Effect of the source of Cu on the structure and performance of Cu-Zn-Al catalysts prepared by complete liquid-phase technology
-
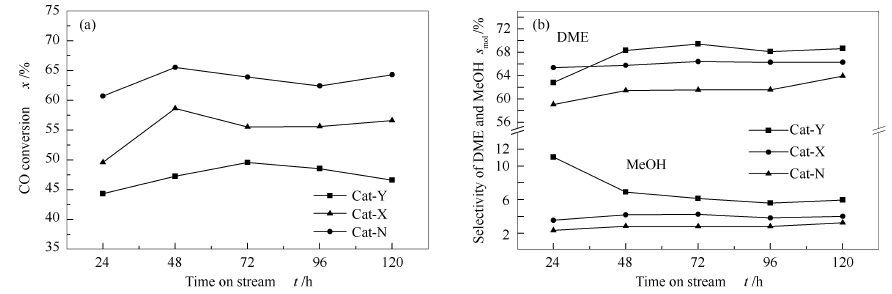
摘要: 采用完全液相法,分别以柠檬酸铜、硝酸铜、乙酸铜为Cu源制备了三种Cu-Zn-Al浆状催化剂,考察了不同铜源对催化剂催化合成气制二甲醚性能的影响,利用XRD、H2-TPR、NH3-TPD、BET、XPS和TEM等技术对催化剂进行了表征。结果表明,铜源对催化剂织构形貌及性能影响显著,用柠檬酸铜为铜源制备的催化剂铜物种分散性最好,铜物种与其他组分间相互作用强,可还原物质的量多,同时催化剂表面弱酸量与强酸量的比较高,催化剂的甲醇脱水能力提升,三种催化剂中柠檬酸铜催化剂性能最好,CO转化率为63.4%,二甲醚选择性为66.0%。
-
关键词:
- 完全液相法 /
- Cu-Zn-Al催化剂 /
- 二甲醚 /
- 柠檬酸铜 /
- 乙酸铜
Abstract: Three kinds of Cu-Zn-Al slurry catalysts were prepared respectively with copper citrate, copper nitrate and copper acetate as the source of copper by complete liquid-phase technology. The effects of different sources of copper on the catalytic performance of dimethyl ether synthesis from syngas were investigated. The catalysts were characterized by XRD, H2-TPR, NH3-TPD, BET, XPS and TEM. The results indicated that the texture morphology and catalytic performance of the catalysts varied significantly. The catalyst prepared with copper citrate showed the best dispersion of Cu species in the catalyst and the largest amount of reducable substance. Meanwhile, the Cu species have strongest interaction with other components. Copper citrate increased the ratio of the amount of weak acid and strong acid on the surface of catalysts, and also improved the performance of the catalyst for methanol dehydration. So the catalytic performance of the catalyst pre-pared with copper citrate was the best. The conversion of CO was 63.4% and the DME selectivity was 66.0%.-
Key words:
- complete liquid-phase technology /
- Cu-Zn-Al catalysts /
- dimethyl ether /
- copper citrate /
- copper acetate
-
表 1 不同催化剂的活性评价
Table 1 Catalytic performance of Cat-N, Cat-X, Cat-Y
Catalyst DME yield/(mg·h-1·gcat-1) CO conversionxmol/% Selectivity smol/% DME MeOH CO2 CH Cat-N 36.84 63.39 66.03 3.97 25.94 4.06 Cat-X 29.93 55.21 61.52 2.82 30.41 5.24 Cat-Y 28.09 47.26 67.45 7.12 20.28 5.15 表 2 催化剂反应前后平均Cu晶粒粒径
Table 2 Copper grain size of different catalysts before and after reaction
Catalyst Cat-N Cat-X Cat-Y DCu /nm(before reaction) 20.0 19.9 25.6 DCu /nm(after reaction) 21.6 22.6 28.6 FWHM(before reaction)/(°) 0.439 0.441 0.348 FWHM(after reaction)/(°) 0.408 0.392 0.315 表 3 催化剂反应前的H2-TPR耗氢量
Table 3 Hydrogen consumption calculated from H2-TPR
Catalyst Reduction temperature t/℃ H2 consumption w/mol Cat-N 255.4 1.634×10-3 Cat-X 245.6 1.257×10-3 Cat-Y 242.9 4.770×10-4 表 4 不同催化剂的织构性质
Table 4 Specific surface area and pore properties of different catalysts
Catalyst Before reaction After reaction BET surface area A/(m2·g-1) pore volumev/(cm3·g-1) average pore diameter d/nm BET surface area A/(m2·g-1) pore volumev/(cm3·g-1) average pore diameter d/nm Cat-N 253.9 0.22 3.43 426.9 0.35 3.27 Cat-X 128.0 0.30 9.21 321.3 0.53 6.55 Cat-Y 392.0 0.30 3.08 457.5 0.34 2.95 表 5 反应前催化剂的XPS和XAES数据
Table 5 XPS and XAES data of fresh catalysts
Catalyst EB/eV EK/eV α′(Cu) Cu 2p3/2 Al 2p Zn 2p3/2 Cu L3VV Zn L3M45M45 Cat-N 932.2 74.2 1 021.5 916.7 987.6 1 848.9 Cat-X 932.5 74.6 1 021.8 916.2 987.5 1 848.7 Cat-Y 932.7 73.8 1 021.4 916.3 987.9 1 849.0 表 6 催化剂反应前表面元素物质的量比
Table 6 Mol ratios between relevant elements on fresh catalysts
Catalyst Mol ratio Cu/Zn Cu/Al (Cu+Zn)/Al Cat-N 1.28 0.08 0.14 Cat-X 1.42 0.09 0.16 Cat-Y 0.43 0.06 0.19 -
[1] 李忠, 谢克昌.现代煤化工技术丛书(第十分册)煤基醇醚燃料[M].北京:化学工业出版社, 2011.LI Zhong, XIE Ke-chang.Modern Coal Chemical Technology Books-Coal Base Methanol-Ether Fue l (Vol.8)[M].Beijing:Chemical Industry Press, 2001. [2] FLEISCH T H, BASU A, GRADASSI M J, MASIN J G.Dimethyl ether:A fuel for the 21st century[J].Stud Surf Sci Catal, 1997, 107(6):117-125. [3] 胡益之, 李洪晋, 韩冬青.21世纪洁净燃料-二甲醚[J].煤化工, 2006, 34(5):10-14. http://www.cnki.com.cn/Article/CJFDTOTAL-MHGZ200605001.htmHU Yi-zhi, LI Hong-jin, HAN Dong-qing.Dimethyl ether-the clean fuel in the 21st century[J].Coal Chem Ind, 2006, 34(5):10-14. http://www.cnki.com.cn/Article/CJFDTOTAL-MHGZ200605001.htm [4] ADACHI Y, KOMOTO M, WATANABE I, OHNO Y, FUJIMOTO K.Effective utilization of remote coal through dimethyl ether synthesis[J].Fuel, 2000, 79(3):229-234. https://www.deepdyve.com/lp/elsevier/effective-utilization-of-remote-coal-through-dimethyl-ether-synthesis-PPS2gmF3Dy [5] FLEISCH T H, BASU A, SILLS R A.Introduction and advancement of a new clean global fuel:The status of DME developments in China and beyond[J].J Nat Gas Sci Eng, 2012, 9(2):94-107. http://linkinghub.elsevier.com/retrieve/articleSelectSinglePerm?Redirect=http%3A%2F%2Fwww.sciencedirect.com%2Fscience%2Farticle%2Fpii%2FS1875510012000650%3Fvia%253Dihub&key=eddc164b80b084b9a4e2deb6a39fbd2e19e1256d [6] ZHOU L, HU S, LI Y, ZHOU Q.Study on co-feed and co-production system based on coal and natural gas for producing DME and electricity[J].Chem Eng J, 2008, 136(1):31-40. doi: 10.1016/j.cej.2007.03.025 [7] 别良伟, 王华.合成气一步法制二甲醚催化剂研究进展[J].工业催化, 2009, 17(2):34-39. http://www.cnki.com.cn/Article/CJFDTOTAL-GYCH200305009.htmBIE Liang-wei, WANG Hua.Advances in developments of the catalysts for one-step synthesis of dimethyl ether from syngas[J].Ind Catal, 2009, 17(2):34-39. http://www.cnki.com.cn/Article/CJFDTOTAL-GYCH200305009.htm [8] 王和平.合成气一步法制二甲醚工艺及催化剂研究进展[J].工业催化, 2003, 11(5):34-38. http://www.cnki.com.cn/Article/CJFDTOTAL-GYCH200305009.htmWANG He-ping.Advances in one-step synthesis of dimethyl ether from syngas[J].Ind Catal, 2003, 11(5):34-38. http://www.cnki.com.cn/Article/CJFDTOTAL-GYCH200305009.htm [9] 谭猗生, 解红娟, 崔海涛, 韩怡卓, 钟炳.浆态床二甲醚合成中V2O5、Sm2O3对脱水组分γ-Al2O3的修饰作用[J].燃料化学学报, 2005, 33(5):602-606. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract16901.shtmlTAN Yi-sheng, XIE Hong-juan, CUI Hai-tao, HAN Yi-zhuo, ZHONG Bing.Effect of V2O5/Sm2O3 modification on alum ina performance for slurry phase dimethyl ether synthesis[J].J Fuel Chem Technol, 2005, 33(5):602-606. http://rlhxxb.sxicc.ac.cn/CN/abstract/abstract16901.shtml [10] PRASAD P S S, BAE J W, KANG S H, LEE Y H, JUN K W.Single-step synthesis of DME from syngas on Cu-ZnO-Al2O3/zeolite bifunctional catalysts:The superiority of ferrierite over the other zeolites[J].Fuel Process Technol, 2008, 89(12):1281-1286. doi: 10.1016/j.fuproc.2008.07.014 [11] MORADI G R, NOSRATI S, YARIPOR F.Effect of the hybrid catalysts preparation method upon direct synthesis of dimethyl ether fromsynthesis gas[J].Catal Commun, 2007, 8(3):598-606. doi: 10.1016/j.catcom.2006.08.023 [12] 黄伟, 高志华, 郝利峰, 阴丽华, 谢克昌.浆态床用催化剂及制备方法:中国, 1613560[P].2005-05-11.HUANG Wei, GAO Zhi-hua, HAO Li-fen, YIN Li-hua, XIE Ke-chang.The liquid phase preparation technology of catalyst used in slurry reactor:CN, 1613560[P].2005-05-11. [13] GAO Z H, HAO L F, WEI H, XIE K C.A novel liquid-phase technology for the preparation of slurry catalysts[J].Catal Lett, 2005, 102(3):139-141. [14] 高志华, 黄伟, 郝利峰, 谢克昌.浆态床用CuO/ZrO2催化剂的完全液相法合成及其对CO加氢的催化性能[J].催化学报, 2006, 27(1):86-90.GAO Zhi-hua, HUANG Wei, HAO Li-feng, XIE Ke-chang.Liquid-phase preparation of CuO/ZrO2 catalyst used in slurry reactors and Its catalytic activity for CO hydrogenation[J].Chin J Catal, 2006, 27(1):86-90. [15] 樊金串, 吴慧, 黄伟, 石宇, 谢克昌.完全液相法制备中原料配比对二甲醚合成催化剂结构和性能的影响[J].催化学报, 2007, 28(12):1062-1066. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA200712010.htmFAN Jin-chuan, WU Hui, HUANG Wei, SHI Yu, XIE Ke-chang.Effect of composition of raw materials on structure and dimethyl ether synthesis activity of Cu-Zn-Al catalyst prepared by complete liquid phase method[J].Chin J Catal, 2007, 28(12):1062-1066. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA200712010.htm [16] GAO Z H, HUANG W, YIN L, XIE K C.Liquid-phase preparation of catalysts used in slurry reactors to synthesize dimethyl ether from syngas:Effect of heat-treatment atmosphere[J].Fuel Process Technol, 2009, 90(12):1442-1446. doi: 10.1016/j.fuproc.2009.06.022 [17] 高志华, 黄伟, 王将永, 阴丽华, 谢克昌.Cu-Zn-Al-Zr合成二甲醚浆状催化剂的完全液相制备及表征[J].化学学报, 2008, 66(3):295-300. http://www.cnki.com.cn/Article/CJFDTOTAL-HXXB200803002.htmGAO Zhi-hua, HUANG Wei, WANG Jiang-yong, YIN Li-hua, XIE Ke-chang.Complete liquid-phase preparation and characterization of Cu-Zn-Al-Zr slurry catalysts for synthesis of dimethyl ether[J].Acta Chim Sin, 2008, 66(3):295-300. http://www.cnki.com.cn/Article/CJFDTOTAL-HXXB200803002.htm [18] FAN J C, CHEN C Q, ZHAO J, HUANG W, XIE K C.Effect of surfactant on structure and performance of catalysts for DME synthesis in slurry bed[J].Fuel Process Technol, 2010, 91(4):414-418. doi: 10.1016/j.fuproc.2009.05.005 [19] 王鹏, 黄伟, 唐钰, 孙凯, 张小雨.完全液相制备中醇溶剂及醇用量对催化剂性能的影响[J].太原理工大学学报, 2013, 44(5):551-556. http://www.cnki.com.cn/Article/CJFDTOTAL-TYGY201305002.htmWANG Peng, HUANG Wei, TANG Yu, SUN Kai, ZHANG Xiao-yu.Effect of alcohols and usage amount on performance of catalyst prepared by complete liquid phase technology[J].J Taiyuan Univ Technol, 2013, 44(5):551-556. http://www.cnki.com.cn/Article/CJFDTOTAL-TYGY201305002.htm [20] 樊金串, 杨瑞卿, 赵杰, 黄伟.液体石蜡体系中含铜物种的化学变化[J].应用化学, 2013, 30(1):67-72. http://www.cnki.com.cn/Article/CJFDTOTAL-YYHX201301013.htmFAN Jin-chuan, YANG Rui-qing, ZHAO Jie, HUANG Wei.Chemical change of copper species in liquid paraffin[J].Chin J Appl Chem, 2013, 30(1):67-72. http://www.cnki.com.cn/Article/CJFDTOTAL-YYHX201301013.htm [21] LIU L, HUANG W, GAO Z, YIN L.The dehydration of methanol to dimethyl ether over a novel slurry catalyst[J].Energ Source, Part A, 2010, 32(15):1379-1387. doi: 10.1080/15567030903030724 [22] 孙凯, 张小雨, 张琳, 边仲凯, 黄伟, 赵志换.酸碱性硅溶胶对浆状Cu/Zn/Al催化剂性能的影响[J].燃料化学学报, 2015, 43(10):1221-1229. doi: 10.1016/S1872-5813(15)30037-2SUN Kai, ZHANG Xiao-yu, ZHANG Lin, BIAN Zhong-kai, HUANG Wei, ZHAO Zhi-huan.Influence of acid and alkaline silica sol on the performance of Cu/Zn/Al slurry catalysts[J].J Fuel Chem Technol, 2015, 43(10):1221-1229. doi: 10.1016/S1872-5813(15)30037-2 [23] 张智宏, 王玉峰, 张少瑜.用柠檬酸通过固相反应法制备氧化镍[J].机械工程材料, 2010, 34(1):49-51. http://www.cnki.com.cn/Article/CJFDTOTAL-GXGC201001014.htmZHANG Zhi-hong, WANG Yu-feng, ZHANG Shao-yu.Nickel oxide prepared by solid state reaction with citric acid[J].Mater Mech Eng, 2010, 34(1):49-51. http://www.cnki.com.cn/Article/CJFDTOTAL-GXGC201001014.htm [24] 马强, 黄伟, 樊金串, 赵杰, 任杰.完全液相法制备的Cu-Zn-Si-Al浆状催化剂一步法合成二甲醚的失活研究[J].分子催化, 2009, 23(6):499-505. http://www.cnki.com.cn/Article/CJFDTOTAL-FZCH200906003.htmMA Qiang, HUANG Wei, FAN Jin-chuan, ZHAO Jie, REN Jie.Study on the deactivation of Cu-Zn-Si-Al slurry catalyst prepared by complete liquid-phase for one-step dimethyl ether synthesis[J].J Mol Catal, 2009, 23(6):499-505. http://www.cnki.com.cn/Article/CJFDTOTAL-FZCH200906003.htm [25] 樊金串, 吴慧, 黄伟, 谢克昌.表面活性剂对完全液相法制Cu-Zn-Al浆状催化剂结构和性能的影响[J].高等学校化学学报, 2008, 29(5):993-999. http://www.cnki.com.cn/Article/CJFDTOTAL-GDXH200805026.htmFAN Jin-chuan, WU Hui, HUANG Wei, XIE Ke-chang.Effect of surfactants on structure and performance of Cu-Zn-Al catalyst prepared by complete liquid-phase technology[J].Chem J Chin Univ, 2008, 29(5):993-999. http://www.cnki.com.cn/Article/CJFDTOTAL-GDXH200805026.htm [26] 黄伟, 刘勇军, 左志军.Al含量对完全液相法制备Cu-Zn-Al催化剂合成甲醇的影响[J].太原理工大学学报, 2012, 43(4):401-405. http://www.cnki.com.cn/Article/CJFDTOTAL-TYGY201204002.htmHUANG Wei, LIU Yong-jun, ZUO Zhi-jun.Effect of Al content on synthesis of methanol over Cu-Zn-Al catalyst prepared by complete liquid-phase technology[J].J Taiyuan Univ Technol, 2012, 43(4):401-405. http://www.cnki.com.cn/Article/CJFDTOTAL-TYGY201204002.htm [27] RAMOS F S, FARIAS A M, BORGES L E P, MONTEIRO J L, FRAGA M A, SOUSA A, APPEL L G.Role of dehydration catalyst acid properties on one-step DME synthesis over physical mixtures[J].Catal Today, 2005, 101(1):39-44. doi: 10.1016/j.cattod.2004.12.007 [28] YARIPOUR F, SHARIATINIA Z, SAHEBDELFAR S, IRANDOUKHT A.The effects of synthesis operation conditions on the properties of modifiedγ-alumina nanocatalysts in methanol dehydration to dimethyl ether using factorial experimental design[J].Fuel, 2015, 139(1):40-50. [29] LI Z Z, ZUO Z J, HUANG W, XIE K C.Research on Si-Al based catalysts prepared by complete liquid-phase method for DME synthesis in a slurry reactor[J].Appl Surf Sci, 2011, 257(6):2180-2183. doi: 10.1016/j.apsusc.2010.09.069 [30] SUH Y W, MOON S H, RHEE H K.Active sites in Cu/ZnO/ZrO2, catalysts for methanol synthesis from CO/H2[J].Catal Today, 2000, 63(2):447-452. [31] VELU S, SUZUKI K, VIJAYARAJ M, BARMAN S, GOPINATH C S.In situ XPS investigations of Cu1-xNixZnAl mixed metal oxide catalysts used in the oxidative steam reforming of bio-ethanol[J].Appl Catal B:Environ, 2005, 55(4):287-299. doi: 10.1016/j.apcatb.2004.09.007 [32] 李志红, 黄伟, 左志军, 宋雅君, 谢克昌.用XPS研究不同方法制备的CuZnAl一步法二甲醚合成催化剂[J].催化学报, 2009, 30(2):171-177. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA200902018.htmLI Zhi-hong, HUANG Wei, ZUO Zhi-jun, SONG Ya-jun, XIE Ke-chang.XPS study on CuZnAl catalysts prepared by different methods for direct synthesis of dimethyl ether[J].Chin J Catal, 2009, 30(2):171-177. http://www.cnki.com.cn/Article/CJFDTOTAL-CHUA200902018.htm [33] SUN K, LU W, WANG M, XU X.Low-temperature synthesis of DME from CO2/H2 over Pd-modified CuO-ZnO-Al2O3-ZrO2/HZSM-5 catalysts[J].Catal Commun, 2004, 5(7):367-370. doi: 10.1016/j.catcom.2004.03.012 [34] OKAMOTO Y, FUKINO K, IMANAKA T, TERANISHI S.Surface characterization of CuO-ZnO methanol-synthesis catalysts by X-ray photoelectron spectroscopy.1.Precursor and calcined catalysts[J].J Phys Chem, 1983, 87(19):3740-3747. doi: 10.1021/j100242a034 [35] DENISE B, SNEEDEN R P A, BEGUIN B, CHERIFI O.Supported copper catalysts in the synthesis of methanol:N2O-titrations[J].Appl Catal, 1987, 30(2):353-363. doi: 10.1016/S0166-9834(00)84125-1 [36] 樊金串, 黄伟, 吉鹏.完全液相法与溶胶-凝胶法制备Cu-Zn-Al双功能催化剂对浆态床一步法合成二甲醚的催化性能比较[J].高等学校化学学报, 2011, 32(6):1360-1365. http://www.cnki.com.cn/Article/CJFDTOTAL-GDXH201106026.htmFAN Jin-chuan, HUANG Wei, JI Peng.Comparison of catalytic performances between complete liquid-phase's catalyst and Sol-gel's catalyst for slurry synthesis dimethyl ether[J].Chem J Chin Univ, 2011, 32(6):1360-1365. http://www.cnki.com.cn/Article/CJFDTOTAL-GDXH201106026.htm -





 下载:
下载: