Mechanism analysis of synergy behavior during blended char co-gasification of bituminous coal and rice straw
-
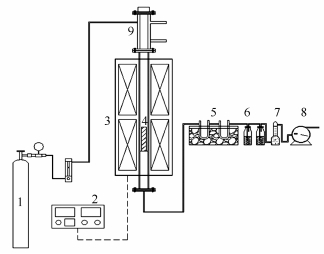
摘要: 基于热重分析仪考察了神府烟煤焦、稻草焦和神府烟煤-稻草混合焦样气化反应活性及共气化过程协同行为。并借助电感耦合等离子体发射光谱仪和扫描电子显微镜-能谱仪联用装置探讨了共气化过程活性矿物组分的迁移转化特性,以关联解释共气化协同行为演变。结果表明,与煤焦单独气化相比,稻草焦掺混有利于提高煤焦整体气化反应活性。混合焦样共气化过程协同行为随碳转化率的提高呈先逐渐减弱的抑制作用,达到某一碳转化率(记为转折碳转化率)后呈不断增强的协同促进作用,且转折碳转化率随气化温度升高而提高。神府烟煤-稻草混合焦样共气化过程协同行为演变主要归因于共气化过程活性K和Ca转化特性的共同影响。神府烟煤-稻草混合焦样共气化整体协同行为呈协同促进作用,并随气化温度的升高而减弱。Abstract: The gasification reactivity of Shenfu bituminous coal char, rice straw char and their blended char and the synergy behavior in blend were studied using TGA. Additionally, inductively coupled plasma emission spectrometer and scanning electron microscopy coupled with energy disperse spectroscopy were employed to explore the active AAEM transformation characteristics in co-gasification process, so as to correlate and explain the synergy behavior variations during co-gasification. The results show that the addition of rice straw char to coal char was favorable for promoting the overall char gasification reactivity compared with individual coal char gasification. The synergy behavior at different co-gasification conversions changes from the gradually decreasing inhibition effect at the early stage of co-gasification to the gradually enhanced synergistic effect when reaching a turning conversion that is increased at higher gasification temperature. The synergy behavior variations during blended char co-gasification are attributed to the combined effect of active K and Ca transformation characteristics in co-gasification process. Furthermore, the overall synergy behavior of blended char co-gasification is shown as a positive synergistic effect, and it is weakened with the increase of gasification temperature.
-
Key words:
- coal /
- biomass /
- co-gasification /
- synergy behavior /
- mechanism
-
表 1 样品的工业分析、元素分析和灰熔融温度
Table 1 Proximate analysis, ultimate analysis and ash fusion temperature of tested samples
Sample Proximate analysis wd/% Ultimate analysis wd /% Ash fusion temperature t/℃ V FC A C H N O S DT ST HT FT SF 35.42 58.29 6.29 79.14 2.32 1.12 10.36 0.77 1152 1167 1175 1179 RS 75.05 15.35 9.60 44.08 6.24 1.13 38.57 0.38 1198 1257 1290 1380 表 2 样品的灰组成
Table 2 Ash composition of tested samples
Sample Ash composition w/% SiO2 Al2O3 K2O Na2O CaO Fe2O3 MgO SF 33.36 12.44 0.67 1.73 27.78 9.11 1.34 RS 58.88 0.18 21.97 1.13 4.20 0.26 2.73 表 3 焦样气化反应活性指数
Table 3 Gasification reactivity index of char samples
Sample R0.9/min-1 800 ℃ 850 ℃ 900 ℃ 950 ℃ SF-800P 0.003 0.011 0.030 0.067 RS-800P 0.008 0.022 0.052 0.115 SF:RS-1:1-800P 0.006 0.019 0.044 0.087 表 4 焦样及混合焦样气化半焦中活性AAEM含量
Table 4 Active AAEM content in chars and gasification semi-chars of blended char
Sample Element content /(mg·g-1) (raw char/blended char) active K-exp active K-cal active Ca-exp active Ca-cal RS-800P 38.80 - 6.36 - SF-800P 0.12 - 35.38 - SF:RS-1:1-800P 11.72 - 26.67 - SF:RS-1:1-800P-10% 7.84 10.72 12.61 14.71 SF:RS-1:1-800P-30% 7.46 7.70 12.22 14.30 SF:RS-1:1-800P-50% 6.42 1.84 12.01 14.08 SF:RS-1:1-800P-70% 4.19 0.76 11.66 13.86 SF:RS-1:1-800P-90% 1.75 0.31 10.96 13.50 note: SF:RS-1:1-800P-x% represents semi-char (conversion, x%) of SF:RS-1:1-800P 表 5 神府煤焦及混合焦样气化半焦中神府煤焦颗粒表面元素组成
Table 5 Elemental composition of the particle surface of SF-800P and SF gasification semi-char of SF:RS-1:1-800P
Sample Elemental composition /% C O Si K K/Si RS-800P-epidermis 19.50 44.29 33.21 0.78 0.02 RS-800P-ground tissue 88.81 8.53 0.36 1.04 2.89 SF-800P 86.38 6.57 0.26 0.09 0.35 SF:RS-1:1-800P-50%-SF 78.49 11.64 0.46 0.83 1.80 -
[1] 王辅臣, 于广锁, 龚欣, 刘海峰, 王亦飞, 周志杰, 陈雪莉.大型煤气化技术的研究与发展[J].化工进展, 2009, 28(2):173-180. doi: 10.3969/j.issn.1008-4800.2015.09.056WANG Fu-chen, YU Guang-suo, GONG Xin, LIU Hai-feng, WANG Yi-fei, ZHOU Zhi-jie, CHEN Xue-li. Research and development of large-scale coal gasification technology[J]. Chem Ind Eng Prog, 2009, 28(2):173-180. doi: 10.3969/j.issn.1008-4800.2015.09.056 [2] 任海君, 张永奇, 房倚天, 王洋.煤焦与生物质焦共气化反应特性研究[J].燃料化学学报, 2012, 40(2):143-148. http://rlhxxb.sxicc.ac.cn/CN/Y2012/V40/I02/143REN Hai-jun, ZHANG Yong-qi, FANG Yi-tian, WANG Yang. Co-gasification properties of coal char and biomass char[J]. J Fuel Chem Technol, 2012, 40(2):143-148. http://rlhxxb.sxicc.ac.cn/CN/Y2012/V40/I02/143 [3] RIZKIANA J, GUAN G Q, WIDAYATNO W B, HAO X G, HUANG W, TSUTSUMI A, ABUDULA A. Effect of biomass type on the performance of co-gasification of low rank coal with biomass at relatively low temperatures[J]. Fuel, 2014, 134:414-419. doi: 10.1016/j.fuel.2014.06.008 [4] 陈鸿伟, 杨新, 韩悦, 赵振虎.制焦条件对稻秆与大同烟煤共气化特性影响的实验研究[J].热能动力工程, 2017, 32(8):94-99. http://mall.cnki.net/magazine/magadetail/RNWS201708.htmCHEN Hong-wei, YANG Xin, HAN Yue, ZHAO Zhen-hu. Experimental study on the influence of pyrolysis condition on co-gasification characteristics of rice straw and Datong bituminous coal[J]. J Eng Therm Energ Power, 2017, 32(8):94-99. http://mall.cnki.net/magazine/magadetail/RNWS201708.htm [5] ZHANG Y, ZHENG Y, YANG M J, SONG Y C. Effect of fuel origin on synergy during co-gasification of biomass and coal in CO2[J]. Bioresour Technol, 2016, 200:789-794. doi: 10.1016/j.biortech.2015.10.076 [6] DING L, ZHANG Y Q, WANG Z Q, HUANG J J, FANG Y T. Interaction and its induced inhibiting or synergistic effects during co-gasification of coal char and biomass char[J]. Bioresour Technol, 2014, 173:11-20. doi: 10.1016/j.biortech.2014.09.007 [7] HABIBI R, KOPYSCINSKI J, MASNADI M S, LAM J, GRACE J R, MIMS C A, HILL J M. Co-gasification of biomass and non-biomass feedstocks:Synergistic and inhibition effects of switchgrass mixed with sub-bituminous coal and fluid coke during CO2 gasification[J]. Energy Fuels, 2012, 27:494-500. https://www.researchgate.net/publication/263656626_Co-gasification_of_Biomass_and_Non-biomass_Feedstocks_Synergistic_and_Inhibition_Effects_of_Switchgrass_Mixed_with_Sub-bituminous_Coal_and_Fluid_Coke_During_CO2_Gasification [8] 邱朋华, 杜昌帅, 刘栗.热解酸洗煤焦结构特性以及与其反应性关系研究[J].煤炭学报, 2017, 42(S1):233-239. http://mall.cnki.net/magazine/magadetail/MTXB2017S1.htmQIU Peng-hua, DU Chang-shuai, LIU Li. Structural characteristics of char derived from acid-washed coal pyrolysis and its corrections with char reactivity[J]. J China Coal Soc, 2017, 42(S1):233-239. http://mall.cnki.net/magazine/magadetail/MTXB2017S1.htm [9] CHEN H D, CHEN X L, QIAO Z, LIU H F. Release and transformation characteristics of K and Cl during straw torrefaction and mild pyrolysis[J]. Fuel, 2016, 167:31-39. doi: 10.1016/j.fuel.2015.11.059 [10] KANG K, AZARGOHAR R, DALAI A K, WANG H. Hydrogen production from lignin, cellulose and waste biomass via supercritical water gasification:Catalyst activity and process optimization study[J]. Energy Convers Manage, 2016, 117:528-537. doi: 10.1016/j.enconman.2016.03.008 [11] MARCHAND D J, SCHNEIDER E, WILLIAMS B P, JOO Y L, KIM J, KIM G T, KIM S H. Physical and chemical changes of coal during catalytic fluidized bed gasification[J]. Fuel Process Technol, 2015, 130:292-298. doi: 10.1016/j.fuproc.2014.10.039 [12] GIL M V, RIAZA J, ÁLVAREZ L, PEVIDA C, RUBIERA F. Biomass devolatilization at high temperature under N2 and CO2:Char morphology and reactivity[J]. Energy, 2015, 91:655-662. doi: 10.1016/j.energy.2015.08.074 [13] HUO W, ZHOU Z J, CHEN X L, DAI Z H, YU G S. Study on CO2 gasification reactivity and physical characteristics of biomass, petroleum coke and coal chars[J]. Bioresour Technol, 2014, 159:143-149. doi: 10.1016/j.biortech.2014.02.117 [14] MA Z, BAI J, LI W, BAI Z Q, KONG L X. Mineral transformation in char and its effect on coal char gasification reactivity at high temperatures, Part 1:Mineral transformation in char[J]. Energy Fuels, 2013, 27:4545-4554. doi: 10.1021/ef4010626 [15] 向银花, 王洋, 张建民, 董众兵, 李斌.煤焦气化过程中比表面积和孔容积变化规律及其影响因素研究[J].燃料化学学报, 2002, 30(2):108-112. http://d.wanfangdata.com.cn/Periodical_rlhxxb200202003.aspxXIANG Yin-hua, WANG Yang, ZHANG Jian-min, DONG Zhong-bing, LI Bin. Study on structural properties and their affecting factors during gasification of chars[J]. J Fuel Chem Technol, 2002, 30(2):108-112. http://d.wanfangdata.com.cn/Periodical_rlhxxb200202003.aspx [16] LI S, WHITTY K J. Physical phenomena of char-slag transition in pulverized coal gasification[J]. Fuel Process Technol, 2012, 95(2):127-136. https://www.sciencedirect.com/science/article/pii/S0378382011004176 [17] 张凯, 汤达祯, 陶树, 刘彦飞, 陈世达.不同变质程度煤吸附能力影响因素研究[J].煤炭科学技术, 2017, 45(5):192-197. http://www.cnki.com.cn/Article/CJFDTotal-MKKC201501006.htmZHANG Kai, TANG Da-zhen, TAO Shu, LIU Yan-fei, CHEN Shi-da. Study on influence factors of adsorption capacity of different metamorphic degree coals[J]. Coal Sci Technol, 2017, 45(5):192-197. http://www.cnki.com.cn/Article/CJFDTotal-MKKC201501006.htm [18] OKUNO T, SONOYAMA N, HAYASHI J, LI C Z, SATHE C, CHIBA T. Primary release of alkali and alkaline earth metallic species during the pyrolysis of pulverized biomass[J]. Energy Fuels, 2005, 19:2164-2171. doi: 10.1021/ef050002a [19] LU X C, LI F C, WASTON A T. Adsorption measurements in devomianshales[J]. Fuel, 1995, 74:599-603. doi: 10.1016/0016-2361(95)98364-K -





 下载:
下载: