Promoting effect of oxidation reaction on steam gasification reaction in Shengli lignite gasification process Ⅰ: Macroscopic reaction characteristic
-
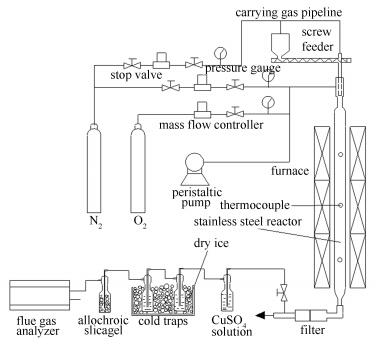
摘要: 在φ80×3 000 mm耐高温不锈钢管气流床反应器中,以150-180 μm胜利褐煤为气化原料,考察了800和900℃时添加氧气前后褐煤转化率的变化,研究了氧化反应对水蒸气气化反应影响的宏观特征。结果表明,添加氧气后褐煤转化率明显大于O2和H2O气氛下褐煤转化率之和,即向水蒸气气氛添加氧气后褐煤转化率的增幅大于氧气氧化作用导致的褐煤转化率的增幅,随着H2O含量增大以及温度的升高此现象愈加明显。该协同作用主要是氧化反应对水蒸气气化反应的促进作用造成的。利用φ40×200 mm石英圆筒流化床反应器进行了类似的实验,也发现了该协同作用。同时,借鉴收缩核模型并结合气流床气化实验条件推导了水蒸气气化宏观动力学方程,得到的速率方程$ (Z-{{(1-x)}^{\frac{1}{3}}})=\frac{t\beta {{k}_{{{\text{H}}_{2}}\text{O}}}}{R{{\rho }_{\text{C}}}}{{\varphi }_{{{\text{H}}_{2}}\text{O}}}={{K}_{{{\text{H}}_{2}}\text{O}}}{{\varphi }_{{{\text{H}}_{2}}\text{O}}} $与实验值吻合较好,添加氧气后水蒸气气化反应速率和水蒸气气化反应表观速率常数KH2O明显增大,这是氧气对水蒸气气化反应促进作用的动力学特征。Abstract: Shengli brown coal in 150-180 μm was gasified at 800-900℃ in a simulated entrained-flow reactor, φ80×3 000 mm. Conversion and kinetics of steam gasification reaction of the lignite were discussed to investigate synergistic effects of oxidation reaction on steam gasification reaction. The results show that lignite conversion under H2O+1%O2 atmospheres is greater significantly than the sum of that under H2O atmosphere and 1%O2 atmosphere, i.e., the increase of lignite conversion from H2O atmosphere to H2O+1%O2 atmospheres is greater than that from N2 atmosphere to N2+1%O2 atmospheres. The synergistic effects are caused by promoting effect of oxidation reaction on steam gasification reaction, and are more obvious as H2O content increasing and temperature rising. Moreover, the similar experiments were carried out in φ40×200 mm cylindrical quartz fluidized bed, and the synergistic effects are also found. The steam gasification reaction rate equation, $ (Z-{{(1-x)}^{\frac{1}{3}}})=\frac{t\beta {{k}_{{{\text{H}}_{2}}\text{O}}}}{R{{\rho }_{\text{C}}}}{{\varphi }_{{{\text{H}}_{2}}\text{O}}}={{K}_{{{\text{H}}_{2}}\text{O}}}{{\varphi }_{{{\text{H}}_{2}}\text{O}}} $, is in good agreement with experimental data. This indicates that the apparent rate constant KH2O increases obviously after O2 adding to water vapor, which is the kinetic characteristics of promoting effect of oxidation reaction on steam gasification reaction.
-
Key words:
- mild gasification /
- synergistic effect /
- steam gasification /
- conversion rate /
- kinetics
-
表 1 胜利褐煤的工业分析和元素分析
Table 1 Proximate and ultimate analysis of material
Material Proximate analysis wad/% Elemental analysis wdaf/% M A V FC C H S O N Shengli lignite 5.89 9.87 36.23 53.90 62.26 6.12 0.66 29.85 1.11 -
[1] 王毅.块状褐煤高温蒸汽热解的宏细观特性分析及应用[M].徐州:中国矿业大学出版社, 2012.WANG Yi. Macro & Micro Characteristic Analysis and Application for Massive Lignite High Temperature Steam Pyrolysis[M]. Xuzhou:China University of Mining and Technology Press, 2012. [2] 戴和武, 谢可玉.褐煤利用技术[M].北京:煤炭工业出版社, 1999.DAI He-wu, XIE Ke-yu. Brown Coal Utilization Technology[M]. Beijing:China Coal Industry Publishing House, 1999. [3] LI C Z. Importance of volatile-char interactions during the pyrolysis and gasification of low-rank fuels-A review[J]. Fuel, 2013, 112:609-623. doi: 10.1016/j.fuel.2013.01.031 [4] TAY H L, KAJITAANI S, ZHANG S, LI C Z. Effects of gasifying agent on the evolution of char structure during the gasification of Victorian brown coal[J]. Fuel, 2013, 103:22-28. doi: 10.1016/j.fuel.2011.02.044 [5] 周晨亮, 刘全生, 李阳, 智科端, 滕英跃, 宋银敏.胜利褐煤水蒸气气化制富氢合成气及其固有矿物质的催化作用[J].化工学报, 2013, 64(6):2092-2102. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ201306031.htmZHOU Chen-liang, LIU Quan-sheng, LI Yang, ZHI Ke-duan, TENG Ying-yue, SONG Yin-min. Production of hydrogen-rich syngas by steam gasification of Shengli lignite and catalytic effect of inherent minerals[J]. CIESC, 2013, 64(6):2092-2102. http://www.cnki.com.cn/Article/CJFDTOTAL-HGSZ201306031.htm [6] 任海君, 张永奇, 房倚天, 黄戒介, 王洋.褐煤焦中矿物质对气化动力学的影响[J].化学工程, 2010, 38(10):132-135. http://www.cnki.com.cn/Article/CJFDTOTAL-IMIY201010030.htmREN Hai-jun, ZHANG Yong-qi, FANG Yi-tian, HUANG Jie-jie, WANG Yang. Effect of minerals in lignite char on kinetics of steam gasification[J]. Chem Eng (China), 2010, 38(10):132-135. http://www.cnki.com.cn/Article/CJFDTOTAL-IMIY201010030.htm [7] CRNOMARKOVIC N, REPIC B, MLADENOVIC R, NESKOVIC O, VELJKOVIC M. Experimental investigation of role of steam in entrained flow coal gasification[J]. Fuel, 2007, 86(1):194-202. https://www.researchgate.net/publication/222072017_Experimental_investigation_of_role_of_steam_in_entrained_flow_coal_gasification [8] LEE J G, KIM J H, LEE H G. Characteristics of entrained flow coal gasification in a drop tube reactor[J]. Fuel, 1996, 75(9):1035-1042. doi: 10.1016/0016-2361(96)00084-1 [9] TAY H L, LI C Z. Changes in char reactivity and structure during the gasification of a Victorian brown coal:Comparison between gasification in O2 and CO2[J]. Fuel Process Technol, 2010, 91(8):800-804. doi: 10.1016/j.fuproc.2009.10.016 [10] LI T T, ZHANG L, DONG L, LI C Z. Effects of gasification atmosphere and temperature on char structural evolution during the gasification of Collie sub-bituminous coal[J]. Fuel, 2014, 117(part B):1190-1195. [11] ZHANG S, MIN Z H, TAY H L, ASADULLAH M, LI C Z. Effects of volatile-char interactions on the evolution of char structure during the gasification of Victorian brown coal in steam[J]. Fuel, 2011, 90(4):1529-1535. doi: 10.1016/j.fuel.2010.11.010 [12] WU H W, LI X J, HAYASHI J I, CHIBA T, LI CZ. Effects of volatile-char interactions on the reactivity of charsfrom NaCl-loaded Loy Yang brown coal[J]. Fuel, 2005, 84(10):1221-1228. doi: 10.1016/j.fuel.2004.06.037 [13] ZHANG S, HAYASHI J I, LI C Z.Volatilization and catalytic effects of alkali and alkaline earth metallic species during the paralysis and gasification of Victorian brown coal. Part IX. Effects of volatile-charinteractions on char-H2O and char-O2reactivates[J]. Fuel, 2011, 90(4):1655-1661. doi: 10.1016/j.fuel.2010.11.008 [14] WANG F J, ZHANG S, CHEN Z D, LIU C, WANG Y G. Tar reforming using char as catalyst during pyrolysis and gasification of Shengli brown coal[J]. J Anal Appl Pyrolysis, 2014, 105:269-275. doi: 10.1016/j.jaap.2013.11.013 [15] KOMAROVA E, GUHL S, MEYER B. Brown coal char CO2-gasification kinetics with respect to the char structure. Part Ⅰ:Char structure development[J]. Fuel, 2015, 152:38-47. doi: 10.1016/j.fuel.2015.01.107 [16] KAJITANI S, TAY H L, ZHANG S, LI C Z. Mechanisms and kinetic modeling of steam gasification of brown coal in the presence of volatile-char interactions[J]. Fuel, 2013, 103:7-13. doi: 10.1016/j.fuel.2011.09.059 [17] 王永刚, 孙加亮, 张书.反应气氛对褐煤气化反应性及半焦结构的影响[J].煤炭学报, 2014, 39(8):1765-1771. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201408051.htmWANG Yong-gang, SUN Jia-liang, ZHANG Shu. Impacts of the gas atmosphere on the gasification reactivity and char structure of the brown coal[J]. J China Coal Soc, 2014, 39(8):1765-1771. http://www.cnki.com.cn/Article/CJFDTOTAL-MTXB201408051.htm [18] 贺永德.现代煤化工技术手册[M]. 2版.北京:化学工业出版社, 2011.HE Yong-de. Modern Coal Chemical Industry Technical Manuals[M]. 2nd ed. Beijing:Chemical Industry Press, 2001. [19] LONG F J, SYKES K W.The mechanism of the steam-carbon reaction[J]. Proc R Soc, London, 1948, A193:377-99. [20] WEN C Y, LEE E S. Coal Conversion Technology[M]. New Jersey:Addisonwesley Publishing Co, Inc, 1979. [21] KWON T W, KIM J R, KIM S D, PARK W H. Catalytic steam gasification of lignite char[J]. Fuel, 1988, 68(4):416-421. [22] MATSUI I, KUNII D, FURUSAWA T. Study of fluidized bed steam gasification of char by thermogravimetrically obtained kinetics[J]. J Chem Eng Jpn, 1985, 18(2):105-113. doi: 10.1252/jcej.18.105 -





 下载:
下载: