/23046291.html [13] 董世伟, 秦玉才, 阮艳军, 王源, 宇文广, 张磊, 范跃超, 宋丽娟.改性Y型分子筛对FCC汽油脱硫性能的研究[J].燃料化学学报, 2013, 41(3):341-346. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18147.shtmlDONG Si-wei, QIN Yu-cai, RUAN Yan-jun, WANG Yuan, YU Wen-guan, ZHANG Lei, FAN Yue-chao, SONG Li-juan. Performance of adsorptive desulfurization for FCC gasoline over modified Y zeolites[J]. J Fuel Chem Technol, 2013, 41(3):341-346. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18147.shtml [14] 董世伟, 秦玉才, 王源, 宋丽娟.汽油选择性吸附脱硫过程中硫化物吸附脱除规律研究[J].石油炼制与化工, 2013, 44(3):26-30. https://www.wenkuxiazai.com/doc/9aa6c64859eef8c75ebfb31f...DONG Shi-wei, QIN Yu-cai, WANG Yuan, SONG Li-juan. Study on the removal of sulfer compounds in gasoline by selective desulferization process[J]. Pet Proce Petroche, 2013, 44(3):26-30. https://www.wenkuxiazai.com/doc/9aa6c64859eef8c75ebfb31f... [15] KIM J H, MA X L, ZHOU A A, SONG C S. Ultra-deep desulfurization and denitrogenation of diesel fuel by selective adsorption over three different adsorbents:A study on adsorptive selectivity and mechanism[J]. Catal Today, 2006, 111:74-83. doi: 10.1016/j.cattod.2005.10.017 [16] 鞠秀芳, 王洪国, 徐静, 靳玲玲, 宋丽娟.噻吩、苯在Ce(Ⅳ)Y分子筛上的吸附行为[J].石油学报(石油加工), 2009, 25(5):655-660. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=sxjg200905008&dbname=CJFD&dbcode=CJFQJU Xiu-fang, WANG Hong-guo, XU Jing, JI Ling-ling, SONG Li-juan. Thiophene and benzene adsorption on CeY zeolites[J]. Acta Pet Sin(Pet Process Sect), 2009, 25(5):655-660. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=sxjg200905008&dbname=CJFD&dbcode=CJFQ [17] JACOBS P A, UYTTERHOEVEN, J B, BEYER H K. Some unusual properties of activated and reduced AgNaA zeolites[J]. J Chem Soc Faraday Trans., 1979, 175:56-64. https://core.ac.uk/display/34346936 [18] 段林海, 孟秀红, 施岩, 鞠秀芳, 靳玲玲, 徐静, 陈晓陆, 宋丽娟.不同硫化物在改性Y分子筛上的选择性吸附脱硫性能及机理[J].石油学报(石油加工), 2009, 增刊:70-76. http://www.wenkuxiazai.com/doc/dbe5524fe518964bcf847c0c-2.htmlDUAN Lin-hai, MENG Xiu-hong, SHI Yan, JU Xiu-fang, JIN Ling-ling, XU Jing, CHEN Xiao-lu, SONG Li-juan. Selective adsorption desulfurization performance and mechanism of sulfur compounds over modified Y zeolite[J]. Acta Pet Sin(Pet Process Sect), 2009, 25(Suppl):s70-s76. http://www.wenkuxiazai.com/doc/dbe5524fe518964bcf847c0c-2.html [19] 陈晓陆, 孙兆林, 王洪国, 王阔, 宋丽娟.基于分子模拟技术的Cu(Ⅰ)Y分子筛对硫化物及烯烃选择性吸附行为的分析[J].石油学报(石油加工), 2010, 26(1):82-86. http://industry.wanfangdata.com.cn/dl/Detail/Periodical?id=...CHEN Xiao-lu, SUN Zhao-lin, WANG Hong-guo, WANG Kuo, SONG Li-juan. Molecular simulation studies of selective adsorption behaviour of sulphur and olefin compounds on Cu(Ⅰ)Y zeolite[J]. Acta Pet Sin(Pet Process Sect), 2010, 26(1):82-86. http://industry.wanfangdata.com.cn/dl/Detail/Periodical?id=... [20] SONG C S, MA X L.New design approaches to ultra-clean diesel fuels by deep desulfurization and dearomatization[J]. App Cata B:Environ, 2003, 41:207-238. doi: 10.1016/S0926-3373(02)00212-6 [21] HERNANDEZ-MALDONADO A J, YANG R T. Desulfurization of commercial jet fuels by adsorption via π-complexation with vapor phase ion exchanged Cu(Ⅰ)-Y zeolites[J]. Ind Eng Chem Res, 2004, 43:6142-6149. doi: 10.1021/ie049533x [22] VELU S, MA X L, SONG C S. Selective adsorption for rmoving sulfur from jet fuel over zeolite-based adsorbents[J]. Ind Eng Chem Res, 2003, 42(21):5293-5304. doi: 10.1021/ie020995p -





 下载:
下载: