-
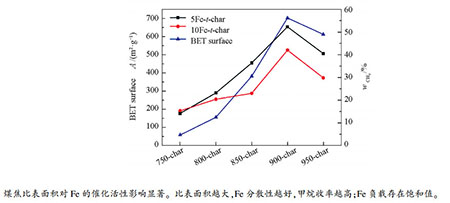
摘要: 在加压固定床反应器上研究了Fe催化剂在不同比表面积煤焦中分散性对催化加氢气化性能的影响,利用XRD、BET、H2-TPR、FT-IR、TEM、拉曼光谱对煤焦及催化剂进行了分析表征。结果表明,煤焦活性位点和石墨化程度并非影响催化气化反应的唯一因素,而催化剂的分散性对反应影响更大。煤焦的比表面积越大,Fe催化剂在煤焦表面的分散更均匀,催化剂活性组分平均晶粒粒径越小,并可以促进煤催化加氢气化中间相产物Fe3C的生成,甲烷收率越高。对于比表面积较高的900-char,在氢气压力为2 MPa,温度为750 ℃,Fe负载量为5%(质量分数)时,催化加氢气化甲烷收率可达53%。在900-char上考察了Fe催化剂负载量对催化加氢气化的影响,甲烷收率呈先增加后降低的趋势,Fe负载量存在饱和点。Abstract: The effect of dispersibility of Fe on coal chars with different specific surface areas on the catalytic hydrogasification performance was studied in a pressurized fixed bed reactor. The chars and catalysts were characterized by XRD, BET, H2-TPR, FT-IR, TEM and Raman spectroscopy. The results show that the active site and graphitization degree of coal char are not the only factors affecting the catalytic gasification reaction, but the dispersion of catalyst has a greater impact on the reaction. The larger the specific surface area of coal char is, the more uniform the Fe catalyst disperses on the surface of coal char and the smaller the average grain size of the active component of catalyst is, which can promote the formation of the catalytically hydrogenated mesophase product Fe3C and methane yield. For 900-char with higher specific surface area, the methane yield can reach 53% when the hydrogen pressure is 2 MPa, the temperature is 750℃ and the Fe loading is 5%. The effect of Fe catalyst loading on the catalytic hydrogasification was investigated with 900-char. It is found that the methane yield increases first and then decreases, and the Fe loading has a saturation point.
-
Key words:
- iron /
- hydrogasification /
- catalytic /
- dispersion /
- saturation point
-
图 1 催化加氢气化加压固定床反应装置示意图
Figure 1 Schematic diagram of pressurized fixed bed reactor for catalytic hydrogasification
1: mass flowmeter; 2: cutoff valve; 3: pressure gauge; 4: electric furnace; 5: ball valve; 6: reactor; 7: K thermocouple; 8: sample; 9: quartz sand; 10: reducing pressure valve; 11: back pressure valve; 12: circulating cooling system; 13: temperature control system; 14: GC; 15: wet air flowmeter
表 1 不同温度预处理煤焦的元素分析
Table 1 Ultimate analysis of char samples at different pretreatment temperatures
Sample Ultimate analysis wdaf/% C H N S O* De-char 93.200 0.450 1.210 0.150 4.990 750-char 94.220 1.334 1.660 0.214 2.572 800-char 93.420 1.282 1.320 0.214 3.764 850-char 91.920 1.684 1.890 0.270 4.506 900-char 91.630 1.326 1.550 0.220 5.027 950-char 92.550 1.334 1.270 0.249 4.597 *: by difference 表 2 不同温度预处理煤焦的孔结构
Table 2 Pore structure parameters of char samples pretreated at different temperatures
Sample BET surface area A/(m2·g-1) Pore volume v/(cm3·g-1) Average pore size d/nm De-char 4.8 0.003 7.75 750-char 58.3 0.042 2.88 800-char 155.7 0.061 1.56 850-char 381.8 0.156 1.63 900-char 701.8 0.302 1.72 950-char 612.1 0.263 1.72 表 3 不同温度预处理煤焦的拉曼分峰拟合面积比
Table 3 Parameters of Raman peak fitting area ratio of coal char pretreated at different temperatures
Sample ID1/IG ID3+D4/IG IG/IAll De-char 1.95 4.23 0.139 750-char 2.96 3.68 0.131 800-char 2.08 5.98 0.120 850-char 1.65 4.93 0.132 900-char 2.23 4.57 0.128 950-char 1.98 4.96 0.126 表 4 不同比表面积煤焦负载5%Fe-1%Ca的XRD表征相关参数
Table 4 XRD characterization parameters of coal chars with different specific surfaces loaded with 5%Fe-1%Ca
Sample Chem form Average crystallite size d/nm dFe dFe3C 5Fe-750-char FeO,Fe2O3, Fe3O4 - - 5Fe-800-char FeO - - 5Fe-850-char Fe, Fe3C 52.94 35.18 5Fe-900-char Fe, Fe3C 43.62 21.05 5Fe-950-char Fe, Fe3C 49.43 26.38 -
[1] 洪冰清, 战书鹏, 王兴军, 王辅臣, 于广锁.不同金属化合物催化呼和浩特煤加氢气化实验研究[J].燃料化学学报, 2012, 40(7):782-789. doi: 10.3969/j.issn.0253-2409.2012.07.003HONG Bing-qing, ZHAN Shu-peng, WANG Xing-jun, WANG Fu-cheng, YU Guang-suo. Experimental study on Hohhot coal hydrogasification catalysed by different metal compounds[J]. J Fuel Chem Technol, 2012, 40(7):782-789. doi: 10.3969/j.issn.0253-2409.2012.07.003 [2] JIANG J, LIU Z, LIU Q. Synergetic catalysis of calcium oxide and iron in hydrogasification of char[J]. Energy Fuels, 2017, 31:198-204. doi: 10.1021/acs.energyfuels.6b02026 [3] OHTSUKA Y, TAMAI Y, TOMITA A. Iron-catalyzed gasification of brown coal at low temperatures[J]. Energy Fuels, 1987, 1(1):32-36. doi: 10.1021/ef00001a006 [4] MATSUMOTO S. Catalyzed hydrogasification of Yallourn char in the presence of supported hydrogenation nickel catalyst[J]. Energy Fuels, 1991, 5(1):60-63. doi: 10.1021/ef00025a009 [5] MATSUMOTO S, SAKAGAMI S. Catalytic gasification activity of iron enhanced by spilt-over hydrogen[J]. Stud Surf Sci Catal, 1993, 77:409-412. doi: 10.1016/S0167-2991(08)63223-1 [6] YAN S, BI J, QU X. The behavior of catalysts in hydrogasification of sub-bituminous coal in pressured fluidized bed[J]. Appl Energy, 2017, 206:401-412. doi: 10.1016/j.apenergy.2017.08.189 [7] HAGA T, NISHIYAMA Y. Promotion of iron-group catalysts by a calcium salt in hydrogasification of carbons at elevated pressures[J]. Ind Eng Chem Res, 1987, 26:1202-1206. doi: 10.1021/ie00066a023 [8] YUAN S, ZHANG N, QU X, BI J, CAO Q, WANG J. Promoted catalysis of calcium on the hydrogasification reactivity of iron-loaded subbituminous coal[J]. Fuel, 2017, 200:153-161. doi: 10.1016/j.fuel.2017.03.066 [9] YUAN S, QU X, ZHANG R, BI J. Effect of calcium additive on product yields in hydrogasification of nickel-loaded Chinese sub-bituminous coal[J]. Fuel, 2015, 147:133-140. doi: 10.1016/j.fuel.2015.01.004 [10] SUZUKI T, MINAMI H, YAMADA T, HOMMA T. Catalytic activities of ion-exchanged nickel and iron in low temperature hydrogasification of raw and modified birch chars[J]. Fuel, 1994, 73(12):1836-1841. doi: 10.1016/0016-2361(94)90208-9 [11] ASAMI K, OHTSUKA Y. Catalytic behavior of iron in the gasification of coal with hydrogen[J]. Stud Surf Sci Catal, 1993, 77:413-416. doi: 10.1016/S0167-2991(08)63224-3 [12] JAWHARI T, ROID A, CASADO J. Raman spectroscopic characterization of some commercially available carbon black materials[J]. Carbon, 1995, 33(11):1561-1565. doi: 10.1016/0008-6223(95)00117-V [13] BEYSSAC O, GOFFÉ B, PETITET J P, FROIGNEUX E, MOREAU M, ROUZAUD J N. On the characterization of disordered and heterogeneous carbonaceous materials by Raman spectroscopy[J]. Spectrochim Acta A, 2003, 59(10):2267-2276. doi: 10.1016/S1386-1425(03)00070-2 [14] CHABALALA V P, WAGNER N, POTGIETER-VERMAAK S. Investigation into the evolution of char structure using Raman spectroscopy in conjunction with coal petrography; Part1[J].Fuel Process Technol, 2011, 92(4):750-756. doi: 10.1016/j.fuproc.2010.09.006 [15] HE X, ZHANG X, JIAO Y, ZHU J S, CHEN X W, LI C Y, LI H S. Complementary analyses of infrared transmission and diffuse reflection spectra of macerals in low-rank coal and application in triboelectrostatic enrichment of active maceral[J]. Fuel, 2017, 192:93-101. doi: 10.1016/j.fuel.2016.12.009 [16] 李娜, 李阳, 班延鹏, 宋银敏, 周华从, 智科端, 何润霞, 滕英跃, 杨柯利, 刘全生.胜利褐煤焦钙催化水蒸气气化反应中活性微结构分析[J].燃料化学学报, 2016, 44(11):1297-1303. doi: 10.3969/j.issn.0253-2409.2016.11.003LI Na, LI Yang, BAN Yan-peng, SONG Yin-min, ZHOU Hua-cong, ZHI Ke-rui, HE Run-xia, TENG Ying-yue, YANG Ke-li, LIU Quan-sheng. Analysis of active microstructure during steam gasification of Shengli char catalyzed by calcium component[J]. J Fuel Chem Technol, 2016, 44(11):1297-303. doi: 10.3969/j.issn.0253-2409.2016.11.003 [17] HASEGAWA Y, OTANI R, YONEZAWA S, TAKASHIM M. Reaction between carbon dioxide and elementary fluorine[J]. J Fluorine Chem, 2008, 128(1):17-28. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ023658796/ [18] LIU L, CAO Y, LIU Q. Kinetics studies and structure characteristics of coal char under pressurized CO2gasification conditions[J]. Fuel, 2015, 146:103-110. doi: 10.1016/j.fuel.2015.01.002 [19] KLOSE W, WÖLKI M. On the intrinsic reaction rate of biomass char gasification with carbon dioxide and steam[J]. Fuel, 2005, 84(7):885-892. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=fd88d51e3f60dbcaf331889c7c65f22b [20] MCKEE D W. Effect of metallic impurities on the gasification of graphite in water vapor and hydrogen[J]. Carbon, 1974, 12(4):453-464. doi: 10.1016/0008-6223(74)90011-6 [21] CHEN Z, WANG F, LI H, YANG Q, WANG L, LI X. Low-temperature selective catalytic reduction of NOx with NH3over Fe-Mn mixed-oxide catalysts containing Fe3Mn3O8 phase[J]. Ind Eng Chem Res, 2012, 51(1):202-212. doi: 10.1021/ie201894c [22] PINEAU A, KANARI N, GABALLAH I. Kinetics of reduction of iron oxides by H2:Part I:Low temperature reduction of hematite[J]. Thermochim Acta, 2006, 447(1):89-100. doi: 10.1016/j.tca.2005.10.004 [23] MATSUMOTO S, JR P L W. char gasification in steam at 1123 K catalyzed by K, Na, Ca and Fe-effect of H2, H2S and COS[J]. Carbon, 1986, 24(3):277-285. doi: 10.1016/0008-6223(86)90228-9 [24] LECEA S M D, ALMELA-ALARCÍN M, LINARES-SOLANO A. Calcium-catalysed carbon gasification in CO2 and steam[J]. Fuel, 1990, 69(1):21-27. doi: 10.1016/0016-2361(90)90253-M [25] 战书鹏, 王兴军, 洪冰清, 于广锁, 王辅臣.褐煤催化加氢气化实验研究[J].燃料化学学报, 2012, 40(1):8-14. doi: 10.3969/j.issn.0253-2409.2012.01.002ZHAN Shu-peng, WANG Xing-jun, HONG Bing-qing, YU Guang-suo, WANG Fu-cheng. Experimental study on catalytic hydrogasification of lignite[J]. J Fuel Chem Technol, 2012, 40(1):8-14. doi: 10.3969/j.issn.0253-2409.2012.01.002 -





 下载:
下载: