Changes in pyrolysis characteristics of agricultural residues before and after water washing
-
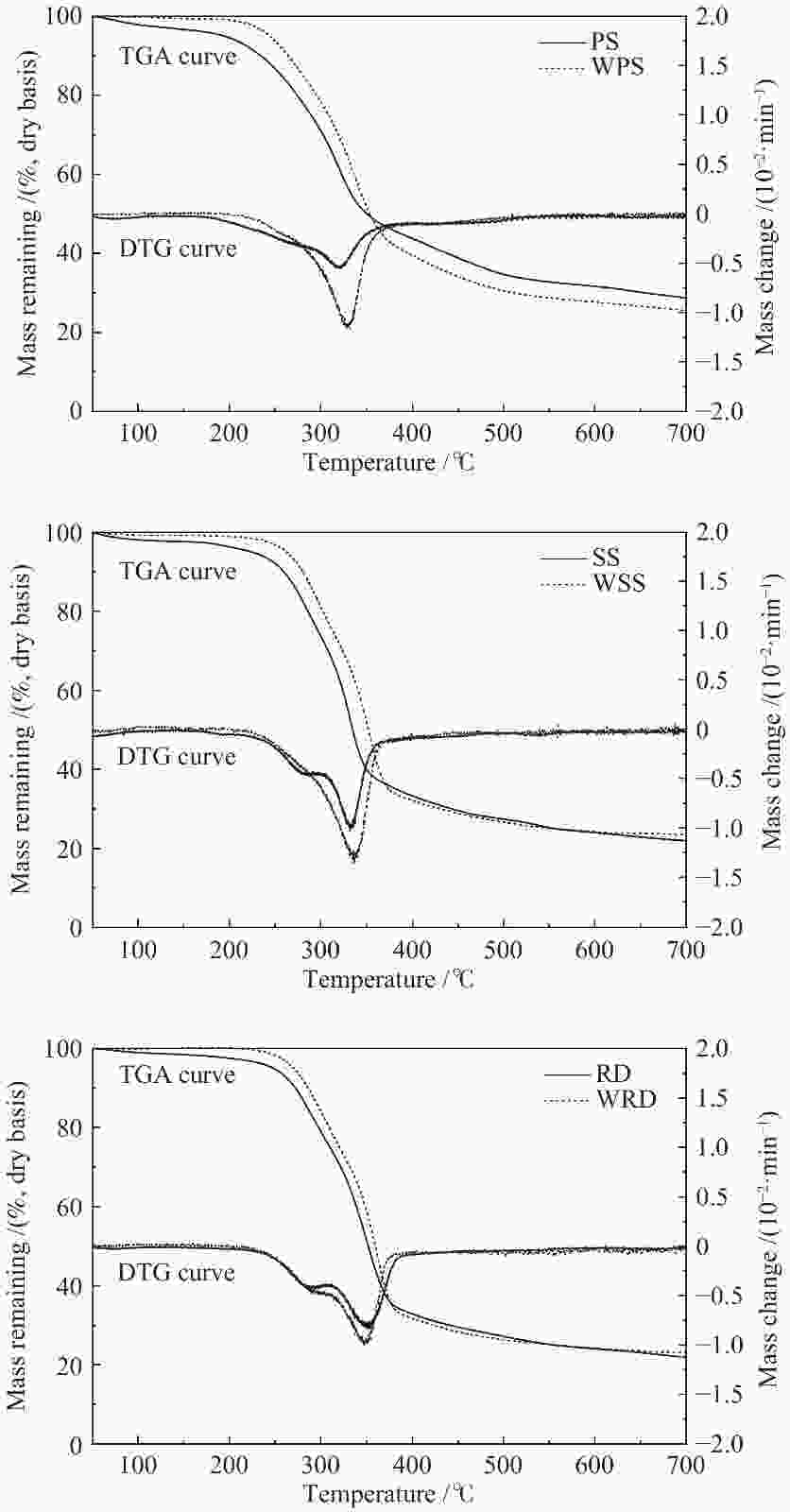
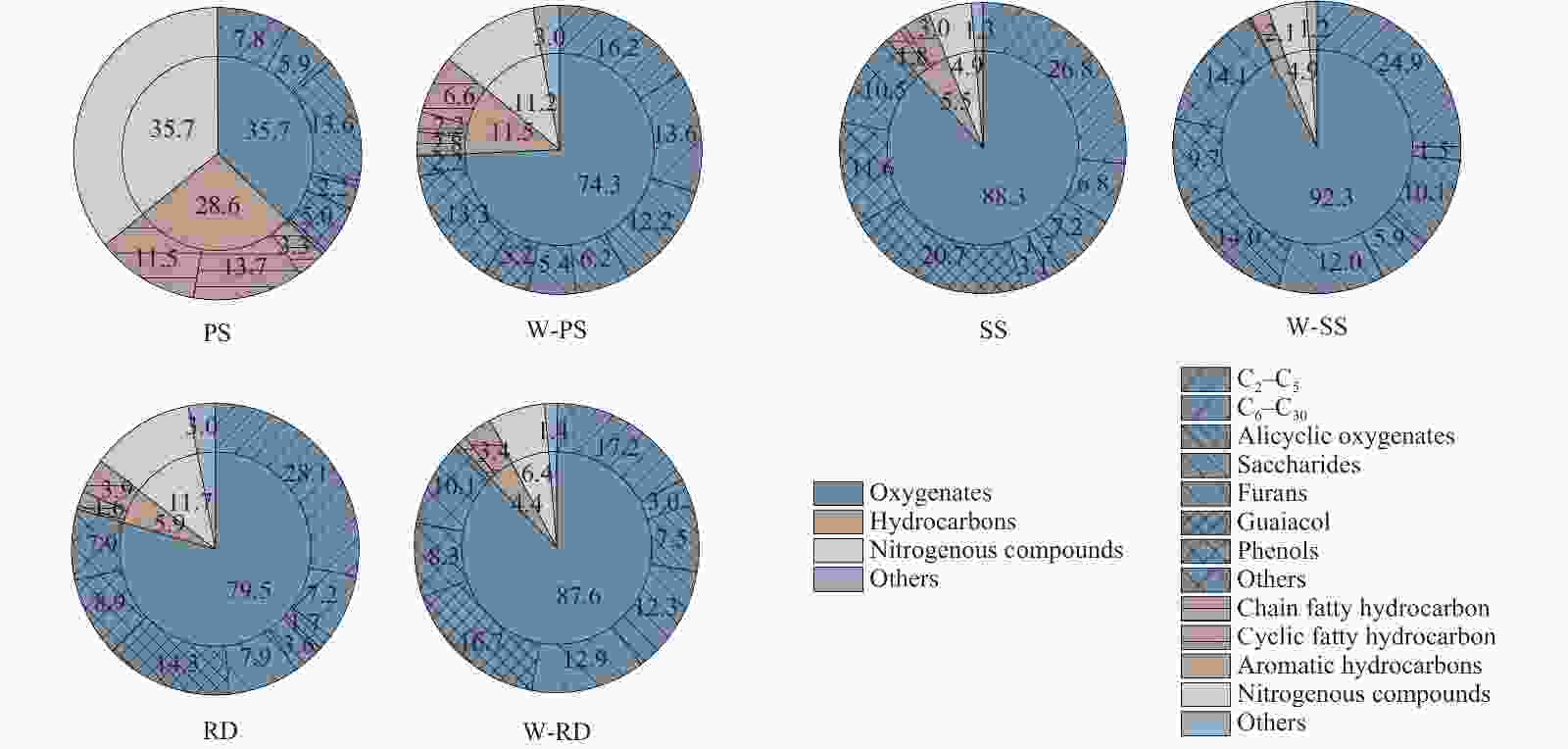
摘要: 首先对花生秸秆、高粱秸秆和芦苇三种农业废弃物进行了水洗预处理,再利用固定床反应器进行了热解实验,借此考察水洗前后碱金属和碱土金属(AAEMs)和纤维组成的变化及其对农业废弃物热解特性的影响。研究发现,水洗可脱除52.7%−92.6%的钾和一半左右的中性溶解物(NDS)。AAEMs和NDS的脱除对热解农业废弃物热解产生综合影响,含AAEMs和NDS较多的花生秸秆影响尤为明显。AAEMs的脱除遏制脱羧基、脱羰基、脱氢及挥发分二次缩聚反应,而NDS的脱除直接影响气体和液体的产率和组成。对于所有三种农业废弃物,水洗后均有利于提高生物油和生物炭产率,但气体产率降低;水洗后生物油中含氧化合物增多,其中,糖类和呋喃类增幅最大,而烃类、含氮化合物减少。水洗后,花生秸秆的CO2、CO和CH4产率均明显降低,而生物油中长链脂肪酸的占比增大。Abstract: Three agricultural residues (peanut straw, sorghum stalk and reed) were first pretreated by water washing, with which the pyrolysis experiments were carried out in a fixed bed reactor to investigate how the pyrolysis characteristics are changed by removing the alkali earth metals (AAEMs) and a part of fibrous components by water washing. The result shows that the water washing removes 52.7%–92.6% potassium and approximately half of neutral detergent solute (NDS) from three agricultural residues. The removal of AAEMs and NDS has a complex influence on the pyrolysis of agricultural residues, especially for peanut straw due to its higher contents of both AAEMs and NDS. However, the removal of AAEMs has an inhibitory effect on the reactions such as decarboxylation, decarbonylation, dehydrogenation and polycondensation, and the elution of NDS directly affects the yields and composition of gas and liquid products. For all three agricultural residues, overall, the water washing promotes the production of bio-oil and bio-char, but it lowers the gas yield. The bio-oils generated from the water-washed agricultural residues have higher proportions of oxygenates, especially sugars and furans, with lower proportions of hydrocarbons and nitrogenous compounds. Besides, after washing, the yields of CO2, CO and CH4 with peanut straw decrease significantly, while the proportion of long-chain fatty acids in bio oil increases.
-
Key words:
- agricultural residues /
- pyrolysis /
- water washing /
- inorganic matter /
- fibrous components
-
表 1 水洗前后农业废弃物的工业分析和元素分析
Table 1 Proximate and ultimate analyses of the agricultural residues before and after water washing
Residues Proximate analysis (%, dry basis) Ultimate analysis (%, dry basis) volatile ash fixed carbona C H N S Oa PS 66.39 17.04 16.57 46.51 6.79 2.77 0.06 43.87 W-PS 72.87 15.50 11.63 49.46 6.92 2.34 0.00 41.28 SS 72.20 9.12 18.68 48.10 6.79 0.59 0.00 44.52 W-SS 76.77 7.06 16.17 48.10 6.38 0.54 0.00 44.98 RD 73.03 7.97 19.00 47.64 6.63 0.83 0.00 44.90 W-RD 77.51 6.54 15.95 48.94 6.44 0.66 0.00 43.96 a: by difference 表 2 水洗前后农业废弃物中的无机矿物质含量
Table 2 Contents of AAEMs in agricultural residues before and after water washing
Residues AAEMs (%, dry basis) Na K Ca Mg PS 0.08 1.79 1.51 1.05 W-PS 0.03 0.70 1.60 0.81 SS 0.03 1.10 0.34 0.19 W-SS 0.02 0.63 0.32 0.16 RD 0.02 0.27 0.23 0.06 W-RD 0.01 0.02 0.16 0.02 表 3 水洗前后农业废弃物的纤维组成
Table 3 Contents of fibrous components in agricultural residues before and after water washing
Residues Fibrous components (%, dry basisa) NDS hemicellulose cellulose lignin silicate PS 47.4 14.3 26.8 7.6 3.9 W-PS 29.4
(21.0)21.3
(15.2)34.7
(24.8)11.1
(7.9)3.5
(2.4)SS 16.8 29.9 23.6 24.8 4.9 W-SS 9.5
(7.9)30.7
(25.5)28.1
(23.4)26.7
(22.2)5.0
(4.2)RD 15.6 24.3 42.1 12.5 5.5 W-RD 8.9
(8.0)27.6
(24.7)44.5
(39.9)13.9
(12.4)5.1
(4.6)a: data in parenthesis are based on the mass of dried raw biomass 表 4 水洗前后农业废弃物热解总产物分布及各气体产率
Table 4 Distributions of overall products and yields of individual gases produced by pyrolysis of the three agricultural residues before and after water washing (%, dry biomass basis)
Products PS W-PS SS W-SS RD W-RD Overall products gas 22.9 17.1 17.9 14.8 15.9 12.3 water 16.2 17.5 24.2 26.1 21.6 23.9 bio-oil 24.3 29.5 27.6 32.0 31.5 37.2 biochar 35.5 34.7 29.3 26.3 29.3 25.3 total 98.9 98.8 99.0 99.2 98.3 98.7 Gases CO2 14.50 10.92 11.15 8.79 9.19 6.99 CO 6.22 4.43 5.15 4.58 4.98 3.94 H2 0.46 0.41 0.33 0.32 0.31 0.24 CH4 1.12 0.75 0.92 0.80 1.08 0.93 C2−C3 0.64 0.58 0.41 0.31 0.38 0.26 表 5 水洗前后农业废弃物热解生物油中部分主要化合物的GC-MS分析相对峰面积(%)
Table 5 GC-MS relative peak areas (RPAs) of some main compounds in pyrolysis bio-oils of the three agricultural residues before and after washing (%)
Group/Compound Molecular formula PS W-PS SS W-SS RD W-RD Fatty acids Hexadecenoic acid C16H30O2 − 1.8 − − − − Hexadecanoic acid C16H32O2 − 6.7 1.0 − − 0.5 Methyl hexadecanoate C17H34O2 1.1 0.4 0.1 − − − Oleic acid C18H34O2 − 0.9 1.2 − − − Octadecanoic acid C18H36O2 − 0.4 0.4 − − − Sugars 1,4,3,6-dianhydro-α-d-glucopyranose C6H8O4 0.7 2.8 1.3 1.3 0.8 1.1 1,6-anhydro-β-d-glucopyranos C6H10O5 − − 0.4 4.7 1.2 5.1 D-allose C6H12O6 − 3.3 0.1 − 1.2 6.1 Cyclic hydrocarbons 1,3,5-cycloheptatriene C7H8 3.1 − − − 0.5 − 1,3,5,7-cyclooctatetraene C8H8 5.0 − − − 0.9 − Trimethylbicyclo heptane C10H18 1.4 2.0 − − − − Ethylcyclododecane C14H28 0.9 − 0.1 − − − Nitrogenous compounds Dihydro-trimethyl- oxazole C6H11NO 13.9 − − − − − 1-pyrrolidinylacetic acid C6H11NO2 11.8 − − − − − (Z)-9-octadecenoamide C18H35NO − 0.9 0.4 0.7 − 0.8 (Z)-13-docosenamide C22H43NO − 0.9 0.4 0.7 − 0.7 表 6 水洗前后农业废弃物热解生物油中部分化合物的产率
Table 6 Yields of some main compounds in the bio-oils produced from the three agricultural residues before and after washing (%, dry biomass basis)
Compound PS W-PS SS W-SS RD W-RD Acetic acid 1.10 1.77 4.07 3.86 4.19 3.28 Hexadecanoic acid 0.00 0.35 0.02 0.00 0.00 0.01 Octadecanoic acid 0.00 0.07 0.04 0.00 0.00 0.00 Oleic acid 0.00 0.15 0.47 0.00 0.00 0.00 Hydroxyacetone 0.13 0.38 0.60 0.55 0.62 0.45 Furfural 0.11 0.29 0.52 1.36 0.83 0.79 2-furanmethanol 0.08 0.34 0.33 0.93 0.41 0.78 Guaiacol 0.00 0.10 0.22 0.26 0.16 0.31 Phenol 0.10 0.20 0.29 0.34 0.13 0.14 Cresols 0.10 0.35 0.21 0.31 0.13 0.30 Xylenol 0.07 0.14 0.16 0.29 0.14 0.14 Benzene 0.09 0.06 0.00 0.00 0.00 0.00 Toluene 0.13 0.13 0.10 0.04 0.07 0.24 Xylene 0.00 0.00 0.02 0.00 0.00 0.00 -
[1] LORENZETTI C, CONTI R, FABBRI D, YANIK J. A comparative study on the catalytic effect of H-ZS M5 on upgrading of pyrolysis vapors derived from lignocellulosic and proteinaceous biomass[J]. Fuel,2016,166:446−452. doi: 10.1016/j.fuel.2015.10.051 [2] ARNI S A. Comparison of slow and fast pyrolysis for converting biomass into fuel[J]. Renewable Energy,2017,124:197−201. [3] TAG, TOPTAS A, DUMAN, GOZDE. Effects of feedstock type and pyrolysis temperature on potential applications of biochar[J]. J Anal Appl Pyrolysis,2016,120:200−206. doi: 10.1016/j.jaap.2016.05.006 [4] ZHANG X, LEI H, ZHU L, QIAN M, ZHU X, WU J, CHEN S. Enhancement of jet fuel range alkanes from co-feeding of lignocellulosic biomass with plastics via tandem catalytic conversions[J]. Appl Energy,2016,173:418−430. doi: 10.1016/j.apenergy.2016.04.071 [5] LIU S, XIE Q, ZHANG B, CHENG Y, RUAN R. Fast microwave-assisted catalytic co-pyrolysis of corn stover and scum for bio-oil production with CaO and HZSM-5 as the catalyst[J]. Bioresour Technol,2016,204:164−170. doi: 10.1016/j.biortech.2015.12.085 [6] KAN T, STREZOV V, EVANS T J. Lignocellulosic biomass pyrolysis: A review of product properties and effects of pyrolysis parameters[J]. Renewable Sustainable Energy Rev,2016,57(28):1126−1140. [7] MEHMOOD M, YE G, LUO H, LIU C, MALIK S, AFZAL I. Pyrolysis and kinetic analyses of Camel grass (Cymbopogon schoenanthus) for bioenergy[J]. Bioresour Technol,2017,228:18−24. doi: 10.1016/j.biortech.2016.12.096 [8] BISWAS B, PANDEY N, BISHT Y, SINGH R, KUMAR J, BHASKAR T. Pyrolysis of agricultural biomass residues: Comparative study of corn cob, wheat straw, rice straw and rice husk[J]. Bioresour Technol,2017,237:57−63. [9] 侯宝鑫, 张守玉, 吴巧美. 生物质热解制备木醋液及其性质研究[J]. 燃料化学学报,2015,43(12):1439−1445.HOU Bao-xin, ZHANG Shou-yu, WU Qiao-mei. Wood vinegar and its properties from pyrolysis of biomass[J]. J Fuel Chem Technol,2015,43(12):1439−1445. [10] 郑楠, 史纪龙, 王杰. 生物质铁盐催化加氢热解产生生物油与气态烃的研究[J]. 燃料化学学报,2020,48(4):414−423.ZHENG Nan, SHI Ji-long, WANG Jie. Iron salts-catalyzed biomass hydropyrolysis for production of bio-oil and gaseous hydrocarbons[J]. J Fuel Chem Technol,2020,48(4):414−423. [11] 王贤华, 陈汉平, 王静, 辛芬, 杨海平. 无机矿物质盐对生物质热解特性的影响[J]. 燃料化学学报,2008,36(6):679−683. doi: 10.3969/j.issn.0253-2409.2008.06.007WANG Xian-hua, CHEN Han-ping, WANG Jing, XIN Fen, YANG Hai-ping. Influences of mineral matters on biomass pyrolysis characteristics[J]. J Fuel Chem Technol,2008,36(6):679−683. doi: 10.3969/j.issn.0253-2409.2008.06.007 [12] CEN K, ZHANG J, MA Z, CHEN D, ZHOU J, MA H. Investigation of the relevance between biomass pyrolysis polygeneration and washing pretreatment under different severities: Water, dilute acid solution and aqueous phase bio-oil[J]. Bioresour Technol,2019,278:26−33. doi: 10.1016/j.biortech.2019.01.048 [13] CHANDLER D S, RESENDE F L P. Effects of warm water washing on the fast pyrolysis of & IT; Arundo Donax & IT[J]. Biomass Bioenergy,2018,2018:113, 65−74. [14] CAO B, WANG S, HU Y, ABOMOHRA E F, QIAN L, HE Z. Effect of washing with diluted acids on Enteromorpha clathrata pyrolysis products: Towards enhanced bio-oil from seaweeds[J]. Renewable Energy,2019,138:29−38. doi: 10.1016/j.renene.2019.01.084 [15] CHEN D Y, WANG Y, LIU Y X, CEN K H, XIAO B. Comparative study on the pyrolysis behaviors of rice straw under different washing pretreatments of water, acid solution, and aqueous phase bio-oil by using TG-FTIR and Py-GC/MS[J]. Fuel,2019,252:1−9. doi: 10.1016/j.fuel.2019.04.086 [16] WANG C Y, ZHENG N, WAN S Q, WANG J. Assessment of the modes of occurrence of trace elements in agricultural crop residues and their enrichments and bioavailability in bio-chars[J]. Biomass Convers Biorefin, 2020, DOI: 10.1007/s13399-019-00597-w. [17] MOURANT D, WANG Z, HE M, XIAO S M, GARCIA P M, LING K, LI C Z. Mallee wood fast pyrolysis, Effects of alkali and alkaline earth metallic species on the yield and composition of bio-oil[J]. Fuel,2011,90(9):2915−2922. [18] NOWAKOWSKI D J, JONES J M, BRYDSON R, ROSS A B. Potassium catalysis in the pyrolysis behaviour of short rotation willow coppice[J]. Fuel,2007,86(15):2389−2402. doi: 10.1016/j.fuel.2007.01.026 [19] RAVEENDRAN K, GANESH A, KHILAR K C. Influence of mineral matter on biomass pyrolysis characteristics[J]. Fuel,1995,74(12):1812−1822. doi: 10.1016/0016-2361(95)80013-8 [20] GUREVICH M L I, BONELLI P R, CUKIERMAN A L. Effect of acid pretreatment and process temperature on characteristics and yields of pyrolysis products of peanut shells[J]. Renewable Energy,2017,114:697−707. doi: 10.1016/j.renene.2017.07.065 [21] 王树荣, 廖艳芳, 文丽华. 钾盐催化纤维素快速热裂解机理研究[J]. 燃料化学学报,2004,32(6):694−698. doi: 10.3969/j.issn.0253-2409.2004.06.011WANG Shu-rong, LIAO Yan-fang, WEN Li-hua. Catalysis mechanism of potassium salt during rapid pyrolysis of cellulose[J]. J Fuel Chem Technol,2004,32(6):694−698. doi: 10.3969/j.issn.0253-2409.2004.06.011 [22] 廖艳芳, 王树荣, 骆仲泱. 氯化钙催化纤维素热裂解动力学研究[J]. 燃料化学学报,2005,33(6):692−697. doi: 10.3969/j.issn.0253-2409.2005.06.010LIAO Yan-fang, Wang Shu-rong, LUO Zhong-yang. Kinetics analysis of cellulose pyrolysis catalyzed by calcium chloride[J]. J Fuel Chem Technol,2005,33(6):692−697. doi: 10.3969/j.issn.0253-2409.2005.06.010 [23] WAN S Q, ZHENG N, ZHANG J, WANG J. Role of neutral extractives and inherent active minerals in pyrolysis of agricultural crop residues and bio-oil formations[J]. Biomass Bioenergy,2019,122:53−62. doi: 10.1016/j.biombioe.2019.01.010 [24] SHI L, YU S, WANG F C, WANG J. Pyrolytic characteristics of rice straw and its constituents catalyzed by internal alkali and alkali earth metals[J]. Fuel,2012,96:586−594. doi: 10.1016/j.fuel.2012.01.013 [25] VAMVUKA D, TROULINOS S, KASTANAKI E. The effect of mineral matter on the physical and chemical activation of low rank coal and biomass materials[J]. Fuel,2006,85(12/13):1763−1771. [26] MOHAN D, CHARLES U P J, STEELE P H. Pyrolysis of wood/biomass for bio-oil: A critical review[J]. Energy Fuels,2006,20(3):848−889. doi: 10.1021/ef0502397 [27] MELZER M, BLIN J, BENSAKHRIA A, VALETTE J, BROUST F. Pyrolysis of extractive rich agroindustrial residues[J]. J Anal Appl Pyrolysis,2013,104:448−460. doi: 10.1016/j.jaap.2013.05.027 [28] YU J, PATERSON N, BLAMEY J, MILLAN M. Cellulose, xylan and lignin interactions during pyrolysis of lignocellulosic biomass[J]. Fuel,2017,191:140−149. doi: 10.1016/j.fuel.2016.11.057 [29] SHI X H, WANG J. A comparative investigation into the formation behaviors of char, liquids and gases during pyrolysis of pinewood and lignocellulosic components[J]. Bioresour Technol,2014,170:262−269. doi: 10.1016/j.biortech.2014.07.110 [30] PASANGULAPATI V, RAMACHANDRIYA K D, KUMAR A, WILKINS M R, JONES C L, HUHNKE R L. Effects of cellulose, hemicellulose and lignin on thermochemical conversion characteristics of the selected biomass[J]. Bioresour Technol,2012,114:663−669. doi: 10.1016/j.biortech.2012.03.036 [31] FAHMI R, BRIDGWATER A V, DARVELL L I, JONES J M, YATES N, THAIN S, DONNISON I S. The effect of alkali metals on combustion and pyrolysis of Lolium and Festuca grasses, switchgrass and willow[J]. Fuel,2007,86(10/11):1560−1569. [32] DI BLASI C, GALGANO A, BRANCA C. Influences of the chemical state of alkaline compounds and the nature of alkali metal on wood pyrolysis[J]. Ind Eng Chem Res,2009,48(7):3359−3369. doi: 10.1021/ie801468y [33] STEFANIDIS S D, KALOGIANNIS K G, ILIOPOULOU E F, MICHAILOF C M, PIAVACHI P A, LAPPAS A A. A study of lignocellulosic biomass pyrolysis via the pyrolysis of cellulose, hemicellulose and lignin[J]. J Anal Appl Pyrolysis,2014,105:143−150. doi: 10.1016/j.jaap.2013.10.013 [34] WANG S, GUO X, WANG K, LUO Z. Influence of the interaction of components on the pyrolysis behavior of biomass[J]. J Anal Appl Pyrolysis,2011,91(1):183−189. doi: 10.1016/j.jaap.2011.02.006 [35] KYOTANI T, HAYASHI S, TOMITA A. Study of calcium catalysis on carbon gasification with molecular oxygen-18[J]. Energy Fuels,1991,5(5):565−569. [36] YANG H, YAN R, CHEN H, DONG H L, ZHENG C. Characteristics of hemicellulose, cellulose and lignin pyrolysis[J]. Fuel,2007,86(12/13):1781−1788. doi: 10.1016/j.fuel.2006.12.013 [37] WANG Z Y, CAO J Q, WANG J. Pyrolytic characteristics of pine wood in a slowly heating and gas sweeping fixed-bed reactor[J]. J Anal Appl Pyrolysis,2009,84(2):179−184. doi: 10.1016/j.jaap.2009.02.001 [38] YILDIZ, GÜRAY, RONSSE F, VENDERBOSCH R. Effect of biomass ash in catalytic fast pyrolysis of pine wood[J]. Appl Catal B: Environ,2015,168:203−211. [39] 李帅丹, 陈雪莉, 刘爱彬. 固定床中纤维素热解及其焦油裂解机理研究[J]. 燃料化学学报,2014,42(4):414−419.LI Shuai-dan, CHEN Xue-li, LIU Ai-bin. Mechanism of cellulose pyrolysis and tar decomposition in a fixed bed reactor[J]. J Fuel Chem Technol,2014,42(4):414−419. [40] 黄金保, 吴隆琴, 童红. 半纤维素模型化合物热解机理的理论研究[J]. 燃料化学学报,2016,44(8):911−920. doi: 10.3969/j.issn.0253-2409.2016.08.003HUANG Jin-bao, WU Long-qin, TONG Hong. Theoretical study on thermal degradation mechanism of hemicellulose model compound[J]. J Fuel Chem Technol,2016,44(8):911−920. doi: 10.3969/j.issn.0253-2409.2016.08.003 [41] EOM I Y, KIM J Y, KIM T S, LEE S M, CHOI D, CHOI I G, CHOI J W. Effect of essential inorganic metals on primary thermal degradation of lignocellulosic biomass[J]. Bioresour Technol,2012,104:687−694. doi: 10.1016/j.biortech.2011.10.035 [42] 黄金保, 刘朝, 魏顺安, 黄晓露, 李豪杰. 纤维素热解形成左旋葡聚糖机理的理论研究[J]. 燃料化学学报,2011,39(8):590−594. doi: 10.3969/j.issn.0253-2409.2011.08.006HUANG Jin-bao, LIU Chao, WEI Shun-an, HUANG Xiao-lu, LI Hao-jie. A theoretical study on the mechanism of levoglucosan formation in cellulose pyrolysis[J]. J Fuel Chem Technol,2011,39(8):590−594. doi: 10.3969/j.issn.0253-2409.2011.08.006 [43] PATWARDHAN P R, SATRIO J A, BROWN R C, SHANKS B H. Product distribution from fast pyrolysis of glucose-based carbohydrates[J]. J Anal Appl Pyrolysis,2009,86(2):323−330. doi: 10.1016/j.jaap.2009.08.007 [44] BROWN A L, HAMES B R, DAILY J W, DAYTON D C. Chemical analysis of solids and pyrolytic vapors from wildland trees[J]. Energy Fuels,2003,17(4):1022−1027. doi: 10.1021/ef020229v -





 下载:
下载: