Effect of extraction temperature on depolymerization characteristics of ethanol organosolv lignin in supercritical ethanol
-
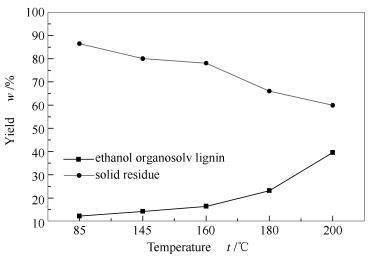
摘要: 通过控制溶出温度(160、180和200℃),制备了三种毛竹乙醇木质素EOL(EOL-160、EOL-180和EOL-200),并采用红外光谱仪(FT-IR)、凝胶渗透色谱(GPC)和热重分析(TG)分别对乙醇木质素的官能团、分子量分布及热稳定性进行了分析;采用微型高温高压反应釜,在超临界乙醇体系,进行三种乙醇木质素的解聚实验,通过气相色谱/质谱联用仪(GC/MS)及FT-IR对解聚产物进行分析,探讨乙醇木质素特性对其超临界乙醇环境解聚产物组成的影响规律。结果表明,随着溶出温度的升高,乙醇木质素溶出率呈上升趋势。EOL-160、EOL-180和EOL-200三种乙醇木质素在超临界条件解聚液相产物主要成分分别为对乙基苯酚、2,6-二甲氧基苯酚和4-羟基-3-甲氧基-苯甲酸酯。Abstract: Effect of extraction temperature (160, 180 and 200 ℃) on depolymerization characteristics of ethanol organosolv lignin (EOL) was investigated with a micro autoclave reactor. Three different EOLs (EOL-160, EOL-180 and EOL-200) were prepared by controlling extraction temperature, and their properties including functional groups, molecular weight distribution and thermogravimetric properties were analyzed by infrared spectroscopy (FT-IR), gel permeation chromatography (GPC) and thermogravimetric analysis (TG). The liquid and solid products obtaining from EOLs depolymerized in supercritical ethanol were analyzed by gas chromatography/mass spectrometry (GC/MS) and FT-IR, respectively. The results show that the main phenolic compound product for EOL-160, EOL-180 and EOL-200 depolymerized in supercritical ethanol were paraethyl phenol, 2,6-dimethoxy phenol and 4-hydroxy-3-methoxy-benzoate, respectively.
-
表 1 毛竹的基础化学成分分析
Table 1 Fundamental chemical component analysis of bamboo

表 2 不同温度下溶出的木质素的分子量及其分子量分布
Table 2 Molecular weight and its distribution of ethanol organosolv lignin

表 3 乙醇木质素超临界乙醇解聚液相产物组成的GC/MS分析
Table 3 GC/MS of ethanol organosolv lignin depolymerization in supercritical ethanol

-
[1] KIFAYAT U, MUSHTAQ A, VINOD K, LU P, ADAM H, MUHAMMAD Z, SHAZIA S. Assessing the potential of algal biomass opportunities for bioenergy industry:A review[J]. Fuel, 2015, 143(4):414-423. http://www.sciencedirect.com/science/article/pii/S001623611401059X [2] 孙勇, 李佐虎, 萧煌炘.木质素分离方法的研究进展[J].中国制浆造纸工业, 2005, 26(10):58-61. http://www.cnki.com.cn/Article/CJFDTOTAL-COKE200510018.htmSUN Yong, LI Zuo-hu, XIAO Huang-xin. The research progress of lignin separation methods[J]. China Pulp Pap Ind, 2005, 26(10):58-61. http://www.cnki.com.cn/Article/CJFDTOTAL-COKE200510018.htm [3] ZHANG J G, HIROYUKI A, JEAPHIANNE V R. Highly efficient, NiAu-catalyzed hydrogenolysis of lignin into phenolic chemicals[J]. Green Chem, 2014, 16(5):2432-2437. doi: 10.1039/C3GC42589D [4] 陈梦薇, 郭大亮, 王林芳, 薛国新.碱木质素超/亚临界乙醇体系解聚机理研究[J].燃料化学学报, 2016, 44(10):1203-1210. doi: 10.3969/j.issn.0253-2409.2016.10.007CHEN Meng-wei, GUO Da-liang, WANG Lin-fang, XUE Guo-xin. Study on the depolymerization mechanism of alkali lignin in sub-and supercritical sthanol[J]. J Fuel Chem Technol, 2016, 44(10):1203-1210. doi: 10.3969/j.issn.0253-2409.2016.10.007 [5] YUAN Z S, CHENG S, LEITCH M. Hydrolytic degradation of alkaline lignin in hot-compressed water and ethanol[J]. Bioresour Technol, 2010, 101(23):9308-9313. doi: 10.1016/j.biortech.2010.06.140 [6] PARK S Y, CHANG Y H, HAN S J. Improvement of oil properties by combination of organic solvents and formic acid during supercritical depolymerization[J]. J Anal Appl Pyrolysis, 2016, 121:113-120. doi: 10.1016/j.jaap.2016.07.011 [7] ANA T, LUIS S, JALEL L. Improving base catalyzed lignin depolymerization byavoiding lignin repolymerization[J]. Fuel, 2014, 116:617-624. doi: 10.1016/j.fuel.2013.08.071 [8] GOSSELINK R J, TEUNISSEN W, VAN D J, DE J E, GELLERSTEDT G, SCOTT E L. Lignin depolymerisation in supercritical carbon dioxide/acetone/water fluid for the production of aromatic chemicals[J]. Bioresour Technol, 2012, 106:173-177. doi: 10.1016/j.biortech.2011.11.121 [9] YUE Y Y, YU Z, JUAN F, JIE C. Novel method for production of phenolics by combining lignin extraction with lignin depolymerization in aqueous ethanol[J]. Ind Eng Chem Res, 2012, 51(1):103-110. doi: 10.1021/ie202118d [10] QIU L Y, JIAN B S, LU L. Characterization of structural changes of lignin in the process of cooking of bagasse with solid alkali and active oxygen as a pretreatment for lignin conversion[J]. Energy Fuels, 2012, 26(11):6999-7004. doi: 10.1021/ef300983h [11] 娄瑞. 非木材纤维木质素的热裂解特性与产物形成的途径调控[D]. 广州: 华南理工大学, 2011.LOU Rui. Thermal cracking characteristics of non-wood fiber lignin and product form the way of control[D]. Guangzhou:South China University of Technology, 2011. [12] YANZ G, JING H Z, JIA L W, GUANG W S, YI J S. Structural transformations of triploid of Populus tomentosa Carr. Lignin during auto-catalyzed ethanol organosolv pretreatment[J]. Ind Crop Prod, 2015, 76:522-529. doi: 10.1016/j.indcrop.2015.06.020 [13] MAURICIO Y S, BETTY M, CAROLINA N, SHAOBO P. Physicochemical characterization of ethanol organosolv lignin (EOL) from Eucalyptus globulus:Effect of extraction conditions on the molecular structure[J]. Polym Degrad Stabil, 2014, 110(1s):184-194. http://www.cabdirect.org/abstracts/20153155786.html [14] MAHMOOD N, YUAN Z, SCHMIDT J, XU C C. Hydrolytic depolymerization of hydrolysis lignin:Effects of catalysts and solvents[J]. Bioresour Technol, 2015, 190:416-419. doi: 10.1016/j.biortech.2015.04.074 [15] MIAO W, JIN H P, XUE M Z. Enhancement of lignin biopolymer Isolation from hybrid poplar by organosolv pretreatments[J]. Int J Polym Sci, 2014, (3):1-10. https://www.researchgate.net/publication/270673139_Enhancement_of_Lignin_Biopolymer_Isolation_from_Hybrid_Poplar_by_Organosolv_Pretreatments -





 下载:
下载: