Study on the formation of 2-pentanone from ethanol over K-CuZrO2 catalysts
-
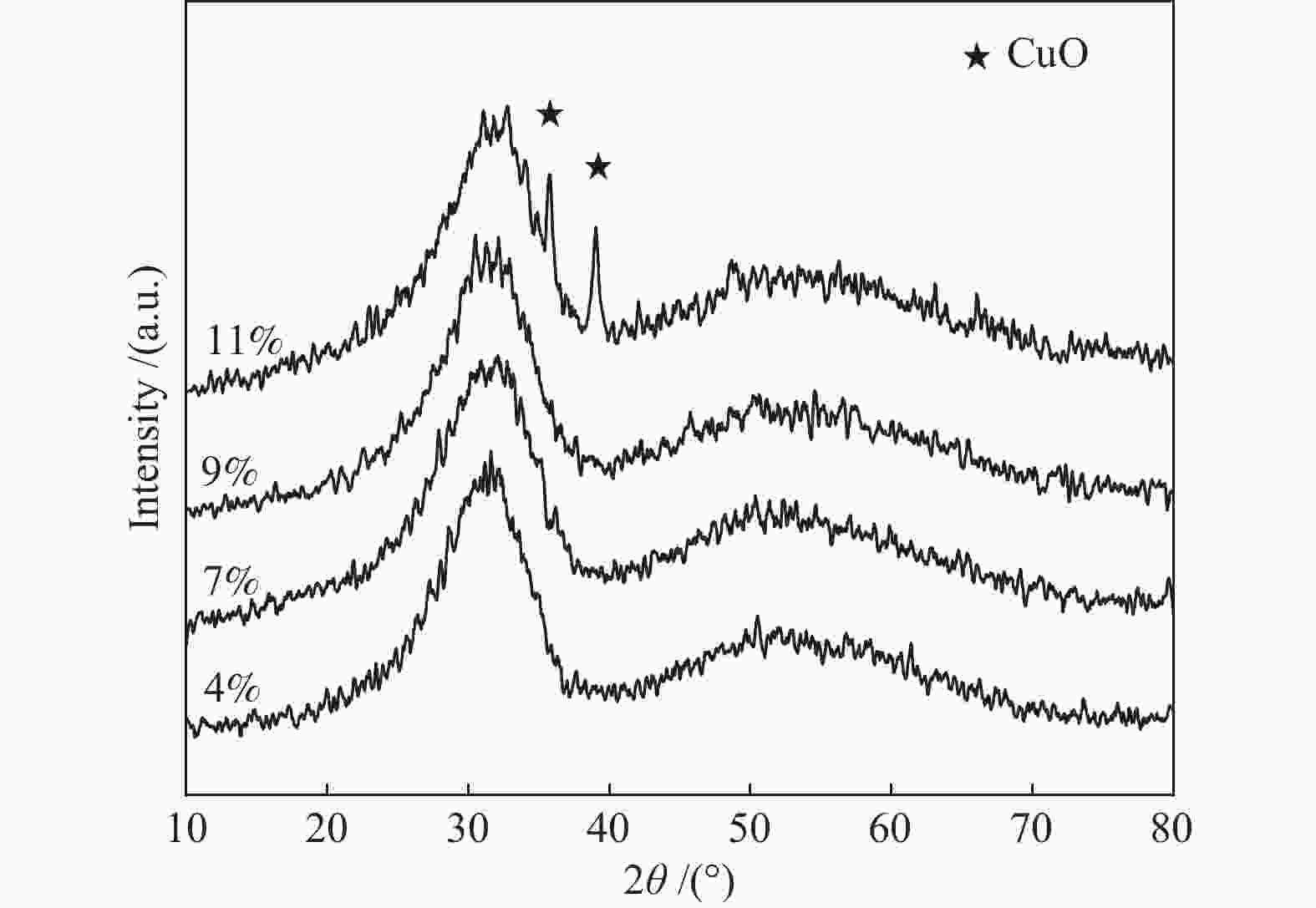
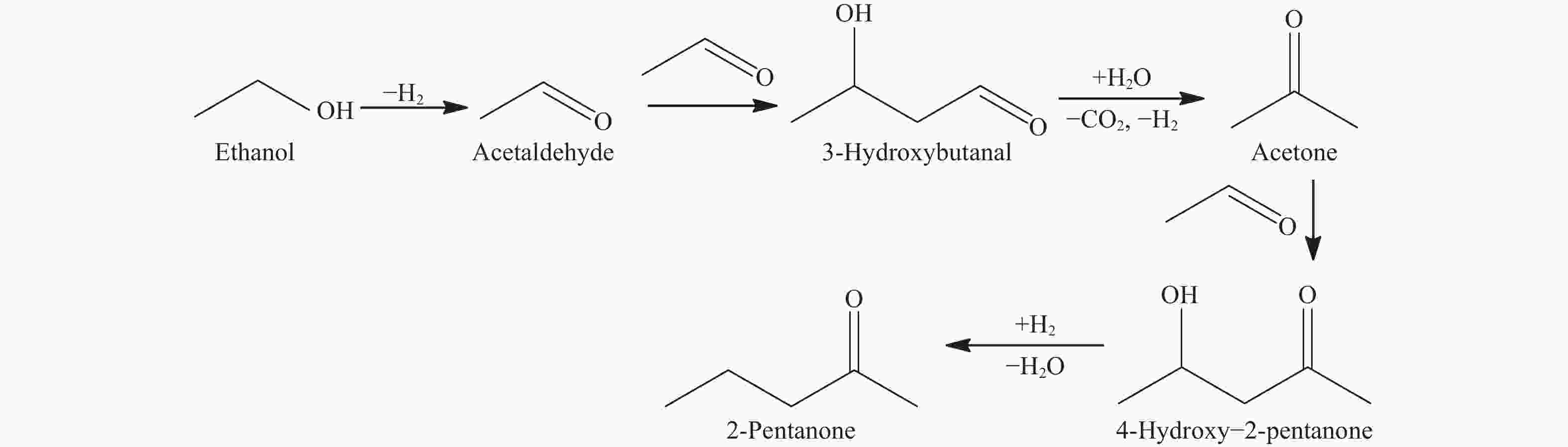
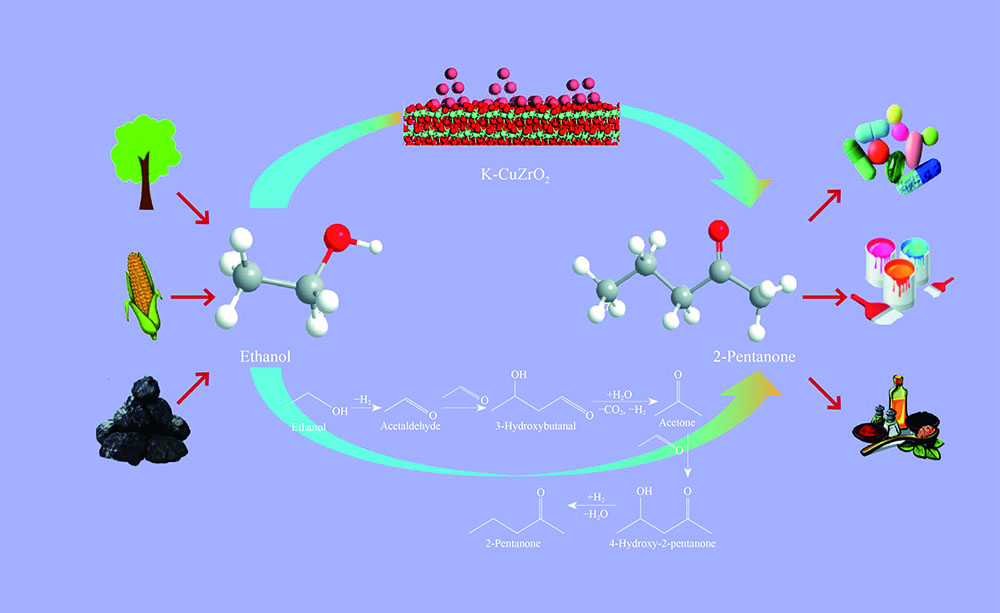
摘要: 制备了不同Cu含量的K-CuZrO2催化剂。以乙醇缩合制备2-戊酮为探针反应,考察了催化剂的催化性能并对反应机理进行了探索;采用BET、XRD、H2-TPR、CO2-TPD、TEM以及XPS等表征技术对催化剂的体相结构、性质进行了研究。结果表明,当Cu含量为9%时,乙醇转化率达到极大值(99.5%),这是由于此时催化剂中各组分分散较好,CuO-ZrO2之间存在较强的相互作用,促进了CuO的还原,使催化剂表面Cu比表面积最大;2-戊酮选择性达到最大值(35.0%),是由于催化剂表面适合缩合反应的中等强度碱性中心碱性最强。通过对反应中间物种分析,推测了K-CuZrO2催化剂上2-戊酮的形成过程:乙醇首先脱氢形成乙醛,之后两分子乙醛经缩合、分解得到丙酮,丙酮进一步与乙醛反应形成目标产物2-戊酮。
-
关键词:
- 铜含量 /
- K-CuZrO2催化剂 /
- 乙醇 /
- 2-戊酮
Abstract: A series of K-CuZrO2 catalysts with different Cu contents were prepared. The catalytic performance and reaction mechanism of 2-pentanone from ethanol condensation were investigated. The structure and properties of the catalysts were studied by N2 sorption, XRD, H2-TPR, CO2-TPD, TEM and XPS. The results showed that when the content of Cu was 9%, the conversion of ethanol reached the maximum (99.5%) due to the strong interaction between CuO and ZrO2 which promoted the reduction of CuO and resulted in the largest specific surface area of Cu on the catalyst surface. The selectivity of 2-pentanone reached the maximum (35.0%) because the strongest basicity related to medium-strength basic sites of the catalyst surface were suitable for the condensation reaction. The formation of 2-pentanone on K-CuZrO2 catalyst was speculated on the basis of the analysis of intermediates: ethanol was dehydrogenated to form acetaldehyde. Then, the acetaldehyde was condensed and decomposed to acetone followed by reaction with acetaldehyde to form 2-pentanone.-
Key words:
- copper content /
- K-CuZrO2 catalyst /
- ethanol /
- 2-pentanone
-
表 1 不同Cu含量K-CuZrO2催化剂比表面积和Cu比表面积
Table 1 BET surface area and surface metallic area of KCuZrO2 catalysts with different Cu loadings
Cu w/
%Average dp/
nmPore volume v/
(cm3·g−1)ABET/
(m2·g−1)SCu/
(m2·g−1)4 4.3 0.2 192 7.2 7 4.4 0.2 208 16.2 9 5.2 0.2 166 19.8 11 4.4 0.2 166 16.8 表 2 不同Cu含量K-CuZrO2催化剂的XPS表征
Table 2 XPS results for K-CuZrO2 catalysts with different Cu loadings
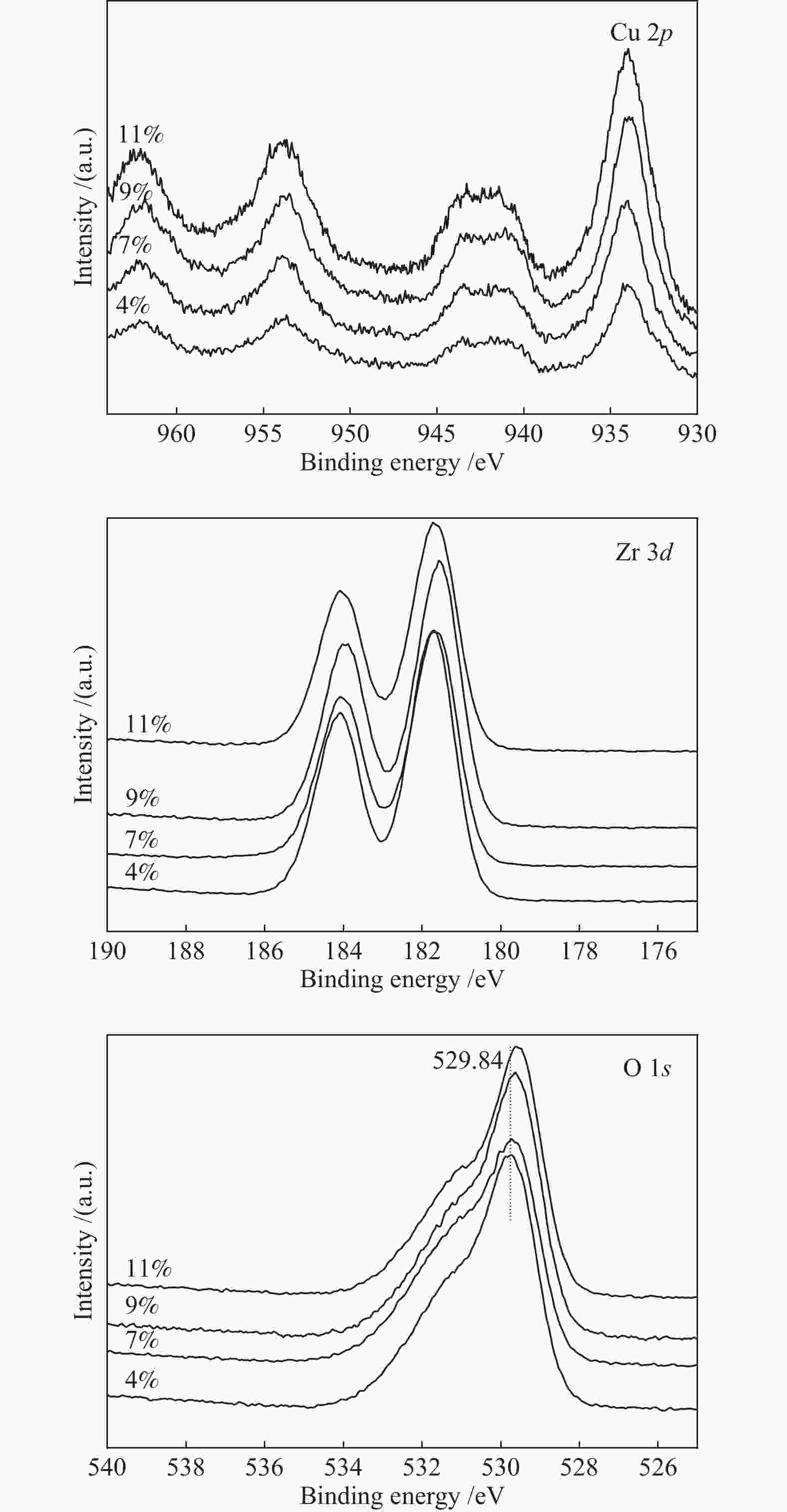
Cu w/% Binding energy E/eV Relative surface concentration of catalysts/% Cu 2p3/2 Zr 3d5/2 O 1s Cu Zr O K 4 933.90 181.73 529.84 2.5 20.6 73.3 3.6 7 933.95 181.67 529.88 4.6 18.9 73.0 3.5 9 934.06 181.58 529.70 5.7 18.9 71.5 3.9 11 933.98 181.67 529.64 7.9 18.0 70.4 3.7 表 3 Cu含量对2-戊酮合成性能的影响
Table 3 Effect of Cu content on the synthesis of 2-pentanone
Samples Conv. x/% Distribution of products/% acetaldehyde acetone 2-pentanone others 4 94.7 0.8 3.6 19.0 76.6 7 98.0 2.9 8.1 27.7 61.3 9 99.5 5.8 7.8 35.0 53.6 11 92.7 3.6 4.4 21.5 68.3 reaction conditions:350 °C,0.1 MPa, carrier gas: N2, GHSV = 2000 h−1, WHSV = 1.8 $ {\rm{m}}{{\rm{L}}_{{{\rm{C}}_2}{{\rm{H}}_5}{\rm{OH}}}} $/(mLcat·h), reaction time:3 h 表 4 乙醇进样量对2-戊酮合成性能的影响
Table 4 Effect of ethanol injection on the synthesis of 2-pentanone
WHSV/
$ ({\rm{m}}{{\rm{L}}_{{{\rm{C}}_2}{{\rm{H}}_5}{\rm{OH}}}} \cdot {\rm{mL}}_{{\rm{cat}}}^{ - 1} \cdot {{\rm{h}}^{ - 1}}) $Conv.
x/%Distribution of products/% acetaldehyde acetone 2-pentanone others 1.2 99.8 3.5 6.5 28.4 76.6 1.8 99.5 3.6 7.8 35.0 53.6 3 91.6 5.4 1.9 12.1 80.6 reaction conditions:350 °C, 0.1 MPa, carrier gas: N2, GHSV = 2000 h−1, 9%Cu-KCuZrO2, reaction time:3 h 表 5 反应温度对2-戊酮合成性能的影响
Table 5 Effect of reaction temperature on the synthesis of 2-pentanone
t/°C Conv. x/% Distribution of products/% acetaldehyde acetone 2-pentanone others 330 90.5 2.5 6.5 28.4 62.6 340 95.5 3.0 8.9 37.5 50.6 350 99.5 3.6 7.8 35.0 53.6 360 99.8 2.8 6.2 30.1 60.9 reaction conditions:9%Cu-KCuZrO2, 0.1 MPa, carrier gas: N2, GHSV = 2000 h−1, WHSV = 1.8 $ {\rm{m}}{{\rm{L}}_{{{\rm{C}}_2}{{\rm{H}}_5}{\rm{OH}}}} $/(mLcat·h), reaction time:3 h -
[1] 陈明明, 刘伟, 陈蒙慈, 曾雪云, 陈明. 3,5-二氯-2-戊酮的合成研究[J]. 精细化工中间体,2015,45(1):36−39.CHEN Ming-ming, LIU Wei, CHEN Meng-ci, ZHEN Xue-yun, CHEN Ming. Synthesis of 3,5-dichloro-2-pentanone[J]. Fine Chem Intermed,2015,45(1):36−39. [2] 任亚宁, 张怡, 门靖. 3,5-二氯-2-戊酮合成方法及在药物制备中的应用[J]. 化工与医药工程,2019,40(2):23−30.REN Ya-ning, ZhANG Yi, MEN Jing. Synthesis method of 3,5-dichloro-2-pentanone and its application in preparation of drugs[J]. Chem Pharm Eng,2019,40(2):23−30. [3] 孙永军, 李硕, 郭春. 5-氯-2-戊酮的合成工艺改进[J]. 精细化工中间体,2015,45(6):45−47.SUN Yong-jun, LI Shuo, GUO Chun. Improvement on the synthesis of 5-chlorine-2-pentanone[J]. Fine Chem Intermed,2015,45(6):45−47. [4] 邓广金. 合成脂肪酮的研究[D]. 北京: 北京化工大学, 2001.Deng Guang-jin. Study on the synthesis of aliphatic ketones[D]. Beijing: Beijing Univ Chem Technol, 2001. [5] WANG Q N, WENG X F, ZHOU B C, LV S P, MIAO S, ZHANG D L, HAN Y, SCOTT S L, SCHÜTH F, LU A H. Direct, selective production of aromatic alcohols from ethanol using a tailored bifunctional cobalt-hydroxyapatite catalyst[J]. ACS Catal,2019,9:7204−7216. doi: 10.1021/acscatal.9b02566 [6] WANG Q N, ZHOU B C, WENG X F, LV S P, SCHÜTH F, LU A H. Hydroxyapatite nanowires rich in [Ca-O-P] sites for ethanol direct coupling showing high C6–12 alcohol yield[J]. Chem Commun,2019,55:10420−10423. doi: 10.1039/C9CC05454E [7] HE D P, DING Y J, CHEN W M, LU Y, LUO H Y. One-step synthesis of 2-pentanone from ethanol over K-Pd/MnOx-ZrO2-ZnO catalyst[J]. J Mol Catal A: Chem,2005,226:89−92. doi: 10.1016/j.molcata.2004.08.002 [8] SUBRAMANIAM S, GUO M F, BATHENA T, GRAY M, ZHANG X, MARTINEZ A, KOVARIK L, GOULAS K A, RAMASAMY K K. Direct catalytic conversion of ethanol to C5+ ketones: Role of PdZn alloy on catalytic activity and stability[J]. Angew Chem Int Ed,2020,59:14550−14557. doi: 10.1002/anie.202005256 [9] LU T, DU Z, LIU J, CHEN C, XU J. Dehydrogenation of primary aliphatic alcohols to aldehydes over Cu-Ni bimetallic catalysts[J]. Chin J Catal,2014,35:1911−1916. doi: 10.1016/S1872-2067(14)60208-4 [10] 闫梦霄, 肖勇山, 石先莹, 葛汉青, 李婷, 宋永红, 刘昭铁, 刘忠文. Cu-SiO2催化环己醇气相脱氢制环己酮的研究[J]. 陕西师范大学学报(自然科学版),2019,47(1):109−116.YAN Meng-xiao, XIAO Yong-shan, SHI Xian-ying, GE Han-qing, LI Ting, SONG Yong-hong, LIU Zhao-tie, LIU Zhong-wen. The gas-phase dehydrogenation of cyclohexanol to cyclohexanone over Cu-SiO2 catalysts[J]. J Shaanxi Normal Univ (Nat Sci Ed),2019,47(1):109−116. [11] 蔺丹丹, 宁艳春, 吴旭, 郭娟娟, 安霞, 谢鲜梅. CuZnAl催化剂中Cu含量对催化环己醇气相脱氢反应性能的影响[J]. 太原理工大学学报,2017,48(1):25−29.LIN Dan-dan, NING Yan-chun, WU Xu, GUO Juan-juan, AN Xia, XIE Xian-mei. Effect of Cu content on the catalytic properties of CuZnAl for gas-phase dehydrogenation of cyclohexanol[J]. J Taiyuan Univ Technol,2017,48(1):25−29. [12] 姜广申, 胡云峰, 蔡俊, 许鹏, 丛亮, 方菲. 仲丁醇脱氢制甲乙酮的Cu-ZnO 催化剂[J]. 化工进展,2013,32(2):352−358.JIANG Guang-shen, HU Yun-feng, CAI Jun, XU Peng, CONG Liang, FANG Fei. Research of Cu-ZnO catalysts for sec-butanol dehydrogenation to methyl ethyl ketone[J]. Chem Ind Eng Progress,2013,32(2):352−358. [13] 吕婷婷. 固体碱催化剂K/ZrO2催化合成羟丁基乙烯基醚的研究[D]. 太原: 山西大学, 2018.LV Ting-ting. Synthesis of hydroxybutyl vinyl ether catalyzed by solid base catalyst K/ZrO2[D]. Taiyuan: Shanxi University, 2018. [14] 何代平, 丁云杰, 尹红梅. 碱金属助剂对MnOx/ZrO2催化合成甲醇及异丁醇反应性能的影响[J]. 催化学报,2003,24(2):111−114. doi: 10.3321/j.issn:0253-9837.2003.02.009HE Dai-ping, DING Yun-jie, YIN Hong-mei, WANG Tao, ZHU He-jun. Effect of alkali promoters on catalytic performance of MnOx/ZrO2 for synthesis of methanol and isobutanol from syngas[J]. Chin J Catal,2003,24(2):111−114. doi: 10.3321/j.issn:0253-9837.2003.02.009 [15] TAN L, YANG G H, YONEYAMA Y, KOU Y L, TAN Y S, VITIDSANTC T, TSUBAKIA N. Iso-butanol direct synthesis from syngas over the alkali metals modified Cr/ZnO catalysts[J]. Appl Catal A: Gen,2015,505:141−149. doi: 10.1016/j.apcata.2015.08.002 [16] TIAN S P, WANG S C, WU Y Q, GAO J W, WANG P, XIE H J, YANG G H, HAN Y Z, TAN Y S. The role of potassium promoter in isobutanol synthesis over Zn-Cr based catalysts[J]. Catal Sci Technol,2016,6:4105−4115. doi: 10.1039/C5CY02030A [17] SATO A G, VOLANTI D P, MEIRA D M, DAMYANOVA S, LONGO E, BUENO J M C. Effect of the ZrO2 phase on the structure and behavior of supported Cu catalysts for ethanol conversion[J]. J Catal,2013,307:1−17. doi: 10.1016/j.jcat.2013.06.022 [18] 谭理, 武应全, 张涛, 解红娟, 陈建刚. 沉淀温度对K-CuLaZrO2催化剂上合成气直接合成异丁醇的影响[J]. 燃料化学学报,2019,47(9):1096−1103.TAN Li, WU Ying-quan, ZHANG Tao, XIE Hong-juan, CHEN Jian-gang. Effect of precipitation temperature on the performance of K-CuLaZrO2 catalyst for isobutanol synthesis from syngas[J]. J Fuel Chem Technol,2019,47(9):1096−1103. [19] WU Y Q, ZHANG J F, ZHANG T, SUN K, WANG L Y, XIE H J, TAN Y S. Effect of potassium on the regulation of C1 intermediates in isobutyl alcohol synthesis from syngas over CuLaZrO2 catalysts[J]. Ind Eng Chem Res,2019,58:9343−9351. doi: 10.1021/acs.iecr.9b01436 [20] HLEIS D, LABAKI M, LAVERSIN H, COURCOT D, ABOUKAIS A. Comparison of alkali-promoted ZrO2 catalysts towards carbon black oxidation[J]. Colloids Surf A,2008,33(2/3):193−200. [21] AGUILA G, VALENZUELA A, GUERRERO S, ARAYA P. WGS activity of a novel Cu-ZrO2 catalyst prepared by a reflux method. Comparison with a conventional impregnation method[J]. Catal Commun,2013,39:82−85. doi: 10.1016/j.catcom.2013.05.007 [22] 吴贵升, 任杰, 孙予罕. 焙烧温度对Cu/ZrO2和Cu-La2O3/ZrO2催化性能的影响[J]. 物理化学学报,1999,15(6):564−567. doi: 10.3866/PKU.WHXB19990616WU Gui-sheng, REN Jie, SUN Yu-han. The effect of calcinations temperature on the performance of Cu/ZrO2 and Cu-La2O3/ZrO2[J]. Acta Phys -Chim Sin,1999,15(6):564−567. doi: 10.3866/PKU.WHXB19990616 [23] 武应全, 解红娟, 冠永利, 谭理, 韩怡卓, 谭猗生. 焙烧温度对K-Cu/Zn/La/ZrO2催化剂异丁醇合成的影响[J]. 燃料化学学报,2013,41(7):868−874. doi: 10.1016/S1872-5813(13)60036-5WU Ying-quan, XIE Hong-juan, KOU Yong-li, Tan Li, HAN Yi-zhuo, TAN Yi-sheng. Effect of calcination temperature on performance of K-Cu/Zn/La/ZrO2 for isobutanol synthesis[J]. J Fuel Chem Technol,2013,41(7):868−874. doi: 10.1016/S1872-5813(13)60036-5 [24] 武应全, 王思晨, 解红娟, 高俊文, 田少鹏, 韩怡卓, 谭猗生. Cu对K-LaZrO2异丁醇合成催化剂的影响[J]. 物理化学学报,2015,31(1):166−172. doi: 10.3866/PKU.WHXB201411241WU Ying-quan, WANG Si-chen, XIE Hong-juan, GAO Jun-wen, TIAN Shao-peng, HAN Yi-zhuo, TAN Yi-sheng. Influence of Cu on the K-LaZrO2 catalyst for isobutanol synthesis[J]. Acta Phys-Chim Sin,2015,31(1):166−172. doi: 10.3866/PKU.WHXB201411241 [25] ORDOMSKY V V, SUSHKEVICH V L, IVANOVA I I. Study of acetaldehyde condensation chemistry over magnesia and zirconia supported on silica[J]. J Mol Catal A: Chem,2010,333:85−93. doi: 10.1016/j.molcata.2010.10.001 [26] PRAŠNIKAR A, PAVLIŠIČ A, RUIZ-ZEPEDA F, KOVAČ J, LIKOZAR B. Mechanisms of copper-based catalyst deactivation during CO2 reduction to methanol[J]. Ind Eng Chem Res,2019,58:13021−13029. doi: 10.1021/acs.iecr.9b01898 -





 下载:
下载: