Effects of activation methods on the activation of natural aluminosilicate minerals and zeolite synthesis
-
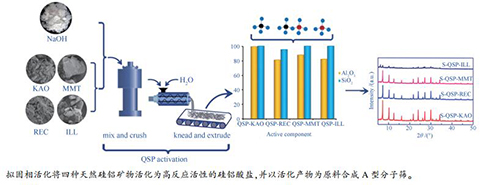
摘要: 选取了高岭土、累托土、蒙脱土和伊利石四种天然硅铝矿物,采用热活化、碱熔活化、亚熔盐活化及拟固相活化四种方法对上述四种矿物分别进行活化,对比了不同活化方法对天然硅铝矿物活化效果的影响。结果表明,亚熔盐活化及拟固相活化都具有良好的活化效果,而且能耗较低,明显优于热活化和碱熔活化。其中,拟固相活化由于能耗更低且更有利于实现工业操作,因此,是最具发展前景的天然硅铝矿物活化方法。对比四种天然硅铝矿物,高岭土、累托土及蒙脱土的晶相结构更容易被解聚,而伊利石稳定性更高,经亚熔盐活化及拟固相活化后,活化产物中也只有极少量的高反应活性的硅、铝物种,因此,伊利石不是理想的分子筛合成原料。Abstract: Four natural aluminosilicate minerals including kaolin, rectorite, montmorillonite and illite were activated by thermal activation, alkali fusion activation, sub-molten salt activation and quasi-solid-phase activation method, respectively. Comparing the activation effects of the four methods, it is found that both sub-molten salt method and quasi-solid-phase method present better activation effect at lower energy consumption, which are obviously superior to the other two activation methods. The quasi-solid-phase activation method is the most promising method for activating the natural aluminosilicate minerals due to its much lower energy consumption and more feasible industrial operation. Comparatively, the framework structure of kaolinite rectorite and montmorillonite are relatively more easily to be depolymerized, the structure of illite is much stable, after being sub-molten salt activated and quasi-solid-phase activated, the resulted illite has little active SiO2 and Al2O3 which can be used to synthesize zeolites due to their high chemical reactivity. Therefore, different from the other three natural aluminosilicate minerals, illite is not an ideal raw material for zeolite synthesis.
-
图 8 以不同拟固相活化天然硅铝矿物为原料所得合成产物的XRD谱图
Figure 8 XRD patterns of the samples from different QSP-activated aluminosilicate minerals(S-QSP-KAO, S-QSP-REC, S-QSP-MMT-1 and S-QSP-ILL-1 are the samples obtained without supplemental alumina sources; S-QSP-MMT-2 and S-QSP-ILL-2 are the samples obtained with supplemental alumina sources)
表 1 不同天然硅铝矿物的化学组成
Table 1 Chemical composition of the different natural aluminosilicate minerals
Natural mineral Component w/% SiO2/Al2O3
(molar ratio)Na2O MgO Al2O3 SiO2 P2O5 K2O CaO TiO2 Fe2O3 KAO 0.3 0.1 44.2 52.7 0.4 0.6 0.2 0.3 0.3 2.03 REC 1.8 0.6 39.0 48.5 0.3 0.8 0.8 2.4 2.2 2.11 MMT 0.1 0.4 40.1 55.8 0.04 2.3 - - 1.1 2.37 ILL 1.6 0.3 20.8 72.9 0.03 3.7 - - 0.6 5.96 -
[1] LIU Y, LIN C X, WU Y G. Characterization of red mud derived from a combined bayer process and bauxite calcination method[J]. J Hazard Mater, 2007, 146(1):255-261. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=b181a8cbe09790f3e0a6c4555eaddcdd [2] ANDERSON M W, HOLMES S M, HANIF N, CUNDY C S. Hierarchical pore structures through diatom zeolitization[J]. Angew Chem Int Ed, 2000, 39(15):2707-2710. doi: 10.1002/1521-3773(20000804)39:15<2707::AID-ANIE2707>3.0.CO;2-M [3] LI T S, LIU H Y, FAN Y, YUAN P, SHI G, BI X T, BAO X J. Synthesis of zeolite Y from natural aluminosilicate minerals for fluid catalytic cracking application[J]. Green Chem, 2012, 14(12):3255-3259. doi: 10.1039/c2gc36101a [4] YUE Y Y, LIU H Y, YUAN P, LI T S, YU C, BI H, BAO X J. From natural aluminosilicate minerals to hierarchical ZSM-5 zeolites:A nanoscale depolymerization-reorganization approach[J]. J Catal, 2014, 319:200-210. doi: 10.1016/j.jcat.2014.08.009 [5] DING J J, LIU H Y, YUAN P, SHI G, BAO X J. Catalytic properties of a hierarchical zeolite synthesized from a natural aluminosilicate mineral without the use of a secondary mesoscale template[J]. ChemCatChem, 2013, 5(8):2258-2269. doi: 10.1002/cctc.201300049 [6] REYES C A R, WILLIAMS C D, ALARCON C O M. Synthesis of zeolite LTA from thermally treated kaolinite[J]. Rev Fac Ing Univ Antioquia, 2010, 53:30-41. http://cn.bing.com/academic/profile?id=cc3ea594cacde3f73c2925642d7c3718&encoded=0&v=paper_preview&mkt=zh-cn [7] CHO K, NA K, KIM J, TERASAKI O, RYOO R. Zeolite synthesis using hierarchical structure-directing surfactants:Retaining porous structure of initial synthesis gel and precursors[J]. Chem Mater, 2012, 24(14):2733-2738. doi: 10.1021/cm300841v [8] LIU H Y, SHEN T, LI T S, YUAN P, SHI G, BAO X J. Green synthesis of zeolites from a natural aluminosilicate mineral rectorite:Effects of thermal treatment temperature[J]. Appl Clay Sci, 2014, 90:53-60. doi: 10.1016/j.clay.2014.01.006 [9] LIU H Y, SHEN T, WANG W, LI T S, YUE Y Y, BAO X J. From natural aluminosilicate minerals to zeolites:Synthesis of ZSM-5 from rectorites activated via different methods[J]. Appl Clay Sci, 2015, 115:201-211. doi: 10.1016/j.clay.2015.07.040 [10] YANG J B, LIU H Y, DIAO H J, LI B S, YUE Y Y, BAO X J. A Quasi-solid-phase approach to activate natural minerals for zeolite synthesis[J]. ACS Sustainable Chem Eng, 2017, 5(4):3233-3242. doi: 10.1021/acssuschemeng.6b03031 [11] YUE Y Y, LIU H Y, YUAN P, YU C, BAO X J. One-pot synthesis of hierarchical FeZSM-5 zeolites from natural aluminosilicates for selective catalytic reduction of NO by NH3[J]. Sci Rep, 2015, 5:9270-9280. doi: 10.1038/srep09270 [12] WHITE C E, PROVIS J L, THOMAS P, RILEY D P, DEVENTER J J. Density functional modeling of the local structure of kaolinite subjected to thermal dehydroxylation[J]. J Phys Chem, 2010, 114(14):4988-4996. doi: 10.1021/jp911108d [13] RíOS C A, WILLIAMS C D, FULLEN M A. Nucleation and growth history of zeolite LTA synthesized from kaolinite by two different methods[J]. Appl Clay Sci, 2009, 42(3):446-454. http://cn.bing.com/academic/profile?id=d40d974b1c20c18ef9c8b2277e60ac04&encoded=0&v=paper_preview&mkt=zh-cn [14] LIU Y, PINNAVAIA T J. Metakaolin as a reagent for the assembly of mesoporous aluminosilicates with hexagonal, cubic and wormhole framework structures from proto-faujasitic nanoclusters[J]. J Mater Chem, 2004, 14(23):3416-3420. doi: 10.1039/b410337h [15] SAIKIA N. Characterization, beneficiation and utilization of a kaolinite clay from Assam, India[J]. Appl Clay Sci, 2003, 24(1/2):93-103. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=d6b7416e2d1df83cae115f0e2ccbc7b7 [16] WEI B Y, LIU H Y, LI T S, CAO L Y, FAN Y, BAO X J. Natural rectorite mineral:A promising substitute of kaolin for in-situ synthesis of fluid catalytic cracking catalysts[J]. AIChE J, 2010, 56(11):2913-2922. doi: 10.1002/aic.12195 [17] GARRELS R M. Montmorillonite/illite stability diagrams[J]. Clays Clay Miner, 1984, 32(3):161-166. doi: 10.1346/CCMN.1984.0320301 [18] HE C L, MAKOVICKY E, ØSBØCK B. Thermal stability and pozzolanic activity of calcined illite[J]. Appl Clay Sci, 1995, 9(5):337-354. doi: 10.1016/0169-1317(94)00033-M [19] OKADA K, KAMESHIMA Y, YASUMORI A. Chemical shifts of silicon X-ray photoelectron spectra by polymerization structures of silicates[J]. J Am Ceram Soc, 1998, 81(7):1970-1972. http://cn.bing.com/academic/profile?id=afe634467c4b53134560999f38f6c722&encoded=0&v=paper_preview&mkt=zh-cn [20] BARR T L, SEAL S, WOZNIAK K, KLINOWSKI J. ESCA studies of the coordination state of aluminium in oxide environments[J]. J Chem Soc Faraday Trans, 1997, 93(1):181-186. doi: 10.1039/a604061f [21] LIPPMAA E, MÄGI M, SAMOSON A, ENGELHARDT G, GRIMMER A. Structural studies of silicates by solid-state high-resolution silicon-29 NMR[J]. J Am Chem Soc, 1980, 102(15):4889-4893. doi: 10.1021/ja00535a008 [22] BERTERMANN R, KROGER N, TACKE R. Solid-state 29Si MAS NMR studies of diatoms:Structural characterization of biosilica deposits[J]. Anal Bioanal Chem, 2003, 375(5):630-634. doi: 10.1007/s00216-003-1769-5 [23] MADANI A, AZNAR A, SANZ J, SERRATOSA J. 29Si and 27Al NMR study of zeolite formation from alkali-leached kaolinites:Influence of thermal preactivation[J]. J Phys Chem, 1990, 94(2):760-765. doi: 10.1021/j100365a046 [24] KOUASSI S, ANDJI J, BONNET J, ROSSIGNOL S. Dissolution of waste glasses in high alkaline solutions[J]. Ceram Silik, 2010, 54(3):235-240. https://www.irsm.cas.cz/materialy/cs_content/2010/Kouassi_CS_2010_0000.pdf [25] ENGELHARDT G. Silicon-29 NMR of Solid Silicates[M]. New York:John Wiley & Sons Ltd, 2007:8-36. [26] ROCHA J, KLINOWSKI J. 29Si and 27Al magic-angle-spinning NMR studies of the thermal transformation of kaolinite[J]. Phys Chem Miner, 1990, 17(2):179-186. doi: 10.1007/BF00199671 [27] MüLLER D, GESSNER W, BEHRENS H J, SCHELER G. Determination of the aluminium coordination in aluminium-oxygen compounds by solid-state high-resolution 27Al NMR[J]. Chem Phys Lett, 1981, 79(1):59-62. doi: 10.1016/0009-2614(81)85288-8 [28] CHANDRASEKHAR S. Influence of metakaolinization temperature on the formation of zeolite 4A from kaolin[J]. Clay Miner, 1996, 31(2):253-261. doi: 10.1180/claymin.1996.031.2.11 -





 下载:
下载: