Preparation and desulfurization performance of molecularly imprinted composite with dibenzothiophene as template
-
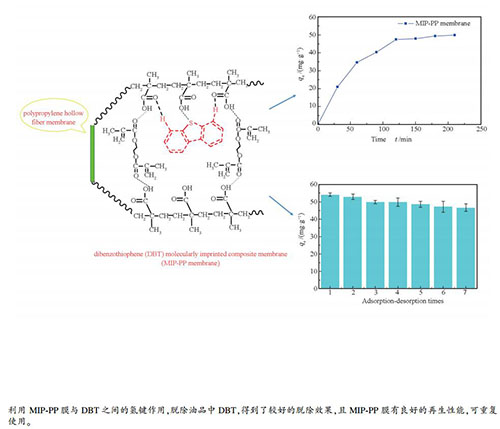
摘要: 聚丙烯中空纤维膜经多巴胺氧化、硅烷化两步表面改性处理后,以甲基丙烯酸为功能单体进行表面分子印迹聚合,制备了中空纤维膜支撑-二苯并噻吩分子印迹复合膜(MIP-PP膜)。利用红外光谱、扫描电镜对印迹复合膜形态结构进行表征,测定了MIP-PP膜的脱硫性能。结果表明,在298 K时,MIP-PP膜对DBT的吸附在180 min达到平衡,最大吸附容量为133.32 mg/g;MIP-PP膜对DBT的吸附符合Lagergren准一级动力学模型及Langmuir吸附等温线,是可自发进行的放热过程。Abstract: The polypropylene hollow fiber membrane was subjected to dopamine oxidation and silanization two-step surface modification treatment, and then the surface molecular molecularly imprinted polymerization was carried out using methacrylic acid as a functional monomer to prepare a hollow fiber membrane supported-dibenzothiophene molecularly imprinted composite membrane (MIP-PP membrane).The morphology of the imprinted composite membrane was characterized by infrared spectroscopy and scanning electron microscopy, and the desulfurization performance of MIP-PP membrane was measured. The results show that at 298 K, the adsorption of DBT by MIP-PP membrane reaches equilibrium at 180 min, and the maximum adsorption capacity is 133.32 mg/g. The adsorption of DBT by MIP-PP membrane conforms to the Lagergren quasi-first-order kinetic model and the Langmuir adsorption isotherm, which is a spontaneous exothermic process.
-
图 4 不同聚合条件下制备的MIP-PP膜对DBT的吸附性能
Figure 4 Adsorption of DBT by MIP-PP membranes prepared under different polymerization conditions
(a): polymerization conditions is DBT:EGDMA=1:10, polymerization pemperature at 65 ℃ (b): polymerization conditions is DBT:MAA=1:4, polymerization pemperature at 65 ℃ (c): polymerization conditions is DBT:MAA:EGDMA=1:4:10 (d): polymerization conditions is DBT:functional monomer:EGDMA=1:4:10 and polymerization pemperature at 65 ℃
表 1 MIP-PP膜和NIP-PP膜对DBT吸附等温线参数
Table 1 Adsorption isotherm parameters of MIP-PP membrane and NIP-PP membrane to DBT
Isotherm model Parameter MIP-PP membrane NIP-PP membrane 298 K 308 K 318 K 298 K 308 K 318 K Langmuir model kL 0.68 0.73 0.57 0.86 0.77 0.65 qm 133.32 114.74 116.86 62.89 60.24 57.81 R2 0.991 0.999 0.993 0.996 0.999 0.997 Freundlich model kF 1.48 1.49 1.41 1.46 1.45 1.37 n 48.87 44.06 38.85 24.82 24.27 21.57 R2 0.973 0.961 0.966 0.989 0.986 0.986 表 2 MIP-PP膜对DBT的吸附热力学参数
Table 2 Thermodynamic parameters for the adsorption of MIP-PP membrane to DBT
T/K kc ΔG0 ΔH0 ΔS0 298 56.09 -9.97 -11.96 -6.75 308 47.76 -9.90 318 41.38 -9.84 表 3 MIP-PP膜及NIP-PP膜选择性吸附参数
Table 3 Selective recognition parameters of MIP-PP membrane and NIP-PP membrane
Compounds MIP-PP membrane NIP-PP membrane k' kd k kd k DBT 0.0553 - 0.0293 - - BT 0.0384 1.44 0.0282 1.03 1.39 T 0.0327 1.69 0.0273 1.07 1.57 -
[1] GB18352.6-2016, 轻型汽车污染物排放限值及测方法[S].GB18352.6-2016, Limits and measurement methods for emissions from light-duty vehicles[S]. [2] 董立霞, 夏步田, 罗凯威, 赵亮, 高金森, 郝天臻.清洁油品升级背景下加氢脱硫技术研究进展[J].化工进展, 2019, 38(1):208-216. http://d.old.wanfangdata.com.cn/Periodical/hgjz201901019DONG Li-xia, XIA Bu-tian, LUO Kai-wei, ZHAO Liang, GAO Jin-seng, HAO Tian-qin. Review of hydrodesulfurization technology based on the upgrading requirement of clean gasoline[J]. Chem Prog, 2019, 38(1):208-216. http://d.old.wanfangdata.com.cn/Periodical/hgjz201901019 [3] LIU W F, ZHAO H J, YANG Y Z, ZHANG Y, XU B S. Selective removal of benzothiophene and dibenzothiophene from gasoline using double-template molecularly imprinted polymers on the surface of carbon microspheres[J]. Fuel, 2014, 117:184-190. doi: 10.1016/j.fuel.2013.09.031 [4] LI X, XU Y L, WANG H, SONG Y J, ZHANG W, LI C P. H2O2 assisted hydrothermal synthesis of TiO2-SiO2 and its enhanced photocatalytic adsorptive desulfurization performance for model fuel[J]. Fuel, 2018, 226:527-535. doi: 10.1016/j.fuel.2018.04.046 [5] FATEMEH D D, MANOUCHEH V, ABED A Z. Biodesulfurization of dibenzothiophene by a newly isolated Rhodococcus erythropolis strain[J]. Bioresour Technol, 2010, 101:1102-1105. doi: 10.1016/j.biortech.2009.08.058 [6] YANG Y Z, ZHUANG Y, LI S, LIU X G, XU B S. Grafting molecularly imprinted poly (2-acrylamido-2-methylpropanesulfonic acid) onto the surface of carbon microspheres[J]. Appl Surf Sci, 2012, 258:6441-6450. doi: 10.1016/j.apsusc.2012.03.058 [7] HUA S J, ZHAO L, CAO L Y, WANG X Q, GAO J S, XU C M. Fabrication and evaluation of hollow surface molecularly imprinted polymer for rapid and selective adsorption of dibenzothiophene[J]. Chem Eng J, 2018, 345:414-424. doi: 10.1016/j.cej.2018.03.128 [8] LIZ N M S, BRUNO E L B, MARCIO C P, LUAZ C A O, ADILSON C D S. Thermodinamic study of a magnetic molecular imprinted polymer for removal of nitrogenous pollutant from gasoline[J]. Fuel, 2017, 210:380-389. doi: 10.1016/j.fuel.2017.08.087 [9] ESMAIL M, MOHAMMAD R Y, HASSAN S J, ABBASALI Z, FARIDEH P. Molecularly imprinted pol (4, 4'-methylenedianiline) as electrochemical sensor for determination of 1-benzothiophene[J]. Synth Met, 2020, 259:116252. doi: 10.1016/j.synthmet.2019.116252 [10] YANG W M, LIU L K, ZHOU Z P, LIU H, XIE B Z, XU W Z. Rational preparation of dibenzothiophene imprinted polymers by surface imprinting technique combined with atom transfer radical polymerization[J]. Appl Surf Sci, 2013, 282:809-819. doi: 10.1016/j.apsusc.2013.06.063 [11] XU W Z, ZHOU W, XU P P, PAN J M, WU X Y, YAN Y S. A molecularly imprinted polymer based on TiO2 as a sacrificial support for selective recognition of dibenzothiophene[J]. Chem Eng J, 2011, 172:191-198. doi: 10.1016/j.cej.2011.05.089 [12] LIU W F, QIN L, YANG Y Z, LIU X G, XU B S. Synthesis and characterization of dibenzothiophene imprinted polymers on the surface of iniferter-modified carbon microspheres[J]. Mater Chem Phys, 2014, 148:605-613. doi: 10.1016/j.matchemphys.2014.08.024 [13] 司汇通, 王兵, 单娟娟.磁性分子印迹微球的合成及其对二苯并噻吩的选择性吸附研究[J].功能材料, 2016, 47(SⅠ):188-192. http://d.old.wanfangdata.com.cn/Periodical/gncl2016z1034SI Hui-tong, WANG Bing, SHAN Juan-juan. Synthesis of magnetic molecularly imprinted microspheres and it selective adsorption of dibenzothiophene[J]. Funct Mater, 2016, 47(SⅠ):188-192. http://d.old.wanfangdata.com.cn/Periodical/gncl2016z1034 [14] ALI H R, EI-MAGHRABI H H, ZAHRAN F, YASSER M M. A novel surface imprinted polymer/magnetic hydroxyapatite nanocomposite for selective dibenzothiophene scavenging[J]. Appl Surf Sci, 2017, 426:56-66. doi: 10.1016/j.apsusc.2017.07.105 [15] MARYAM F, MOHAMMAD A T, DARYOUSH A, ALI M. Reparation of molecularly imprinted polymer coated magnetic multi-walled carbon nanotubes for selective removal of dibenzothiophene[J]. Mater Sci Semicond Process, 2015, 40:501-507. doi: 10.1016/j.mssp.2015.07.018 [16] 焦翠翠, 胡小玲, 管萍, 于进洋.复合型分子印迹膜的制备及影响因素[J].化学工业与工程, 2010, 27(4):358-363. doi: 10.3969/j.issn.1004-9533.2010.04.015JIAO Cui-cui, HU Xiao-ling, GUAN Ping, YU Jin-yang. Preparation and influence factors of composite molecular imprinted membranes[J]. Chem Ind Eng, 2010, 27(4):358-363. doi: 10.3969/j.issn.1004-9533.2010.04.015 [17] 邱增英, 钟世安.烟酸分子印迹复合膜的制备及其分离性能研究[J].化学学报, 2010, 68(3):246-250. http://d.old.wanfangdata.com.cn/Periodical/hxxb201003008QIU Zeng-ying, ZHONG Shi-an. Preparation of nicotinic acid molecularly imprinted composite membrane and its separation performance[J]. J Chem, 2010, 68(3):246-250. http://d.old.wanfangdata.com.cn/Periodical/hxxb201003008 [18] 胡廷平, 李伟成, 郑立辉, 郑洁, 王敏.硅胶表面3-甲基噻吩分子印迹聚合物的制备及吸附特性研究[J].离子交换与吸附, 2015, 31(3):250-259. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=lzjhyxf201503007HU Ting-ping, LI Wei-cheng, ZHENG Li-hui, ZHENG Jie, WANG Min. Preparation and adsorption properties of 3-methylthiophene molecularly imprinted polymer on silica surface[J]. Ion Exc Adsorpt, 2015, 31(3):250-259. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=lzjhyxf201503007 [19] YANG W M, LIU L K, ZHOU Z P, YANG C, XU Y Z. Development of surface imprinting polymer as a selective adsorbent for adsorbing and separating dibenzothiophene from fuel oil[J]. Res Chem Intermed, 2015, 41:2619-2633. doi: 10.1007/s11164-013-1375-x [20] 张燕, 刘旭光, 杨永珍, 杨致, 许并社.二苯并噻吩分子印迹聚合物中功能单体的筛选[J].中国科技论文, 2013, 8(6):506-511. doi: 10.3969/j.issn.2095-2783.2013.06.004ZHANG Yan, LIU Xu-guang, YANG Yong-zhen, YANG Zhi, XU Bing-she. Selection of functional monomer for molecularly imprinted polymers with dibenzothiophenen as template[J]. Chin Sci Lett, 2013, 8(6):506-511. doi: 10.3969/j.issn.2095-2783.2013.06.004 -





 下载:
下载: