-
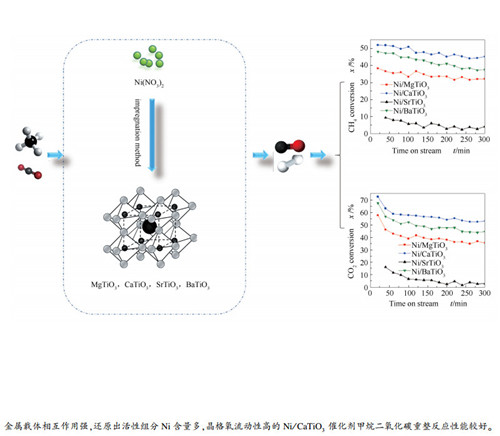
摘要: 采用浸渍法制备了一系列MTiO3(M=Mg、Ca、Sr、Ba)钙钛矿型氧化物负载的Ni催化剂(Ni的负载量为5%,质量分数),通过XRD、氮吸附、H2-TPR、CO2-TPD、XPS和TG等技术对催化剂进行了表征,对其甲烷二氧化碳重整反应的催化性能进行了研究。结果表明,M为不同碱土金属时,催化剂上金属载体相互作用、活性组分的表面原子浓度以及催化剂晶格氧的流动性都发生了变化。Ni/CaTiO3催化剂上金属载体相互作用较强,还原出的活性组分Ni的含量较多,晶格氧流动性较高,因而具有较好的催化性能。SrTiO3载体颗粒粒径较大,Ni/SrTiO3催化剂上Ni的分散度不高,金属载体的相互作用较弱,表面Ni原子相对含量较低,晶格氧的流动性较差,其甲烷二氧化碳重整反应活性也最低。Abstract: A series of MTiO3 (M=Mg, Ca, Sr, Ba) supported Ni catalysts (with a Ni loading of 5%) for methane reforming with carbon dioxide (DRM) were prepared by the impregnation method. The Ni/MTiO3 catalysts were characterized by XRD, N2 sorption, H2-TPR, CO2-TPD, XPS and TG; the effect of alkaline earth metals (M) on the catalytic performance of Ni/MTiO3 in the DRM was then investigated. The results indicate that the metal-support interaction, the surface Ni atomic concentration and the mobility of lattice oxygen species on the Ni/MTiO3 catalysts are related to the alkaline earth metal M used in the MTiO3 supports. The Ni/CaTiO3 catalyst shows superior performance in DRM to other catalysts, which is ascribed to the strong metal-support interaction, large amount of reduced active Ni and relatively high mobility of lattice oxygen species. In contrast, the SrTiO3 support has a relatively large particle size, leading to poor Ni dispersion on the Ni/SrTiO3 catalyst as well as weak metal-support interaction, low lattice oxygen mobility, and less surface active Ni atoms; as a result, the Ni/SrTiO3 catalyst exhibits relatively poor performance in DRM.
-
Key words:
- perovskite /
- alkaline earth metal /
- methane /
- carbon dioxide /
- carbon deposition
-
表 1 钙钛矿载体及负载催化剂的织构性质
Table 1 Textural properties of the perovskite supports and supported Ni catalysts
Sample Surface area
A/(m2·g-1)Pore volume
v/(cm3·g-1)Crystalline
sizea /nmSample Surface area
A/(m2·g-1)Pore volume
v/(cm3·g-1)MgTiO3 4.98 0.0106 44.0 Ni/MgTiO3 6.05 0.0190 CaTiO3 3.65 0.0049 34.0 Ni/CaTiO3 3.05 0.0065 SrTiO3 3.53 0.0048 53.2 Ni/SrTiO3 4.04 0.0070 BaTiO3 2.56 0.0039 34.6 Ni/BaTiO3 3.20 0.0067 a: calculated by Scherrer equation according to the XRD results 表 2 还原后的Ni/MTiO3(M=Mg、Ca、Sr、Ba)的催化剂的XPS拟合
Table 2 XPS fitting results of the reduced Ni/MTiO3 (M=Mg, Ca, Sr, Ba) catalysts
Sample Lattice oxygen Ni elemental
composition /%EB/eV area /% Ni/MgTiO3 529.60 46.20 6.77 Ni/CaTiO3 529.27 48.26 6.90 Ni/SrTiO3 528.96 43.89 6.48 Ni/BaTiO3 529.01 44.77 7.45 -
[1] NEJAT P, JOMEHZADEH F, TAHERI M M, GOHARI M, ABD MAJID M Z. A global review of energy consumption, CO2 emissions and policy in the residential sector (with an overview of the top ten CO2 emitting countries)[J]. Renewable Sustainable Energy Rev, 2015, 43:843-862. http://www.sciencedirect.com/science/article/pii/S1364032114010053 [2] 王辉, 张俊峰, 白云星, 王文峰, 谭猗生, 韩怡卓. NiO@SiO2核壳催化剂在浆态床中低温甲烷化研究[J].燃料化学学报, 2016, 44(5):548-556. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18827.shtmlWANG Hui, ZHANG Jun-feng, BAI Yun-xing, WANG Wen-feng, TAN Yi-sheng, HAN Yi-zhuo. NiO@SiO2 core-shell catalyst for low-temperature methanation of syngas in slurry reactor[J]. J Fuel Chem Technol, 2016, 44(5):548-556. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18827.shtml [3] 石广强, 李君华, 刘宇航, 武则龙.甲烷间接转化制合成气的研究进展[J].天津化工, 2015, 29(4):1-3. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=tjhg201504001SHI Guang-qiang, LI Jun-hua, LIU Yu-hang, WU Ze-long. Research progress on indirect conversion of methane to produce synthesis gas[J]. Tianjin Chem Ind, 2015, 29(4):1-3. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=tjhg201504001 [4] 张小平.介孔Ni/ZrO2催化剂的制备及甲烷二氧化碳重整催化性能[J].山东化工, 2019, 48(10):43-45. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=sdhg201910017ZHANG Xiao-ping. Preparation of mesoporous Ni/ZrO2 catalysts and catalytic performance for CO2 reforming of CH4[J]. Shandong Chem Ind, 2019, 48(10):43-45. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=sdhg201910017 [5] HERMAN R G, SUN Q, SHI C. Development of active oxide catalysts for the direct oxidation of methane to formaldehyde[J]. Catal Today, 1997, 37(1):1-14. http://www.sciencedirect.com/science/article/pii/S0920586196002568 [6] XU J, ZHANG Y, XU X, FANG X, XI R, LIU Y, ZHENG R, WANG X. Constructing La2B2O7 (B=Ti, Zr, Ce) compounds with three typical crystalline phases for oxidative coupling of methane:The effect of phase structures, super oxide anions and alkalinity on the reactivity[J]. ACS Catal, 2019, 9(5):4030-4045. doi: 10.1021/acscatal.9b00022 [7] GUO X, FANG G, LI G, MA H, FAN H, YU L, MA C, WU X, DENG D, WEI M. Direct, nonoxidative conversion of methane to ethylene, aromatics, and hydrogen[J]. Science, 2014, 344(2):616-619. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=175ba9855a651d38b9a5f2eafe15cb15 [8] ROSTRUP-NIELSEN J R. Sulfur-passivated nickel catalysts for carbon-free steam reforming of methane[J]. J Catal, 1984, 85(1):31-43. http://www.sciencedirect.com/science/article/pii/0021951784901076 [9] XIAO Z, LI Y, HOU F, WU C, PAN L, ZOU J, WANG L, ZHANG X, LIU G, LI G. Engineering oxygen vacancies and nickel dispersion on CeO2 by Pr doping for highly stable ethanol steam reforming[J]. Appl Catal B:Environ, 2019, 258:117940. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=2c1f0ab65c9e7fff0b5c13a83fb20e19 [10] YANG J H, YOON Y, RYU M, AN S K, SHIN J, LEE C J, Integrated hydrogen liquefaction process with steam methane reforming by using liquefied natural gas cooling system[J]. Appl Energy, 2019, 255:113840. http://www.researchgate.net/publication/332957134_Analysis_and_assessment_of_partial_re-liquefaction_system_for_liquefied_hydrogen_tankers_using_liquefied_natural_gas_LNG_and_H2_hybrid_propulsion [11] DI GIULIANO A, GALLUCCI K, GIANCATERINO F, COURSON C, FOSCOLO P U. Multicycle sorption enhanced steam methane reforming with different sorbent regeneration conditions:Experimental and modelling study[J]. Chem Eng J, 2019, 377:119874. http://www.sciencedirect.com/science/article/pii/S1385894718317455 [12] MA Y, MA Y, LONG G, LI J, HU X, YE Z, WANG Z, BUCKLEY C E, DONG D, Synergistic promotion effect of MgO and CeO2 on nanofibrous Ni/Al2O3 catalysts for methane partial oxidation[J]. Fuel, 2019, 258: 116103. https://www.researchgate.net/publication/337662135_Synergistic_promotion_effect_of_MgO_and_CeO2_on_nanofibrous_NiAl2O3_catalysts_for_methane_partial_oxidation [13] KIM S, CRANDALL B S, LANCE M J, CORDONNIER N, LAUTERBACH J, SASMAZ E. Activity and stability of NiCe@SiO2 multi-yolk-shell nanotube catalyst for tri-reforming of methane[J]. Appl Catal B:Environ, 2019, 259:118037. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=b145f3506a119f3ea17b530421a53b6c [14] ZHANG X, ZHANG M, ZHANG J, ZHANG Q, TSUBAKI N, TAN Y, HAN Y. Methane decomposition and carbon deposition over Ni/ZrO2 catalysts:Comparison of amorphous, tetragonal, and monoclinic zirconia phase[J]. Int J Hydrogen Energy, 2019, 44(33):17887-17899. http://www.zhangqiaokeyan.com/academic-journal-foreign_other_thesis/0204112987058.html [15] ZHAO Y, KANG Y, LI H, LI H. CO2 conversion to synthesis gas via DRM on the durable Al2O3/Ni/Al2O3 sandwich catalyst with high activity and stability[J]. Green Chem, 2018, 20(12):2781-2787. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6a1d429f40b3005a8f668339bebda088 [16] QIN Z, CHEN J, XIE X, LUO X, SU T, JI H. CO2 reforming of CH4 to syngas over nickel-based catalysts[J]. Environ Chem Lett, 2020, 18(4):997-1017. doi: 10.1007/s10311-020-00996-w [17] ZHANG M, ZHANG J, WU Y, PAN J, ZHANG Q, TAN Y, HAN Y. Insight into the effects of the oxygen species over Ni/ZrO2 catalyst surface on methane reforming with carbon dioxide[J]. Appl Catal B:Environ, 2019, 244:427-437. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=9f75f7017a22bdee905db6cffd8a1ee9 [18] YENTEKAKIS I V, GOULA G, HATZISYMEON M, BETSI-ARGYROPOULOU I, BOTZOLAKI G, KOUSI K, KONDARIDES D I, TAYLOR M J, PARLETT C. M. A, OSATIASHTIANI A, KYRIAKOU G, HOLGADO J P, LAMBERT R M. Effect of support oxygen storage capacity on the catalytic performance of Rh nanoparticles for CO2 reforming of methane[J]. Appl Catal B:Environ, 2019, 243:490-501. http://www.sciencedirect.com/science/article/pii/S092633731831004X [19] LIU C, YE J, JIANG J, PAN Y. Progresses in the preparation of coke resistant Ni-based catalyst for steam and CO2 reforming of methane[J]. Chem Cat Chem, 2011, 3(3):529-541. doi: 10.1002/chin.201127237 [20] ARBAG H, YASYERLI S, YASYERLI N, DOGU G. Activity and stability enhancement of Ni-MCM-41 catalysts by Rh incorporation for hydrogen from dry reforming of methane[J]. Int J Hydrogen Energy, 2010, 35(6):2296-2304. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=692a1e6f29207ab7993c5890af01883f [21] HU Y H, RUCKENSTEIN E. Binary MgO-based solid solution catalysts for methane conversion to syngas[J]. Catal Rev, 2002, 44(3):423-453. doi: 10.1081/CR-120005742 [22] AZIZ M.A.A, JALIL A. A, WONGSAKULPHASATCH S, VO D V N. Understanding the role of surface basic sites of catalysts in CO2 activation in dry reforming of methane:A short review[J]. Catal Sci Technol, 2020, 10(1):35-45. http://pubs.rsc.org/en/content/articlepdf/2020/cy/c9cy01519a [23] WANG S, LU G Q, MILLAR G J. Carbon Dioxide reforming of methane to produce synthesis gas over metal-supported catalysts:State of the art[J]. Energy Fuels, 1996, 10(4):896-904. doi: 10.1021/ef950227t [24] LI Z, WANG Z, JIANG B, KAWI S. Sintering resistant Ni nanoparticles exclusively confined within SiO2 nanotubes for CH4 dry reforming[J]. Catal Sci Technol, 2018, 8(13):3363-3371. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=8d66b0398c5b95c70134acd3dfee7ea1 [25] ZHANG M, ZHANG J, ZHOU Z, CHEN S, ZHANG T, SONG F, ZHANG Q, TSUBAKI N, TAN Y, HAN Y, Effects of the surface adsorbed oxygen species tuned by rare-earth metal doping on dry reforming of methane over Ni/ZrO2 catalyst[J]. Appl Catal B:Environ, 2020, 264:118522. http://www.sciencedirect.com/science/article/pii/S0926337319312688 [26] YAN Q G, WENG W Z, WAN H L, TOGHIANI H, JR C U P. Activation of methane to syngas over a Ni/TiO2 catalyst[J]. Appl Catal A:Gen, 2003, 239(1/2):43-58. http://www.sciencedirect.com/science/article/pii/S0926860X02003514 [27] AKRI M, ZHAO S, LI X, ZANG K, LEE A F, ISAACS M A, XI W, GANGARAJULA Y, LUO J, REN Y, CUI Y T, LI L, SU Y, PAN X, WEN W, PAN Y, WILSON K, LI L, QIAO B, ISHII H, LIAO Y F, WANG A, WANG X, ZHANG T. Atomically dispersed nickel as coke-resistant active sites for methane dry reforming[J]. Nat Commun, 2019, 10(1):5181. http://www.nature.com/articles/s41467-019-12843-w [28] HAYAKAWA T, SUZUKI S, NAKAMURA J, UCHIJIMA T, HAMAKAWA S, SUZUKI K, SHISHIDO T, TAKEHIRA K. CO2 reforming of CH4 over Ni/perovskite catalysts prepared by solid phase crystallization method[J]. Appl Catal A:Gen, 1999, 183(2):273-285. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=345f9468dde335dff482e1085a889d1d [29] LI X, WU M, LAI Z, HE F. Studies on nickel-based catalysts for carbon dioxide reforming of methane[J]. Appl Catal A:Gen, 2005, 290(1/2):81-86. http://www.sciencedirect.com/science/article/pii/S0926860X0500390X [30] GAO N, CHENG M, QUAN C, ZHENG Y. Syngas production via combined dry and steam reforming of methane over Ni-Ce/ZSM-5 catalyst[J]. Fuel, 2020, 273:117702. http://www.sciencedirect.com/science/article/pii/S0016236120306979 [31] WANG Y H, LIU H M, XU B Q. Durable Ni/MgO catalysts for CO2 reforming of methane:Activity and metal-support interaction[J]. J Mol Catal A:Chem, 2009, 299(1):44-52. http://www.sciencedirect.com/science/article/pii/S1381116908004184 [32] LÖFBERG A, GUERRERO-CABALLERO J, KANE T, RUBBENS A, JALOWIECKI-DUHAMEL L. Ni/CeO2 based catalysts as oxygen vectors for the chemical looping dry reforming of methane for syngas production[J]. Appl Catal B:Environ, 2017, 212:159-174. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=4541f58fa1cf4f5628bdadade399cb45 [33] SUTTHIUMPORN K, MANEERUNG T, KATHIRASER Y, KAWI S. CO2 dry-reforming of methane over La0.8Sr0.2Ni0.8M0.2O3 perovskite (M=Bi, Co, Cr, Cu, Fe):Roles of lattice oxygen on C-H activation and carbon suppression[J]. Int J Hydrogen Energy, 2012, 37(15):11195-11207. [34] LI R J, YU C C, JI W J, SHEN S K. Methane oxidation to synthesis gas using lattice oxygen in La1-xSrxFeO3 perovskite oxides instead of molecular oxygen[J]. Stud Surf Sci Catal, 2004, 147:199-204. http://www.sciencedirect.com/science/article/pii/S016729910480051X [35] AU C T, HU Y H, WAN H L. Methane activation over unsupported and La2O3-supported copper and nickel catalysts[J]. Catal Lett, 1996, 36(3/4):159-163. doi: 10.1007/BF00807613 [36] LIU B S, AU C T, Carbon deposition and catalyst stability over La2NiO4/γ-Al2O3 during CO2 reforming of methane to syngas[J]. Appl Catal A Gen, 2003, 244(1):181-195. http://www.sciencedirect.com/science/article/pii/S0926860X02005914 -





 下载:
下载: