Study on the preparation of TiO2-Al2O3 composite support and its application in Co-Mo/TiO2-Al2O3 catalyst for hydro-desulfurization
-
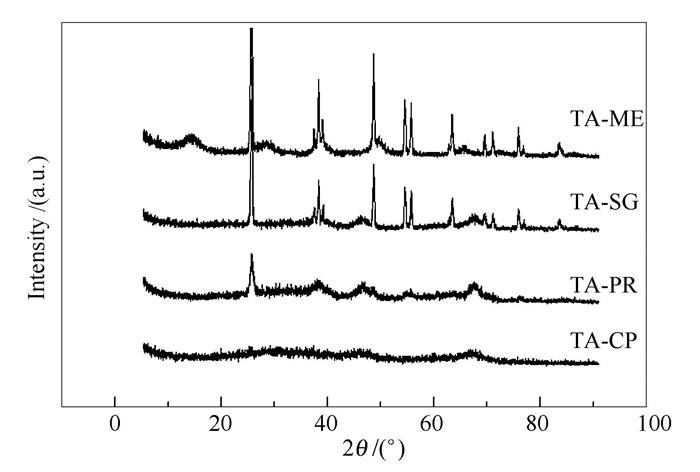
摘要: 通过改进的溶胶-凝胶法(SG)、共沉淀法(CP)、表面沉淀法(PR)及混捏法(ME)制备TiO2-Al2O3复合载体,考察了不同制备方法对复合载体物理性质的影响。采用浸渍法制备Co-Mo/TiO2-Al2O3-X加氢脱硫催化剂,研究了Co-Mo/TiO2-Al2O3-X加氢脱硫催化剂的脱硫性能。利用XRD、BET、SEM等表征手段对复合载体及催化剂进行表征分析。结果表明,SG法制备的复合载体粒径均一,具有较大的比表面积、孔径和孔体积;CP法制备复合载体时TiO2以单层或亚单层的分散状态高度分散于γ-Al2O3中。在氢气压力3.0 MPa、反应温度280℃、反应时间4 h、液时空速1.4 h-1和氢油比600的条件下,SG法制备的Co-Mo/TiO2-Al2O3催化剂具有较高加氢脱硫活性,噻吩转化率达到96.6%。
-
关键词:
- TiO2-Al2O3复合载体 /
- 加氢脱硫 /
- 溶胶-凝胶法 /
- Co-Mo催化剂
Abstract: A variety of TiO2-Al2O3 composite support were prepared using improved sol-gel method (SG), co-precipitation method (CP), surface precipitation (PR) and mechanical kneading method (ME), and the influence of preparation methods on their physical properties were investigated. The hydro-desulfurization activity of Co-Mo/TiO2-Al2O3-X catalysts prepared by impregnation method was studied. XRD, BET and SEM were used to characterize the composite support and the catalysts. Results show that the composite support prepared by SG method has uniform morphology with larger surface area, pore size and pore volume. And the monolayer or sub-monolayer of TiO2 existed in the γ-Al2O3. Under the reaction conditions of 3.0 MPa of hydrogen pressure, 280℃ of reaction temperature, 4 h of reaction time, 1.4 h-1 of the liquid space velocity and 600 of hydrogen to oil ratio, Co-Mo/TiO2-Al2O3 prepared by SG method has highest hydro-desulfurization activity, and the thiophene conversion rate reached 96.6%.-
Key words:
- TiO2-Al2O3composite support /
- hydro-desulfurization /
- sol-gel method /
- Co-Mo catalyst
-
表 1 复合载体的物理性质
Table 1 Physical properties of TA support
Sample ABET/
(m2·g-1)BJH pore volume
v/ (cm3·g-1)Average pore
size d/nmAl2O3 296 0.80 8.01 TiO2 145 0.38 4.40 TA-ME 169 0.63 11.34 TA-SG 242 0.88 12.24 TA-PR 180 0.71 11.89 TA-CP 291 0.26 3.08 表 2 载体制备方法对Co-Mo/TiO2-Al2O3催化剂HDS活性的影响
Table 2 Effect of preparation methods of the support on the activity of the Co-Mo/TiO2-Al2O3 catalyst for HDS
Catalyst TiO2 w/% Thiophene
conversion x/%Co-Mo/TiO2-Al2O3-SG 30 96.6 Co-Mo/TiO2-Al2O3-CP 30 94.4 Co-Mo/TiO2-Al2O3-PR 30 90.4 Co-Mo/TiO2-Al2O3-ME 30 83.5 -
[1] GRZECHOWIAK J R,WERESZCZAKO-ZIELIŃSKA I,MROZIŃSKA K.HDS and HDN activity of molybdenum and nickel-molybdenum catalysts supported on alumina-titania carriers[J].Catal Today,2007,119(1):23-30. https://www.researchgate.net/publication/232400439_HDS_and_HDN_activity_of_molybdenum_and_nickel-molybdenum_catalysts_supported_on_alumina-titania_carriers [2] LU S,YUAN L.Preparation of meso-macroporous carbon nanotube-alumina composite monoliths and their application to the preferential oxidation of CO in hydrogen-rich gases[J].Appl Catal B:Environ,2012,111-112(2):492-501. [3] 杨祝红,李立成,王艳芳,刘金龙,冯新,陆小华.磷化镍/介孔TiO2催化剂的制备及其催化加氢脱硫性能[J].催化学报,2012,33(3):508-517.YANG Zhu-hong,LI Li-cheng,WANG Yan-fang,LIU Jin-long,FENG Xin,LU Xiao-hua.Preparation of nickel phosphide/mesoporous-TiO2 catalyst and its hydrodesulfurization performance[J].Chin J Catal,2012,33(3):508-517. [4] ELEKTOROWICZ M,HABIBI S.Sustainable waste management:Recovery of fuels from petroleum sludge[J].Can J Civ Eng,2005,32(1):164-169. doi: 10.1139/l04-122 [5] 彭会左,杨运泉,王威燕,何兵,钦柏豪.超声波辅助共沉淀法制备大比表面积TiO2-Al2O3复合载体[J].石油化工,2011,40(7):726-731.PENG Hui-zuo,YANG Yun-quan,WANG Wei-yan,HE Bing,QIN Bo-hao.Preparation of TiO2-Al2O3 Composite support with high specific surface area by ultrasound-assisted Co-precipitation method[J].Petrkchem Technol,2011,40(7):726-731. [6] WEI Z,XIN Q,GUO X,SHAM E L,GRANGE P,DELMON B.Titania-modified hydrodesulphurization catalysts:I.Effect of preparation techniques on morphology and properties of TiO2-Al2O3 carrier[J].Appl Catal,1990,63(1):305-317. doi: 10.1016/S0166-9834(00)81721-2 [7] CHOI J,BAN K,CHOUNG S-J,KIM J,ABIMANYU H,YOO K S.Sol-gel synthesis,characterization and photocatalytic activity of mesoporous TiO2/γ-Al2O3 granules[J].J Sol-Gel Sci Technol,2007,44(1):21-28. doi: 10.1007/s10971-007-1592-0 [8] 徐如人.分子筛与多孔材料化学[M].北京:科学出版社,2004.XU Ru-ren.Molecular Sieve and Porous Materials Chemistry[M].Beijing:Science Press,2004. [9] 张成,王永林,杨春雁,隋宝宽,杨刚,王刚,赵国利.NiMo/TiO2-Al2O3催化剂活性相表征及加氢脱硫反应性能研究[J].工业催化,2012,20(5):31-35.ZHANG Cheng,WANG Yong-lin,YANG Chun-yan,SUI Bao-kuan,YANG Gang,WANG Gang,ZHAO Guo-li.Characterization of active phase of NiMo/TiO2-Al2O3 catalyst and its performance in hydrodesulphurization reaction[J].Ind Catal,2012,20(5):31-35. [10] DAAGE M,CHIANELLI R R.Structure-Function relations in molybdenum sulfide catalysts:The "Rim-Edge" Model[J].J Catal,1994,149(2):414-427. doi: 10.1006/jcat.1994.1308 [11] TOPSØE H,CLAUSEN B S,CANDIA R,WIVEL C,MOERUP S.In situ Moessbauer emission spectroscopy studies of unsupported and supported sulfided Co-Mo hydrodesulfurization catalysts:Evidence for and nature of a Co-Mo-S phase[J].J Catal (United States),1981,68(2):433-452. [12] ÍGUTIÉRREZ-ALEJANDRE A,RAMÍREZ J,VAL L J,PEÑUELAS-GALAZA M,SÁNCHEZ-NERIA P,TORRES-MANCERA P.Activity of NiW catalysts supported on TiO2-Al2O3 mixed oxides:Effect of Ti incorporation method on the HDS of 4,6-DMDBT[J].Catal Today,2005,107(44):879-884. [13] WANG D,LI W,ZHANG M,TAO K.Promoting effect of fluorine on titania-supported cobalt-molybdenum hydrodesulfurization catalysts[J].Appl Catal,2007,317(1):105-112. doi: 10.1016/j.apcata.2006.10.020 [14] PAUL J,PAYEN E.Vacancy formation on MoS2 hydrodesulfurization catalyst:DFT study of the mechanism[J].J Phys Chem B,2003,107(17):4057-4064. doi: 10.1021/jp027668f -





 下载:
下载: