Adsorptive separation properties of CO2/CH4/N2 on UZM-9 synthesized by seed-assisted method
-
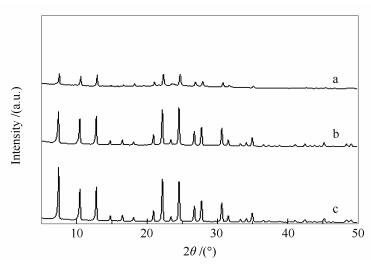
摘要: 以TEAOH和TMAOH为有机模板剂,酸处理的UZM-9分子筛为晶种,采用水热法在48 h内合成出分子筛UZM-9,并对其CO2/CH4/N2的吸附分离性能进行了研究。采用XRD、ICP、TG、SEM与气体吸附等手段对晶种法合成的UZM-9分子筛结构、耐水稳定性与吸附性能进行了研究。结果表明,晶种法可以在2 d内合成出硅铝原子比在3以上、收率达到65%的UZM-9分子筛;所得分子筛的CO2吸附容量可以达到5 mmol/g以上,吸附热为34 kJ/mol,CO2/CH4、CO2/N2与CH4/N2的平均分离因子分别为100、240与2.4,CO2分离性能优良且具有一定耐水性能。Abstract: The UZM-9 zeolite was synthesized in 48 h via a seed-assisted method, in which TEAOH and TMAOH were used as organic structure directing agent (OSDA) while acid-treated UZM-9 zeolites were employed as seeds. The textural properties and hydrophobicity of the as synthesized UZM-9 zeolites and their adsorption properties for CO2/CH4/N2 were characterized by XRD, physical adsorption, SEM, ICP and TG, respectively. The results show that UZM-9 zeolites can be obtained in 2 d with the Si/Al atomic ratio up to 3 and the yield up to 65%. At 298 K and 1atm, the capacity and adsorption heat of CO2 are 5 mmol/g and 34 kJ/mol, and the selectivity of CO2/CH4, CO2/N2 and CH4/N2 are 100, 240 and 2.4 respectively. These results reveal that the as synthesized UZM-9 zeolites have good performances for carbon capture and possess considerable water resistance.
-
Key words:
- acid-treated seed /
- UZM-9 /
- adsorptive separation properties /
- CO2 /
- adsorption heat
-
表 1 分子筛的结构表征及收率
Table 1 Characteristic results and yields of zeolites

表 2 水处理前后分子筛0.3-UZM-9孔结构的变化
Table 2 The textural properties of 0.3-UZM-9 before and after water treatment

表 3 水处理前后分子筛0.3-UZM-9的分离性能
Table 3 Separation performances of 0.3-UZM-9 zeolite before and after water treatment

-
[1] TAGLIABUE M, FARRUSSENG D, VALENCIA S, AGUADO S, RAVON U, RIZZO C, CORMA A, MIRODATOS C. Natural gas treating by selective adsorption:Material science and chemical engineering interplay[J]. Chem Eng J, 2009, 155(3):553-566. doi: 10.1016/j.cej.2009.09.010 [2] BAO Z B, YU L, REN Q, LU X Y, DENG S G. Adsorption of CO2 and CH4 on a magnesium-based metal organic framework[J]. J Colloid Interface Sci, 2011, 353(2):549-556. doi: 10.1016/j.jcis.2010.09.065 [3] RUFFORD T E, SMART S, WATSON G C Y, GRAHAM B F, BOXALL J, DINIZ DA COSTA J C, MAY E F. The removal of CO2 and N2 from natural gas:A review of conventional and emerging process technologies[J]. J Pet Sci Eng, 2012, 94:123-154. http://www.sciencedirect.com/science/article/pii/S0920410512001581 [4] 陈兆元.气体深冷分离工[M].北京:化学工业出版社, 2005.CHEN Zhao-yuan, Gas Deep Cooling Method[M]. Beijing:Chemical Industry Press, 2005. [5] 王学松.气体膜技术[M].北京:化学工业出版社, 2010.WANG Xue-song. Membrane Technology[M]. Beijng:Chemical Industry Press, 2010. [6] 樊栓狮, 程宏远, 陈光进.水合物法分离技术研究[J].现代化工, 1999, 19(2):11-12. http://www.cnki.com.cn/Article/CJFDTOTAL-XDHG902.003.htmFAN Shuan-shi, CHENG Hong-yuan, CHEN Guang-jin. Separation technique based on gas hydrate formation[J]. Mod Chem Ind, 1999, 19(2):11-12. http://www.cnki.com.cn/Article/CJFDTOTAL-XDHG902.003.htm [7] 吕碧洪, 金佳佳, 张莉, 李伟.有机胺溶液吸收CO2的研究现状及进展[J].石油化工, 2011, 40(8):803-809. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=syhg201108000&dbname=CJFD&dbcode=CJFQLV Bi-hong, JIN Jia-jia, ZHANG Li, LI Wei. A systematic review of CO2 adsorption using amine[J]. Petrochem Technol, 2011, 40(8):803-809. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=syhg201108000&dbname=CJFD&dbcode=CJFQ [8] 王洪梅, 罗仕忠, 吴永永, 孙思, 李通.改性硅胶吸附剂用于CO2/CH4吸附分离的研究[J].天然气化工:C1化学与化工, 2012, 37(5):1-5. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=trqh201205000&dbname=CJFD&dbcode=CJFQWANG Hong-mei, LUO Shi-zhong, WU Yong-yong, SUN Si, LI Tong. Study on modified silica gels and their adsorption properties for CO2/CH4 mixed gas separation[J]. Nat Gas Chem Ind:C1, 2012, 37(5):1-5. http://kns.cnki.net/KCMS/detail/detail.aspx?filename=trqh201205000&dbname=CJFD&dbcode=CJFQ [9] ROCHA L A M, ANDREASSEN K A, GRANDE C A. Separation of CO2/CH4 using carbon molecular sieve (CMS) at low and high pressure[J]. Chem Eng Sci, 2017, 164:148-157. doi: 10.1016/j.ces.2017.01.071 [10] 张薄, 辜敏, 鲜学福.碳分子筛的结构和表面性质对其吸附分离CH4/N2和CO2/N2的影响]J].功能材料, 2012, 43(20):2858-2862. doi: 10.3969/j.issn.1001-9731.2012.20.029ZHANG Bo, GU Min, XIAN Xue-fu. Effects structure and surface property on adsorptive separation of carbon molecular sieve for CH4/N2 and CO2/N2[J]. J Funct Mater, 2012, 43(20):2858-2862. doi: 10.3969/j.issn.1001-9731.2012.20.029 [11] HIMENO S, KOMATSU T, FUJITA S. High-pressure adsorption equilibria of methane and carbon dioxide on several activated carbons[J]. J Chem Eng Data, 2005, 50(2):369-376. doi: 10.1021/je049786x [12] PENG X, WANG W, XUE R, SHEN Z. Adsorption separation of CH4/CO2 on mesocarbon microbeads:experiment and modeling[J]. AIChE J, 2006, 52(3):994-1003. doi: 10.1002/(ISSN)1547-5905 [13] BAI B C, CHO S, YU H R, YI K B, KIM K D, LEE Y S. Effects of aminated carbon molecular sieves on breakthrough curve behavior in CO2/CH4separation[J]. J Ind Eng Chem, 2013, 19(3):776-783. doi: 10.1016/j.jiec.2012.10.016 [14] HAO S, ZHANG J, ZHONG Y, ZHU W. Selective adsorption of CO2 on amino-functionalized silica spheres with centrosymmetric radial mesopores and high amino loading[J]. Adsorpt, 2012, 18(5/6):423-430. doi: 10.1007/s10450-012-9428-9 [15] SONGOLZADEH M, SOLEIMANIOl M, TAKHT R M, SONGOLZADEH R. Carbon dioxide separation from flue gases:a technological review emphasizing reduction in greenhouse gas emissions[J]. Sci World J, 2014:828131. http://pubmedcentralcanada.ca/pmcc/articles/PMC3947793/ [16] SUN T, REN X, HU J, WANG S. Expanding pore size of Al-BDC Metal-Organic Frameworks as a way to achieve high adsorption selectivity for CO2/CH4 sseparation[J]. J Phys Chem C, 2014, 118(29):15630-15639. doi: 10.1021/jp411536d [17] VENNA S R, CARREON M A. Highly permeable zeolite imidazolate framework-8 membranes for CO2/CH4 separation[J]. J Am Chem Soc, 2009, 132(1):76-78. http://www.ncbi.nlm.nih.gov/pubmed/20014839 [18] PALOMINO M, CORMA A, REY F, VALENCIA S. New insights on CO2-methane separation using LTA zeolites with different Si/Al ratios and a first comparison with MOFs[J]. Langmuir, 2009, 26(3):1910-1917. http://www.ncbi.nlm.nih.gov/pubmed/19757816 [19] LIU B, SMIT B. Molecular simulation studies of separation of CO2/N2, CO2/CH4, and CH4/N2 by ZIFs[J]. J Phys Chem C, 2010, 114(18):8515-8522. doi: 10.1021/jp101531m [20] JAIME G M, GREGORY J L, JANA L G, MARK A M, LISA M R. Crystalline aluminosilicate zeolitic composition:UZM-9:US, 6713041 B1[P]. 2004-3-30. [21] PARK M B, LEE Y, ZHENG A, XIAO F S, NICHOLAS C P, LEWIS G J, HONG S B. Formation pathway for lta zeolite crystals synthesized via a charge density mismatch approach[J]. J Am Chem Soc, 2012, 135(6):2248-2255. doi: 10.1021/ja309831e [22] KIM S H, PARK M B, MIN H K, HONG S B. Zeolite synthesis in the tetraethylammonium-tetramethylammonium mixed-organic additive system[J]. Microporous Mesoporous Mater, 2009, 123(1):160-168. http://www.sciencedirect.com/science/article/pii/S1387181109001772 [23] TOTH J. Thermodynamical model and prediction of gas/solid adsorption isotherms[J]. J Colloid Interface Sci, 2004, 275(1):2-8. doi: 10.1016/j.jcis.2004.02.073 [24] PETER S A, SEBASTIAN J, JASRA R V. Adsorption of nitrogen, oxygen, and argon in mono-, di-, and trivalent cation-exchanged zeolite mordenite[J]. Ind Eng Chem Res, 2005, 44(17):6856-6864. doi: 10.1021/ie050128v [25] PHAM T D, LIU Q, LOBO R F. Carbon dioxide and nitrogen adsorption on cation-exchanged SSZ-13 zeolites[J]. Langmuir, 2012, 29(2):832-839. http://www.ncbi.nlm.nih.gov/pubmed/23249267 [26] DO D D. Adsorption Analysis:Equilibria and Kinetics[M]. London:Imperial CollegePress, 1998. [27] International Zeolite Association, Structure Commission. http://www.iza-structure.org. [28] ZUKAL A, AREAN C O, DELGADO M R, NACHTIGALL P, PULIDO A, MAYEROVÁ J, Ĉejka J. Combined volumetric, infrared spectroscopic and theoretical investigation of CO2 adsorption on Na-A zeolite[J]. Microporous Mesoporous Mater, 2011, 146(1):97-105. http://www.sciencedirect.com/science/article/pii/S1387181111001417 [29] DUNNE J A, RAO M, SIRCAR S, GORTE R J, MYERS A L. Calorimetric heats of adsorption and adsorption isotherms. 2. O2, N2, Ar, CO2, CH4, C2H6, and SF6 on NaX, H-ZSM-5, and Na-ZSM-5 zeolites[J]. Langmuir, 1996, 12(24):5896-5904. doi: 10.1021/la960496r -





 下载:
下载: