Preparation of honeycombed holmium-modified Fe-Mn/TiO2 catalyst and its performance in the low temperature selective catalytic reduction of NOx
-
摘要: 采用模压法制备了蜂窝状Ho改性的Fe-Mn/TiO2催化剂,研究了结构助剂、黏合剂和造孔剂等对成型催化剂低温选择催化还原(SCR)脱硝性能的影响。优选出一套理想的成型参数:水粉质量比为40%且逐次分批加入;结构助剂玻璃纤维的用量为10%(质量分数);黏合剂羧甲基纤维素的用量为5%(质量分数);助挤剂甘油的添加量为10%(质量分数)且分批加入;造孔剂活性炭粉的用量为2%(质量分数)。该蜂窝状催化剂在120 ℃下脱硝率维持在90%以上,并且在SO2体积分数低于0.02%时具有一定的抗硫抗水性。表征结果表明,成型后蜂窝状催化剂比表面积降低,颗粒分散程度明显减弱,并且表面酸量和表面Mn4+含量下降,对催化活性有一定的影响。Abstract: A series of honeycombed holmium-modified Fe-Mn/TiO2 catalysts were prepared by moulding method and their performance in the low temperature selective catalytic reduction (SCR) of NOx was investigated. The forming process was optimized as:the mass content of water in the forming additives is 40%, in which 10% structure strengthening agent (glass fiber), 5% binder (carboxymethyl cellulose), 10% squeezing agent (glycerin), 5% pore-forming agent (activated carbon) and a little lubricants (liquid paraffin) are added. The honeycombed Fe-Ho-Mn/TiO2 catalyst prepared under these conditions exhibits excellent performance in the low-temperature SCR of NOx; the conversion of NOx exceeds 90% at 120℃ and a good resistance to SO2 and H2O is observed when the content of SO2 in the stream is lower than 0.02%. The characterization results indicated that in comparison with the parent powder holmium-modified Fe-Ho-Mn/TiO2 catalyst, the honeycombed catalyst exhibits lower surface area, more particle aggregation, and less acid sites and Mn4+ species on the surface, which has a certain negative influence on the catalytic performance of Fe-Ho-Mn/TiO2.
-
Key words:
- holmium /
- selective catalytic reduction /
- de-NOx /
- low temperature /
- honeycombed /
- forming
-
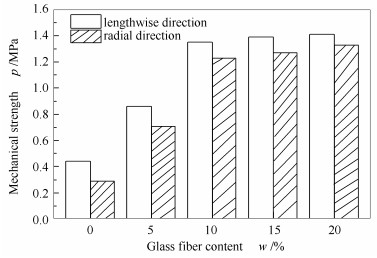
表 1 不同黏合剂含量下成型催化剂的成型效果和机械强度
Table 1 Morphology and mechanical strength of the honeycombed catalysts with different binder contents
Entry Binder Content w/% Morphology Axial mechanical strength p/MPa 1 CMC 2.5 the mud viscosity is low and cracks occur after roasting 1.10 2 CMC 5 the mud viscosity is proper and it is easy to form 1.35 3 CMC 10 the mud viscosity is high and it is difficult to demold 0.94 4 PAM 5 the mud viscosity is low and it is difficult to bond and easy to crack after moulding 0.59 5 PAM 10 the mud viscosity is low and cracks occur after roasting 0.95 6 PAM 15 the viscosity is proper and it is easy to form 1.27 7 PAM 20 the mud viscosity is high and it is difficult to demold 0.98 表 2 不同活性炭粉含量下成型催化剂的机械强度和BET比表面积
Table 2 Mechanical strength and BET surface area of the honeycombed catalysts with different activated carbon contents
Entry AC content w/% Axial direction strength p/MPa ABET/ (m2·g-1) 1 0 1.35 72 2 1 1.34 79 3 2 1.34 83 4 3 1.31 84 5 4 1.25 85 表 3 Fe-Ho-Mn/TiO2催化剂颗粒成型前后的BET比表面积、孔容和孔径
Table 3 Textural properties of the powder and honeycombed Fe-Ho-Mn/TiO2 catalysts
Entry Cataylyst Surface area A/(m2·g-1) Total pore volume v/(cm3·g-1) Average pore width d/nm 1 powder 95 0.28 12.0 2 honeycombed 83 0.24 13.7 表 4 Fe-Ho-Mn/TiO2催化剂颗粒成型前后的NH3-TPD谱图
Table 4 NH3-TPD results of the powder and honeycombed Fe-Ho-Mn/TiO2 catalysts
Entry Catalyst NH3 desorption peak area weak(< 300 ℃) medium (300-500 ℃) strong (> 500 ℃) total 1 powder 6621 6539 6126 19286 2 honeycombed 4204 2005 1931 8140 -
[1] SHAN W, LIU F, HE H, SHI X, ZHANG C. A superior Ce-W-Ti mixed oxide catalyst for the selective catalytic reduction of NOx with NH3[J]. Appl Catal B:Environ, 2012, 115:100-106. http://journal.hep.com.cn/fese/CN/abstract/abstract4885.shtml [2] JIANG B, DENG B, ZHANG Z, WU Z, TANG X, YAO S, LU H. Effect of Zr addition on the low-temperature SCR activity and SO2 tolerance of Fe-Mn/Ti catalysts[J]. J Phys Chem C, 2014, 118(27):14866-14875. doi: 10.1021/jp412828p [3] DUNN J P, KOPPULA P R, STENGER H G, WACHS I E. Oxidation of sulfur dioxide to sulfur trioxide over supported vanadia catalysts[J]. Appl Catal B:Environ, 1998, 19(2):103-117. doi: 10.1016/S0926-3373(98)00060-5 [4] BALLE P, GEIGER B, KURETI S. Selective catlytic reduction of NOx by NH3 on Fe/HBEA zeolite catalysts in oxygen-rich exhaust[J]. Appl Catal B:Environ, 2009, 85(3/4):109-119. https://www.sciencedirect.com/science/article/pii/S092633730800249X [5] LI J, CHANG H, MA L, HAO J, YANG R T. Low-temperature selective catalytic reduction of NOx with NH3 over metal oxide and zeolite catalysts-A review[J]. Catal Today, 2011, 175(1):147-156. doi: 10.1016/j.cattod.2011.03.034 [6] SHEN B, LIU T, ZHAO N, YANG X, DENG L. Lron-doped Mn-Ce/TiO2 catalyst for low temperature selective catalytic reduction of NO with NH3[J]. J Environ Sci-China, 2010, 22(9):1447-1454. doi: 10.1016/S1001-0742(09)60274-6 [7] 熊志波, 路春美.铁铈复合氧化物催化剂SCR脱硝的改性研究[J].燃料化学学报, 2013, 41(3):361-367. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18150.shtmlXIONG Zhi-bo, LU Chun-mei. Study on the modification of iron-cerium mixed oxide catalyst for selective catalytic reduction of NO[J]. J Fuel Chem Technol, 2013, 41(3):361-367. http://manu60.magtech.com.cn/rlhxxb/CN/abstract/abstract18150.shtml [8] 钱怡君, 归柯庭, 梁辉.负载型Fe-Ce催化剂低温SCR脱硝性能的研究[J].工程热物理学报, 2015(01):101-105. http://www.cqvip.com/QK/90922X/201501/1005691851.htmlQIAN Yi-jun, GUI Ke-ting, LIANG Hui. Performance of low-temperature selective catalytic reduction of NO over supported Fe-Ce catalysts[J]. J ENG THERMOPHYS-RUS, 2015(01):101-105. http://www.cqvip.com/QK/90922X/201501/1005691851.html [9] FAN Z, SHI J, GAO C, GAO G, WANG B, NIU C. Rationally designed porous MnOx-FeOx nanoneedles for low-temperature selective catalytic reduction of NOx by NH3[J]. ACS Appl Mater Inter, 2017, 9(19):16117-16127. doi: 10.1021/acsami.7b00739 [10] 张信莉, 路春美, 王栋, 徐丽婷, 彭建升. Fe对MnOx催化剂低温SCR脱硝性能的影响[J].锅炉技术, 2014(06):67-71. doi: 10.3969/j.issn.1672-4763.2014.06.014ZHANG Xin-li, LU Chun-mei, WANG Dong, XU Li-ting, PENG Jian-sheng. The effect of Fe on De-NOx activity of low-temperature SCR MnOx catalyst[J]. Boiler Technology, 2014(06):67-71. doi: 10.3969/j.issn.1672-4763.2014.06.014 [11] MEKHEMER G. Surface acid-base properties of holmium oxide catalyst:In situ infrared spectroscopy[J]. APPL CATAL A-GEN, 2004, 275(1/2):1-7. https://www.sciencedirect.com/science/article/pii/S0926860X04005009 [12] 邱春天, 林涛, 张秋林, 徐海迪, 陈耀强, 龚茂初.改性ZrO2-MnO2基整体式催化剂上NH3选择性催化还原NO[J].催化学报. 2011, 32(7):1227-1233. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA201107020.htmQIU Chun-tian, LIN Tao, ZHANG Qiu-lin, XU Hai-di, CHEN Yao-qiang, GONG Mao-chu. Selective catalytic reduction of NO with NH3 on modified ZrO2-MnO2 monolithic catalysts[J]. Chin J Catal, 2011, 32(7):1227-1233. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA201107020.htm [13] 唐晓龙, 郝吉明, 易红宏, 宁平, 李俊华.活性炭改性整体催化剂上低温选择性还原NOx[J].中国环境科学. 2007, 27(6):845-850. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGHJ200706032.htmTANG Xiao-long, HAO Ji-ming, YI Hong-hong, NING Ping, LI Jun-hua. Selective catalytic reduction of NO with NH3 by monolithic catalyst MnOx/AC/C under low temperature in the presence of excess O2[J]. Chi Environ Sci, 2007, 27(6):845-850. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGHJ200706032.htm [14] 黄海凤, 周小燕, 卢晗锋, 俞河, 陈银飞.原位沉淀技术制备整体型Mn/Ti-Si/堇青石选择性催化还原催化剂[J].中国电机工程学报, 2011, 31(17):50-54. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201117008.htmHUANG Hai-feng, ZHOU Xiao-yan, LU Han-feng, YU He, CHEN Yin-fei. Monolithic Mn/Ti-Si/cordierite catalyst prepared by In-situ deposition for SCR-DeNOx[J]. Proc CSEE, 2011, 31(17):50-54. http://www.cnki.com.cn/Article/CJFDTOTAL-ZGDC201117008.htm [15] ZHU Y, ZHANG Y, XIAO R, HUANG T, SHEN K. Novel holmium-modified Fe-Mn/TiO2 catalysts with a broad temperature window and high sulfur dioxide tolerance for low-temperature SCR[J]. Catal Commun, 2017, 88:64-67. doi: 10.1016/j.catcom.2016.09.031 [16] LIU Q Y, LIU Z Y, HUANG Z G, XIE G Y. A honeycomb catalyst for simultaneous NO and SO2 removal from flue gas:Preparation and evaluation[J]. Catal Today. 2004, 93:833-837. https://www.sciencedirect.com/science/article/pii/S0920586104003165 [17] SHANG X, HU G, HE C, ZHAO J, ZHANG F, XU Y, ZHANG Y, LI J, CHEN J. Regeneration of full-scale commercial honeycomb monolith catalyst (V2O5-WO3/TiO2) used in coal-fired power plant[J]. J IND ENG CHEM, 2012, 18(1):513-519. doi: 10.1016/j.jiec.2011.11.070 [18] BUSCA G, LIETTI L, RAMIS G, BERTI F. Chemical and mechanistic aspects of the selective catalytic reduction of NOx by ammonia over oxide catalysts:A review[J]. Appl Catal B:Environ, 1998, 18(1/2):1-36. https://www.sciencedirect.com/science/article/pii/S092633739800040X [19] YASHNIK S A, ISMAGILOV Z R, KOPTYUG I V, ANDRIEVSKAYA I P, MATVEEV A, MOULIJIN J A. Formation of textural and mechanical properties of extruded ceramic honeycomb monoliths:An H-1 NMR imaging study[J]. Catal Today, 2005, 105(3):507-515. https://www.sciencedirect.com/science/article/pii/S0920586105003020 [20] 李锋. 以纳米TiO2为载体的燃煤烟气脱硝SCR催化剂的研究[D]. 南京: 东南大学, 2006.LI Feng. Study of SCR catalyst for coal-fired flue gas denitrification grafted on nanometer titania[D]. Nanjing: Southeast University, 2006. ) [21] KLIMCZAKM, KERN P, HEINZELMANN T, LUCAS M, CLAUS P. High-throughput study of the effects of inorganic additives and poisons on NH3-SCR catalysts Part I:V2O5-WO3/TiO2 catalysts[J]. Appl Catal B:Environ. 2010, 95(1):39-47. https://www.pubfacts.com/search/vanadium%20oxide/4 [22] KERN P, KLIMCZAKl M, HEINZELMANN T, LUCAS M, CLAUS P. High-throughput study of the effects of inorganic additives and poisons on NH3-SCR catalysts. Part Ⅱ:Fe zeolite catalysts[J]. Appl Catal B:Environ, 2010, 95(1):48-56. http://www.oalib.com/paper/4213498 [23] KHODAYARI R, ODENBRAND C. Selective catalytic reduction of NOx:A mathematical model for poison accumulation and conversion performance[J]. CHEM ENG SCI, 1999, 54(12):1775-1785. doi: 10.1016/S0009-2509(99)00017-2 [24] 李游. 整体式SCR蜂窝催化剂的制备工艺及性能评价[D]. 华东理工大学, 2013.LI You. The Preparation Technology and performance evaluation of monolithic SCR catalyst[D]. Shanghai: East China University of Science and Technology, 2013. ) [25] 于国峰. Mn-Ce/TiO2系低温SCR催化剂脱硝性能及蜂窝状成型制备研究[D]. 浙江工业大学, 2012.YU Guo-feng. Study on the Mn-Ce/TiO2 low-temperature SCR catalysts: Denitration performance and preparation method of honeycombed catalyst[D]. Hang zhou: Zhejiang University of Technology 2012. ) [26] 张雄飞, 谢放华, 王伟, 陈晓宁, 王玉雪.蜂窝状粘土基催化剂成型的研究[J].广州化工, 2012(10):80-82. doi: 10.3969/j.issn.1001-9677.2012.10.031ZHANG Xiong-fei, XIE Fang-hua, WANG Wei, CHEN Xiao-ning, WANG Yu-xue. Study on the molding technology of clay honeycomb catalysts[J]. Guangzhou Chemical Industry, 2012(10):80-82. doi: 10.3969/j.issn.1001-9677.2012.10.031 [27] 马昊. 水泥炉窑烟气低温SCR脱硝成型催化剂优化研究[D]. 浙江大学, 2015.MA Hao. The optimization of molded low-temperature SCR catalysts for NOx removal in cement kiln flue gas[D]. Hangzhou: Zhejiang University, 2015. ) [28] 肖琨. SCR脱硝技术用催化剂性能试验与成型研究[D]. 山东大学, 2008.XIAO Kun. Performance test and molding study of SCR catalyst[D]. Jinan: Shandong University, 2008. ) [29] 陈菲. 3-甲基吡啶氨氧化V2O5/TiO2催化剂研究[D]. 浙江师范大学, 2008.CHEN Fei. Studies on V2O5/TIO2 catalysts for ammoxidation of 3-Picoline[D]. Hangzhou: Zhejiang Normal University, 2008. ) [30] SHEN B, YAO Y, MA H, LIU T. Ceria modified MnOx/TiO2-pillared cays catalysts for the selective catalytic reduction of NO with NH3 at low temperature[J]. Chin J Catal. 2011, 32(11/12):1803-1811. doi: 10.1021/acsanm.7b00320 [31] ZHANG Y, HUANG T, XIAO R, XU H, SHEN K, ZHOU C. A comparative study on the Mn/TiO2-M(M=Sn, Zr or Al) Ox catalysts for NH3-SCR reaction at low temperature[J]. Environ technol, 2017:1-11. doi: 10.1021/jp048431h [32] XIONGY, TANG C, YAO X, ZHANG L, LI L, WANG X, DENG Y, GAO F, DONG L. Effect of metal ions doping (M=Ti4+, Sn4+) on the catalytic performance of MnOx/CeO2 catalyst for low temperature selective catalytic reduction of NO with NH3[J]. APPL CATAL A-GEN. 2015, 495:206-216. doi: 10.1016/j.apcata.2015.01.038 [33] WANG X, LI X, ZHAO Q, SUN W, TADE M, LIU S. Improved activity of W-modified MnOx-TiO2 catalysts for the selective catalytic reduction of NO with NH3[J]. Chem Eng J. 2016, 288:216-222. doi: 10.1016/j.cej.2015.12.002 [34] LUO S, ZHOU W, XIE A, WU F, YAO C, LI X, ZUO S, LIU T. Effect of MnO2 polymorphs structure on the selective catalytic reduction of NOx with NH3 over TiO2-Palygorskite[J]. CHEM ENG J, 2016, 286:291-299. doi: 10.1016/j.cej.2015.10.079 [35] TAN P. Active phase, catalytic activity, and induction period of Fe/zeolite material in nonoxidative aromatization of methane[J]. J CATA. 2016, 338:21-29. doi: 10.1016/j.jcat.2016.01.027 [36] FANG N, GUO J, SHU S, LUO H, CHU Y, LI J. Enhancement of low-temperature activity and sulfur resistance of Fe0.3Mn0.5Zr0.2 catalyst for NO removal by NH3-SCR[J]. CHEM ENG J, 2017, 325:114-123. doi: 10.1016/j.cej.2017.05.053 [37] LIU J, LIU J, ZHAO Z, WEI Y, SONG W, LI J, ZHANG X. A unique Fe/Beta@TiO2 core-shell catalyst by small-grain molecular sieve as the core and TiO2 nanosize thin film as the shell for the removal of NOx[J]. Ind Eng Chem Res, 2017, 56(20):5833-5842. doi: 10.1021/acs.iecr.7b00740 [38] 刘世斌, 王秀光, 李一兵, 郝晓刚, 张忠林, 段东红, 孙彦平. Ho掺杂对Pt-Ru/C电催化氧化甲醇的性能影响(英文)[J].稀有金属材料与工程, 2008(05):909-913. doi: 10.3321/j.issn:1002-185X.2008.05.037LIU Shi-bin, WANG Xiu-guang, LI Yi-bing, HAO Xiao-gang, ZHANG Zhong-lin, DUAN Dong-hong, SUN Yan-ping. Performance of Pt-Ru/C Doped with Ho for methanol electro-catalytic oxidation[J]. RARE METAL MAT ENG, 2008(05):909-913. doi: 10.3321/j.issn:1002-185X.2008.05.037 -





 下载:
下载: