Performance of CO preferential oxidation of CeO2-NiO nanorod catalyst in H2-rich stream
-
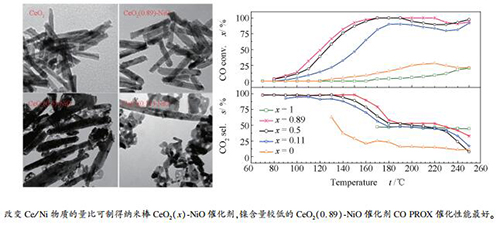
摘要: 采用水热法制备了一系列不同Ce/Ni物质的量比的纳米棒CeO2(x)-NiO催化剂。运用低温N2吸附-脱附、XRD、TEM、拉曼光谱、H2-TPR及XPS等技术对催化剂的形貌、结构进行了表征。考察了Ce/Ni物质的量比对CeO2(x)-NiO催化剂形貌及富氢气氛下CO选择性氧化(CO PROX)反应性能的影响。TEM测试结果表明,调变Ce/Ni物质的量比可制得不同粒径的CeO2(x)-NiO纳米棒催化剂。H2-TPR测试结果表明,将NiO掺入CeO2可提升CeO2(x)-NiO催化剂的氧化还原能力。拉曼光谱及XPS测试结果表明,镍含量较低时,CeO2(x)-NiO催化剂表面活性氧物种及氧空位含量均较多,利于提升其催化性能。CO PROX催化性能测试结果显示,镍含量较低的CeO2(0.89)-NiO纳米棒催化剂的活性和选择性最好,在170-220 ℃的反应条件下,CO转化率为100%,CO2选择性为52%。Abstract: A series of nanorod CeO2(x)-NiO composite oxides catalysts with different Ce/Ni molar ratios have been synthesized by hydrothermal method. Their morphology and structure were characterized by N2 sorption-desorption, XRD, TEM, Raman spectra, H2-TPR and XPS. The effects of Ce/Ni molar ratio on the morphology and catalytic activity of CeO2(x)-NiO composite oxides catalysts for CO preferential oxidation (CO PROX) in hydrogen-rich stream were studied. TEM results indicate that nanorod CeO2(x)-NiO composite oxides catalysts with different sizes can be obtained by adjusting Ce/Ni molar ratio. H2-TPR results show that introduction of NiO into CeO2 enhance the redox ability of CeO2(x)-NiO composite oxides catalysts. Raman spectra and XPS results indicate that CeO2(x)-NiO composite oxides catalysts with low nickel content have much more active oxygen species and oxygen vacancies, which are beneficial to improve its catalytic performance. CeO2(0.89)-NiO nanorod catalysts with low nickel content exhibits the highest activity and CO2 selectivity, the CO conversion is 100% and the CO2 selectivity is about 52% in the reaction temperature range of 170-220 ℃ for CO PROX in hydrogen-rich stream.
-
Key words:
- ceria /
- nickel oxide /
- nanorod /
- CO preferential oxidation /
- hydrogen-rich stream
-
表 1 CeO2(x)-NiO纳米棒催化剂的化学组成、比表面积和XRD分析
Table 1 Chemical composition, surface area and XRD analysis results of the CeO2(x)-NiO nanorods catalysts with different Ce contents
xa xICPb Ce content w/% Ni content w/% ABET /(m2·g-1) Cell parametera/nmc Crystallite size d/nmd x=1 0.99 - - 102.16 0.5416 22.9 x=0.89 0.88 74.48 3.98 97.37 0.5408 26.1 x=0.5 0.50 55.32 23.48 43.12 0.5410 29.8 (27.0) x=0.11 0.10 17.44 61.20 42.23 0.5412 30.4 (28.7) x=0 0 - - 31.30 - -(34.4) a: x is the nominal content of Ce in the composite oxides expressed as the atomic ratio of n(Ce)/(n(Ce) + n(Ni)); b: xICP is the value of the atomic ratio of n(Ce)/(n(Ce) + n(Ni)) determined by ICP analysis; c: calculated from the a value of the ceria (111) planes; d: crystalline size is calculated from the line broadening of CeO2 (111) and NiO (200) (digitals in brackets) diffraction peak by the Scherrer equation from XRD patterns 表 2 CeO2(x)-NiO纳米棒催化剂的H2-TPR表征
Table 2 H2-TPR results of the CeO2(x)-NiO nanorods catalysts with different Ce contents
x Peak position t/℃ H2 uptake/(μmol·g-1) Theoretical H2 uptake/(μmol·g-1)a x=1 450 1734 2904 x=0.89 245, 375 1263, 1372 2756 x=0.5 247, 358 760, 3856 4830 x=0.11 374 8660 8798 x=0 383 13333 13385 a: theoretical H2 uptake for reduction of CeO2(x)-NiO nanorods catalysts, as calculated by assuming that CeO2 and NiO are stoichiometrically reduced to Ce2O3 and Ni, respectively -
[1] 王芳, 吕功煊.助剂改性对贵金属催化剂CO选择氧化活性中心的影响[J].化学进展, 2010, 22(8):1538-1549. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hxjz201008004WANG Fang, LÜ Gong-xuan. Influence of promoters on active centers over nobel metal catalysts for CO selective oxidation[J]. Prog Chem, 2010, 22(8):1538-1549. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=hxjz201008004 [2] HORNÉS A, HUNGRÍA A B, BERA P, LÓPEZ CÁMARA A, FERNÁNDEZ-GARCÍA M, MARTÍNEZ-ARIAS A, BARRIO L, ESTRELLA M, ZHOU G, FONSECA J J, HANSON J C, RODRIGUEZ J A. Inverse CeO2/CuO catalyst as an alternative to classical direct configurations for preferential oxidation of CO in hydrogen-rich stream[J]. J Am Chem Soc, 2010, 132(1):34-35. doi: 10.1021/ja9089846 [3] ZHU H Q, QIN Z F, SHAN W J, SHEN W J, WANG J G. Low-temperature oxidation of CO over Pd/CeO2-TiO2 catalysts with different pretreatments[J]. J Catal, 2005, 233(1):41-50. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=5565d26f854735e2b2155f31282225c6 [4] BAO H Z, CHEN X, FANG J, JIANG Z Q, HUANG W X. Structure-activity relation of Fe2O3-CeO2 composite catalysts in CO oxidation[J]. Catal Lett, 2018, 125(1/2):160-167. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=60f22992be92c9a728afe83567b658d5 [5] LIN S J, SU G J, ZHENG M H, JI D K, JIA M K, LIU Y X. Synthesis of flower-like Co3O4-CeO2 composite oxide and its application to catalytic degradation of 1, 2, 4-trichlorobenzene[J]. Appl Catal B:Environ, 2012, 123/124:440-447. doi: 10.1016/j.apcatb.2012.05.011 [6] AKANDE A J, IDEM R O, DALAI A K. Synthesis, characterization and performance evaluation of Ni/Al2O3 catalysts for reforming of crude ethanol for hydrogen production[J]. Appl Catal A:Gen, 2005, 287(2):159-175. doi: 10.1016/j.apcata.2005.03.046 [7] MARIÑO F, CERRELLA E, DUHALDE S, JOBBAGY M, LABORDE M. Hydrogen from steam reforming of ethanol:Characterization and performance of copper-nickel supported catalysts[J]. Int J Hydrogen Energy, 1998, 23(12):1095-1101. doi: 10.1016/S0360-3199(97)00173-0 [8] SHAN W J, LUO M F, YING P L, SHEN W J, LI C. Reduction property and catalytic activity of Ce1-xNixO2 mixed oxide catalysts for CH4 oxidation[J]. Appl Catal A:Gen, 2003, 246(1):1-9. doi: 10.1016/S0926-860X(02)00659-2 [9] LIU Y M, WANG L C, CHEN M, XU J, CAO Y, HE H Y, FAN K N. Highly selective Ce-Ni-O catalysts for efficient low temperature oxidative dehydrogenation of propane[J]. Catal Lett, 2009, 130(3/4):350-354. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=34060f1b0142b1983d30f407eb9b7dd6 [10] ZHONG L S, HU J S, CAO A M, LIU Q, SONG W G, WAN L J. 3D flowerlike ceria micro/nanocomposite structure and its application for water treatment and CO removal[J]. Chem Mater, 2007, 19(7):1648-1655. doi: 10.1021/cm062471b [11] LI T Y, XIANG G L, ZHANG J, WANG X. Enhanced catalytic performance of assembled ceria necklace nanowires by Ni doping[J]. Chem Commun, 2011, 47(21):6060-6062. doi: 10.1039/c1cc11547b [12] ZHANG D S, FU H X, SHI L Y, PAN C S, LI Q, CHU Y L, YU W J. Synthesis of CeO2 nanorods via ultrasonication assisted by polyethylene glycol[J]. Inorg Chem, 2007, 46(7):2446-2451. doi: 10.1021/ic061697d [13] LI C R, SUN Q T, LU N P, CHEN B Y, DONG W Y. A facile route for the fabrication of CeO2 nanosheets via controlling the morphology of CeOHCO3 precursors[J]. J Cryst Growth, 2012, 343(1):95-100. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=1a7175ba95f73fb57caab45c589265ea [14] HU F Y, CHEN J J, PENG Y, SONG H, LI K Z, LI J H. Novel nanowire self-assembled hierarchical CeO2 microspheres for low temperature toluene catalytic combustion[J]. Chem Eng J, 2018, 331:425-434. doi: 10.1016/j.cej.2017.08.110 [15] ZHOU K B, WANG X, SUN X M, PENG Q, LI Y D. Enhanced catalytic activity of ceria nanorods from well-defined reactive crystal planes[J]. J Catal, 2005, 229(1):206-212. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=a50b9779b191d19a996ddce2039b807d [16] SI R, FLYTZANI-STEPHANOPOULOS M. Shape and crystal-plane effects of nanoscale ceria on the activity of Au-CeO2 catalysts for the water-gas shift reaction[J]. Angew Chem Int Ed, 2008, 47(15):2884-2887. doi: 10.1002/anie.200705828 [17] MAITARAD P, HAN J, ZHANG D, SHI L, NAMUANGRUK S, RUNGROTMONGKOL T. Structure-activity relationships of NiO on CeO2 nanorods for the selective catalytic reduction of NO with NH3:Experimental and DFT studies[J]. J Phys Chem C, 2014, 118(118):9612-9620. https://www.researchgate.net/publication/262188283_Structure-Activity_Relationships_of_NiO_on_CeO_2_Nanorods_for_the_Selective_Catalytic_Reduction_of_NO_with_NH_3_Experimental_and_DFT_Studies [18] ZHANG X, HOUSE S D, TANG Y, NGUYEN L, LI Y, OPALADE A A, YANG J C, SUN Z, FENG TAO F. Complete oxidation of methane on NiO nanoclusters supported on CeO2 nanorods through synergistic effect[J]. ACS Sustainable Chem Eng, 2018, 6(5):6467-6477. doi: 10.1021/acssuschemeng.8b00234 [19] TANG C J LI J C, YAO X J, SUN J F, CAO Y, ZHANG L, GAO F, DENG Y, DONG L. Mesoporous NiO-CeO2 catalysts for CO oxidation:Nickel content effect and mechanism aspect[J]. Appl Catal A:Gen, 2015, 494:77-86. doi: 10.1016/j.apcata.2015.01.037 [20] 李树娜, 宋佩, 张金丽, 贺小霞, 解一昕, 张亚刚, 王瑞义, 李志凯, 朱华青. CeO2-MnOx催化剂形貌对低浓度甲烷催化燃烧反应性能的影响[J].燃料化学学报, 2018, 46(5):615-624. doi: 10.3969/j.issn.0253-2409.2018.05.015LI Shu-na, SONG Pei, ZHANG Jin-li, HE Xiao-xia, XIE Yi-xin, ZHANG Ya-gang, WANG Rui-yi, LI Zhi-kai, ZHU Hua-qing. Morphological effect of CeO2-MnOx catalyst on their catalytic performance in lean methane combustion[J]. J Fuel Chem Technol, 2018, 46(5):615-624. doi: 10.3969/j.issn.0253-2409.2018.05.015 [21] LI S N, ZHU H Q, QIN Z F, WANG G F, ZHANG Y G, WU Z W, LI Z K, CHEN G, DONG W W, WU Z H, ZHENG L R, ZHANG J, HU T D, WANG J G. Morphologic effects of nano CeO2-TiO2 on the performance of Au/CeO2-TiO2 catalysts in low-temperature CO oxidation[J]. Appl Catal B:Environ, 2014, 144(2):498-506. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=0df82c10c28f8fe7ecf202c8e8090c7c [22] 孙敬方, 葛成艳, 姚小江, 曹原, 张雷, 汤常金, 董林.固相浸渍法制备NiO/CeO2催化剂及其在CO氧化反应中的应用[J].物理化学学报, 2013, 29(11):2451-2458. doi: 10.3866/PKU.WHXB201309041SUN Jing-fang, GE Cheng-yan, YAO Xiao-jiang, CAO Yuan, ZHANG Lei, TANG Chang-jin, DONG Lin. Preparation of NiO/CeO2 catalysts by solid state impregnation and their application in CO oxidation[J]. Acta Phys-Chim Sin, 2013, 29(11):2451-2458. doi: 10.3866/PKU.WHXB201309041 [23] BENJARAM M R, ATAULLAH K, YUSUKE Y, TETSUHIKO K, STÉPHANE L, JEAN-CLAUDE V. Structural characterization of CeO2-MO2 (M=Si4+, Ti4+, and Zr4+) mixed oxides by Raman spectroscopy, X-ray photoelectron spectroscopy, and other techniques[J]. J Phys Chem B, 2003, 107(41):11475-11484. doi: 10.1021/jp0358376 [24] NI X M, ZHAO Q B, ZHOU F, ZHENG H G, CHENG J, LI B B. Synthesis and characterization of NiO strips from a single source[J]. J Cryst Growth, 2006, 289(1):299-302. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f3ac569992f605a12f3c68ef5f46b622 [25] MAHAMMADUNNISA Sk, MANOJ K, LINGAIAH N, SUBRAHMANYAM C. NiO/Ce1-xNixO2-δ as an alternative to noble metal catalysts for CO oxidation[J]. Catal Sci Technol, 2013, 3(3):730-736. doi: 10.1039/C2CY20641B [26] SOLSONA B, CONCEPCIÓN P, HERNÁNDEZ S, DENJAMIN B, LÓPEZ NIETO J. Oxidative dehydrogenation of ethane over NiO-CeO2 mixed oxides catalysts[J]. Catal Today, 2012, 180(1):51-58. doi: 10.1016/j.cattod.2011.03.056 [27] ŚWIATOWSKA J, LAIR V, PEREIRA-NABAIS C, COTE G, MARCUS P, CHAGNES A. XPS, XRD and SEM characterization of a thin ceria layer deposited onto graphite electrode for application in lithium-ion batteries[J]. Appl Surf Sci, 2011, 257(21):9110-9119. doi: 10.1016/j.apsusc.2011.05.108 [28] BEHZAD N, MEHRAN R, EBRAHIM N. Preparation of highly active and stable NiO-CeO2 nanocatalysts for CO selective methanation[J]. Int J Hydrogen Energy, 2015, 40(27):8539-8547. doi: 10.1016/j.ijhydene.2015.04.127 -





 下载:
下载: