Low-temperature oxidation of methanol to dimethoxymethane over Mo-Sn catalyst
-
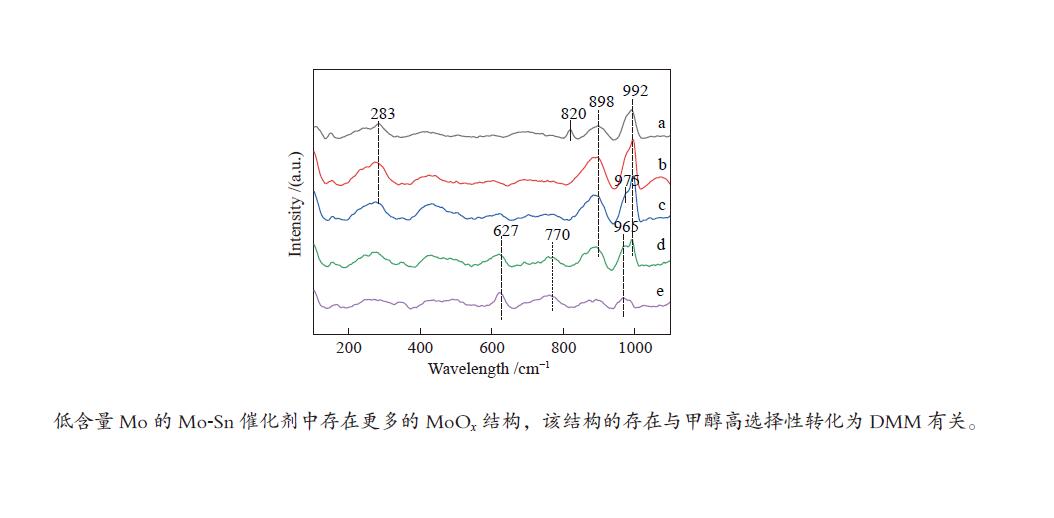
摘要: 采用水热合成法制备了甲醇合成甲缩醛的新型Mo-Sn催化剂。该催化剂可以在低Mo含量时实现甲醇低温氧化高选择性制取甲缩醛。通过考察Mo含量对催化剂结构及甲醇低温氧化制甲缩醛性能的影响,发现Mo1Sn10催化剂在甲醇氧化中表现出了较好的催化性能,在140 ℃、常压反应条件下,甲醇转化率为14.2%,甲缩醛选择性达到了88.9%,并且反应过程中无COx生成。采用XRD、Raman、FT-IR、XPS、NH3-TPD及H2-TPR等表征手段对催化剂进行深入研究。结果表明,不同Mo含量的催化剂结构性能存在着明显的差别,较低含量Mo的存在更有利于Mo5+及MoOx的生成,而由此引起的酸性及氧化还原性的变化是催化剂具有良好性能的重要原因。Abstract: A new Mo-Sn catalyst prepared by hydrothermal method was used for the synthesis of dimethoxymethane (DMM) from methanol oxidation. The catalyst with low Mo content can achieve low-temperature oxidation of methanol to DMM with high selectivity. The influence of Mo content on the structure and the catalytic performance of the catalyst was investigated. It was found that Mo1Sn10 catalyst showed the best catalytic performance under the conditions of 140 °C and atmospheric pressure, the methanol conversion was 14.2%, and the selectivity of DMM reached 88.9% without the formation of COx during the reaction process. The catalysts were characterized by XRD, Raman, FT-IR, XPS, NH3-TPD and H2-TPR. The results showed that the catalysts with different Mo content had obvious differences in structure and performance. Lower Mo content was more conducive to the formation of Mo5+ and MoOx, and the resulting changes in acidity and redox properties were the important reasons for the excellent performance of the catalysts.
-
Key words:
- methanol /
- low temperature oxidation /
- dimethoxymethane /
- Mo-Sn catalyst /
- Mo5+ species
-
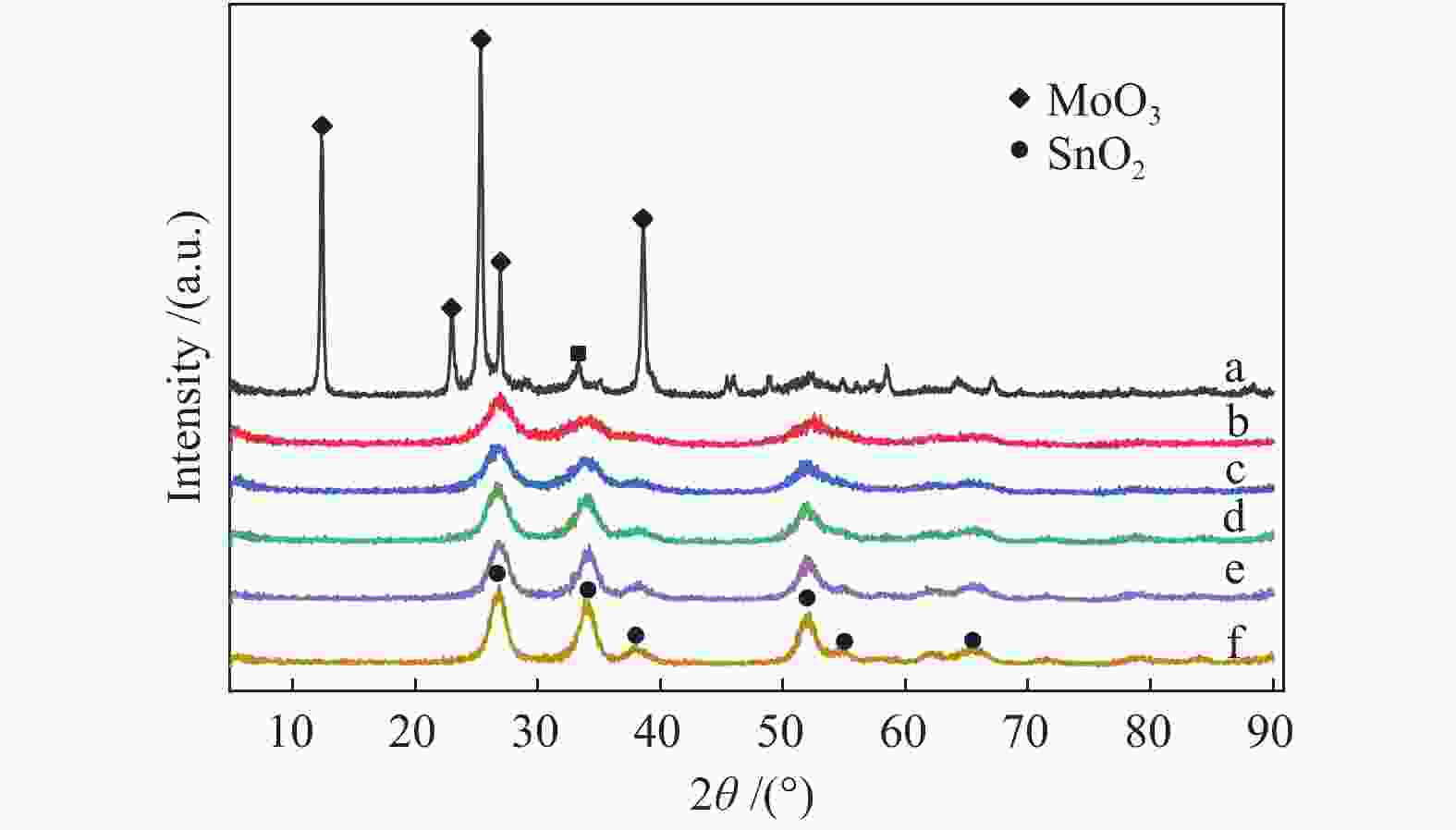
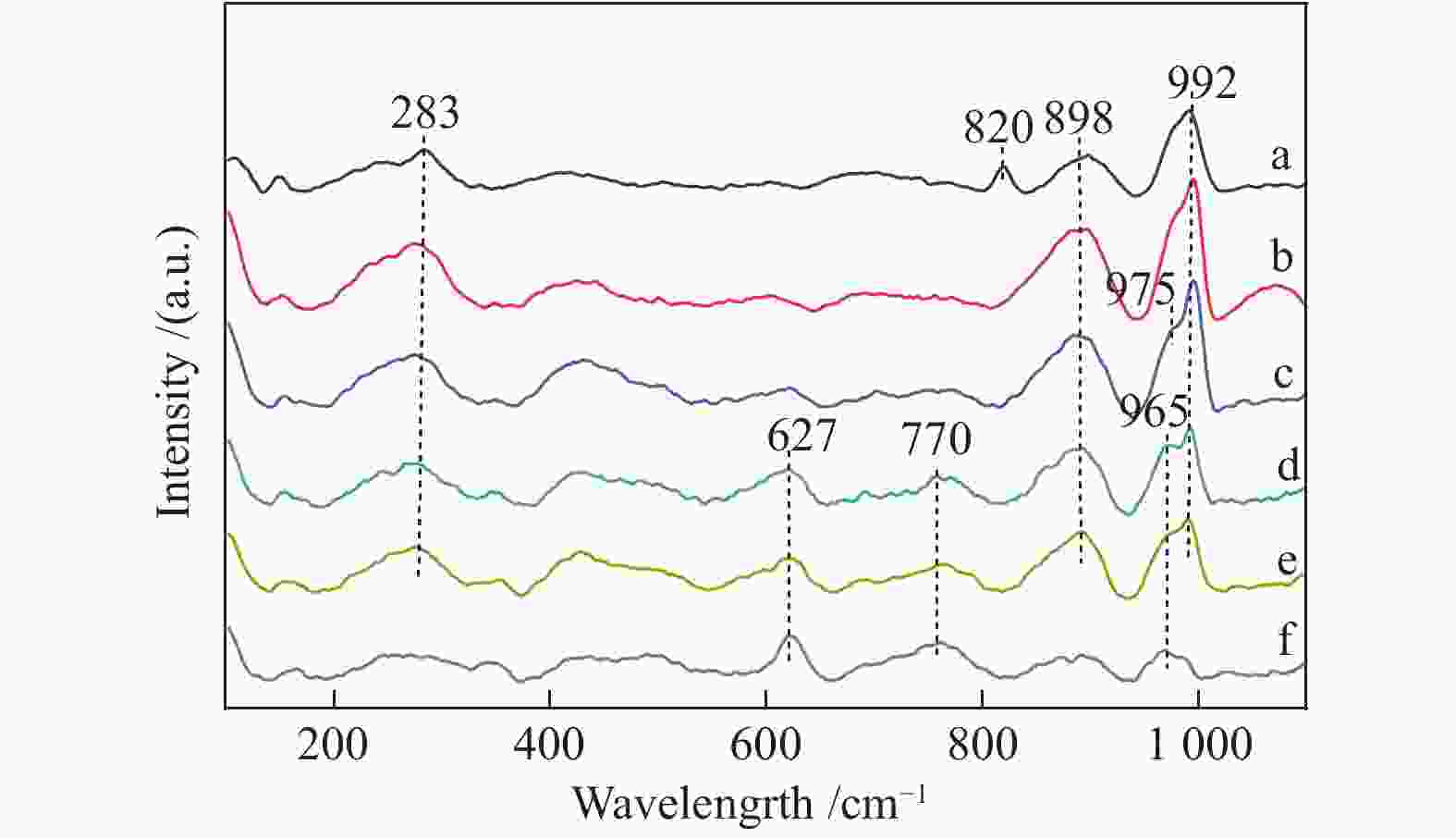
表 1 不同Mo量Mo-Sn催化剂上甲醇转化的反应性能
Table 1 Effect of Mo content on the performance of Mo-Sn catalysts for methanol conversion
Catalyst CH3OH
conversion/%C-mol selectivity/% MF DME FA DMM COx MoO3 0 0 0 0 0 0 Mo2Sn1 17.2 22.4 23.1 0 54.5 0 Mo1Sn1 28.3 45.0 23.5 0 31.5 0 Mo1Sn2 33.9 43.8 17.2 2.9 36.1 0 Mo1Sn3 32.2 42.8 9.2 0 48.0 0 Mo1Sn4 26.5 36.3 5.0 0 58.7 0 Mo1Sn5 21.0 20.9 4.3 2.2 72.6 0 Mo1Sn8 14.5 10.5 1.7 0 87.8 0 Mo1Sn10 14.2 11.1 0 0 88.9 0 Mo1Sn20 10.5 11.8 0 0 88.2 0 SnO2 0 0 0 0 0 0 Reaction conditions: atmospheric pressure, tR = 140 ℃,n(CH3OH)∶ n(O2) = 1∶9.415, CH3OH flow rate = 0.817 mL/h, GHSV = 7200 h−1 表 2 不同Mo含量Mo-Sn催化剂的XPS-Mo 3d谱图分析
Table 2 XPS-Mo 3d analysis of Mo-Sn catalysts with different Mo contents
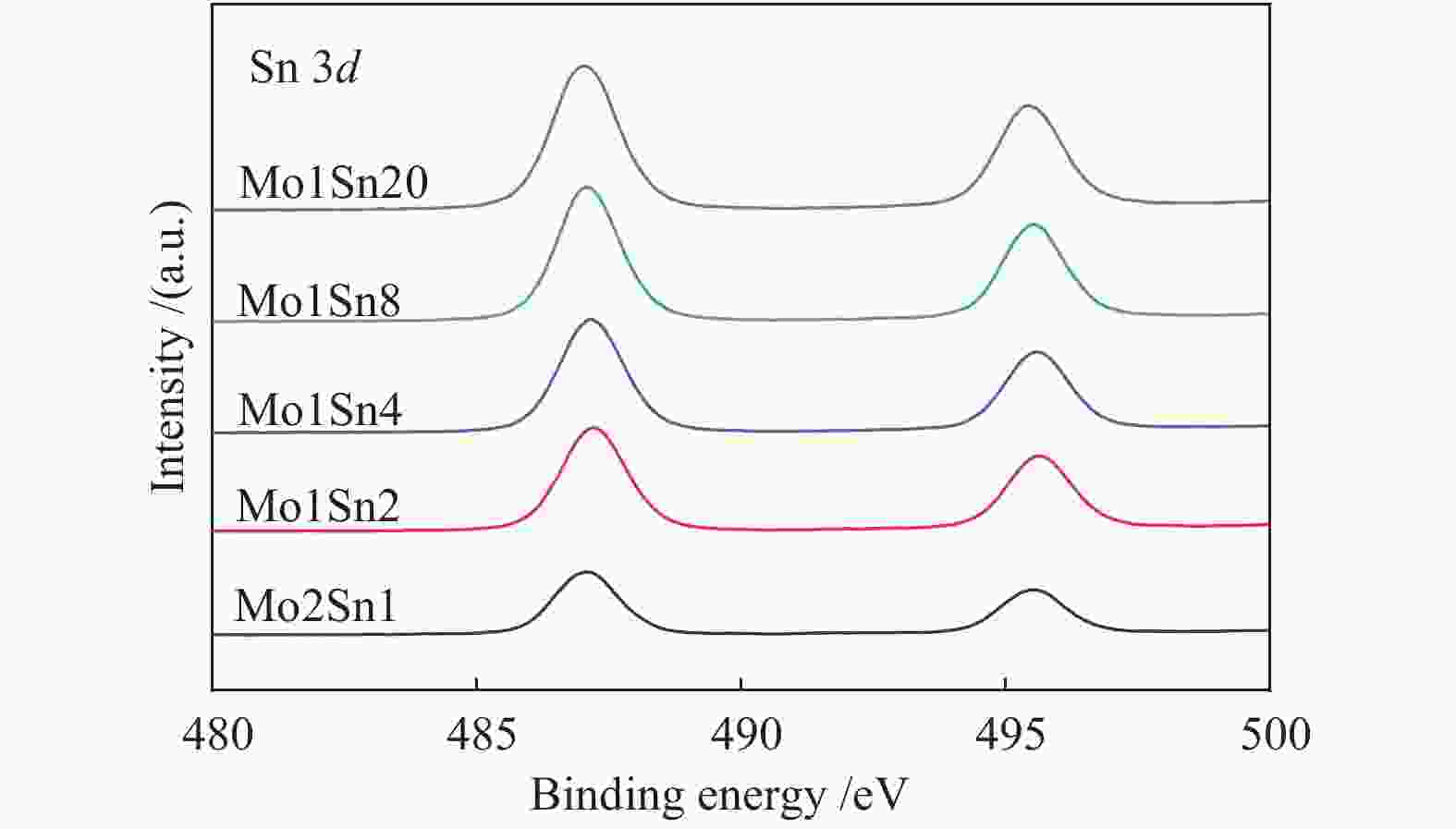
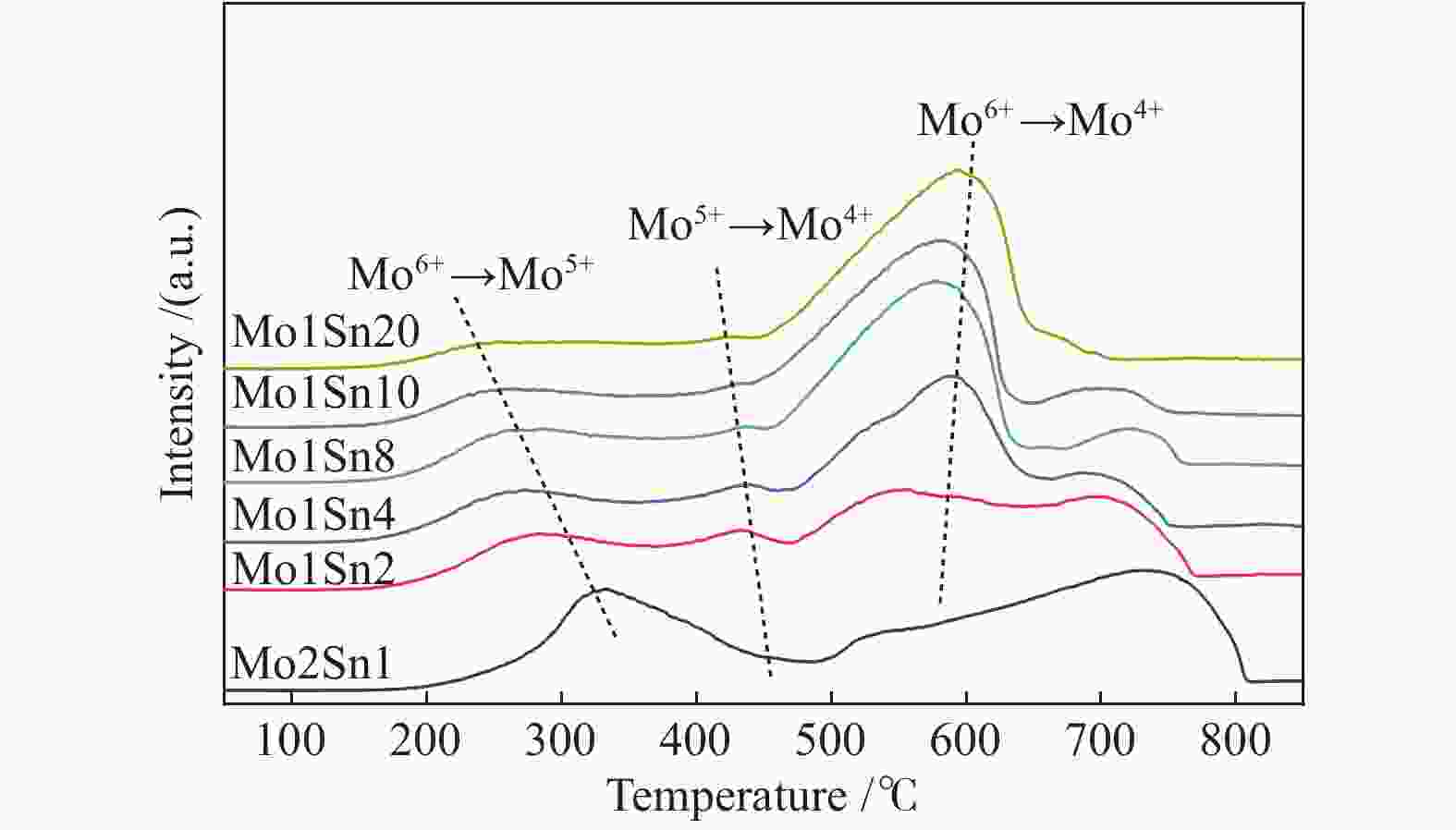
Catalyst Mo6+
3d3/2Mo6+
3d5/2Mo5+
3d3/2Mo5+
3d5/2Mo6+/
%Mo5+/
%Mo2Sn1 236.33 233.07 235.18 231.78 94.5 5.5 Mo1Sn2 236.27 233.10 235.10 231.80 93.1 6.9 Mo1Sn4 236.24 233.10 235.17 231.90 86.3 13.7 Mo1Sn8 236.18 233.02 235.06 231.84 69.6 30.4 Mo1Sn20 236.14 232.94 235.11 231.87 58.9 41.1 表 3 不同Mo含量Mo-Sn催化剂的XPS-O 1s谱图分析
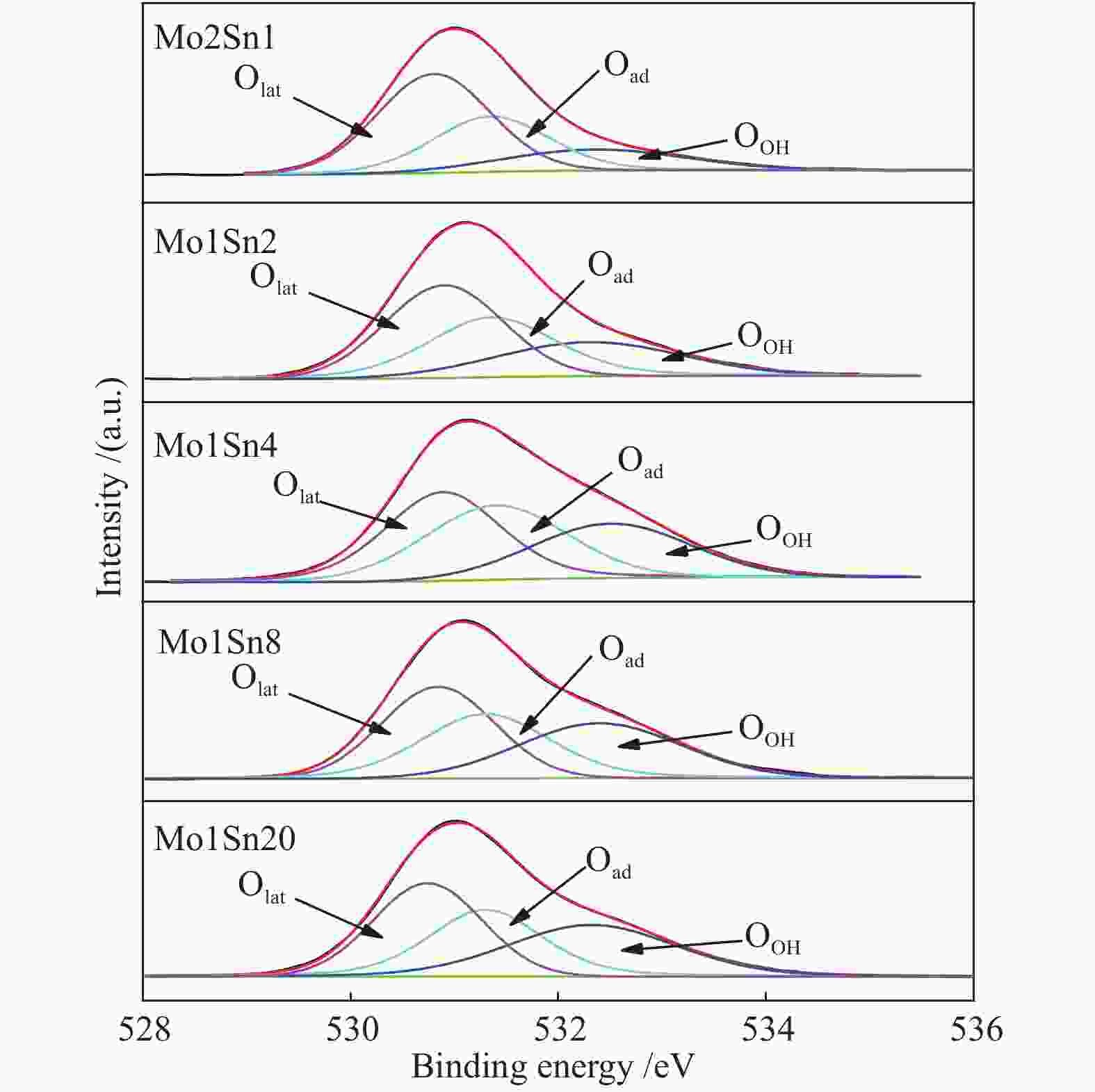
Table 3 XPS-O 1s analysis of Mo-Sn catalysts with different Mo contents
Catalyst OOH Oad Olat OOH/OTotal Oad/OTotal Olat/OTotal Mo2Sn1 532.35 531.35 530.80 0.17 0.31 0.52 Mo1Sn2 532.30 531.36 530.90 0.24 0.34 0.42 Mo1Sn4 532.39 531.39 530.89 0.28 0.38 0.34 Mo1Sn8 523.40 531.30 530.84 0.32 0.32 0.36 Mo1Sn20 532.32 531.30 530.74 0.33 0.31 0.36 -
[1] LIU X M, LU G Q, YAN Z F, YAN Z F, JORGE B. Recent advances in catalysts for methanol synthesis via hydrogenation of CO and CO2[J]. Ind Eng Chem Res,2003,42(25):6518−6530. doi: 10.1021/ie020979s [2] GRABOW L C, MAVRIKAKIS M. Mechanism of methanol synthesis on Cu through CO2 and CO hydrogenation[J]. ACS Catal,2011,1(4):365−384. doi: 10.1021/cs200055d [3] BEHRENS M, STUDT F, KASATKIN I, KUHL S, HAVECKER M, ABILD-PEDERERSEN F, ZANDER S, GIRGSDIES F, KURR P, KNIEP B L, TOVAR M, FISCHER R W, NORSKOV J K, SCHLOGL R. The active site of methanol synthesis over Cu/ZnO/Al2O3 industrial catalysts[J]. Science,2012,336(3):893−897. [4] TIAN P, WEI Y, YE M, LIU Z M. Methanol to olefins (MTO): From fundamentals to commercialization[J]. ACS Catal,2015,5(3):1922−1938. doi: 10.1021/acscatal.5b00007 [5] MARCINIAK A A, ALVES O C, APPEL L G, MOTA J A. Synthesis of dimethyl carbonate from CO2 and methanol over CeO2: Role of copper as dopant and the use of methyl trichloroacetate as dehydrating agent[J]. J Catal,2019,371(1):88−95. [6] YANG Z, FENG J, CHENG H, LIU Y X, JIANG J C. Directional depolymerization of lignin into high added-value chemical with synergistic effect of binary solvents[J]. Bioresour Technol,2021,321(12):124−440. [7] LI S, PENG D, YU J. Morphologically controllable Li plating with stable electrochemistry realized in a newly developed DOL-DMM electrolyte system on Au-modified Cu current collector[J]. Ionics,2020,26(8):3979−3988. doi: 10.1007/s11581-020-03527-3 [8] BADMAEV S D, SMORYGINA A S, PAUKSHTIS E A, BELYAEV V D, SOBYANIN V A, PARMON V N. Gas-phase carbonylation of dimethoxymethane to methyl methoxyacetate on solid acids: The effect of acidity on the catalytic activity[J]. Kinet Catal,2018,59(1):99−103. doi: 10.1134/S0023158418010020 [9] CHONG J M, SHEN L X. Preparation of chloromethyl methyl ether revisited[J]. Synth Commun,1998,28(15):2801−2806. doi: 10.1080/00397919808004855 [10] NOUGUIER R, MIGNON V, GRAS J. Synthesis of methylene acetals in the D-glucose, D-galactose, D-mannose, and D-fructose series by an improved transacetalation reaction from dimethoxymethane[J]. Carbohydr Res,1995,277(2):339−345. doi: 10.1016/0008-6215(95)00218-I [11] YANG Z, FENG J, CHENG H, LIU Y X, JIAN J C. Directional depolymerization of lignin into high added-value chemical with synergistic effect of binary solvents[J]. Bioresour Technol,2021,321(1):124440. [12] WANG J, LIU J, SONG H, CHEN J. Heteropolyacids as efficient catalysts for the synthesis of precursors to ethylene glycol by the liquid-phase carbonylation of dimethoxymethane[J]. Chem Lett,2015,44(6):806−808. doi: 10.1246/cl.150131 [13] LI M, LONG Y, DENG Z, ZHANG H, YANG X G, WANG G Y. Ruthenium trichloride as a new catalyst for selective production of dimethoxymethane from liquid methanol with molecular oxygen as sole oxidant[J]. Catal Commun,2015,68(4):46−48. [14] LIU H C, IGLESIA E. Selective one-step synthesis of dimethoxymethane via methanol or dimethyl ether oxidation on H3+nVnMo12−nPO40 keggin structures[J]. J Phys Chem B,2003,107(39):10840−10847. doi: 10.1021/jp0301554 [15] FU Y, SHEN J. Selective oxidation of methanol to dimethoxymethane under mild conditions over V2O5/TiO2 with enhanced surface acidity[J]. Chem Commun,2007,21(21):2172−2174. [16] TAO M, WANG H, BIN L, ZHAO J X, CAI Q H. Highly selective oxidation of methanol to dimethoxymethane over $ {\rm{SO}}_4^{2 - } $ -V2O5-ZrO2[J]. New J Chem,2017,41(16):8370−8376. doi: 10.1039/C7NJ01295K[17] CHEN S, MA X. The role of oxygen species in the selective oxidation of methanol to dimethoxymethane over VOx/TS-1 catalyst[J]. J Ind Eng Chem,2017,45(9):296−300. [18] AI M. The production of methyl formate by the vapor-phase oxidation of methanol[J]. J Catal,1982,21(77):279−288. [19] LIU G B, ZHANG Q D, HAN Y Z, TSUBAKI N, Tan Y S. Effects of the MoO3 structure of Mo-Sn catalysts on dimethyl ether oxidation to methyl formate under mild conditions[J]. Green Chem,2015,17(2):1057−1064. doi: 10.1039/C4GC01591F [20] ZHANG Z Z, ZHANG Q D, JIA L, WANG W F, SHAO P T, WANG P, HE X, HAN Y Z, TSUBAKI N, TAN Y S. The effects of the Mo-Sn contact interface on the oxidation reaction of dimethyl ether to methyl formate at a low reaction temperature[J]. Catal Sci Technol,2016,15(6):6109−6117. [21] 杨奇, 高秀娟, 冯茹, 李明杰, 张俊峰, 张清德, 韩怡卓, 谭猗生. 水热合成的MoO3-SnO2催化剂催化氧化二甲醚的性能研究[J]. 燃料化学学报,2019,47(8):934−941. doi: 10.3969/j.issn.0253-2409.2019.08.005YANG Qi, GAO Xiu-juan, FENG Ru, LI Ming-jie, ZHANG Jun-feng, ZHANG Qing-de, HAN Yi-zhuo, TAN Yi-sheng. MoO3-SnO2 catalyst prepared by hydrothermal synthesis method for dimethyl ether catalytic oxidation[J]. J Fuel Chem Technol,2019,47(8):934−941. doi: 10.3969/j.issn.0253-2409.2019.08.005 [22] 陈文龙, 刘海超. 甲醇选择氧化金属氧化物催化剂的结构与其催化性能的关系[J]. 物理化学学报,2012,28(10):2315−2356. doi: 10.3866/PKU.WHXB201209146CHEN Wen-long, LIU Hai-chao. Relationship between the structures of metal oxide catalysts and their properties in selective oxidation of methanol[J]. Acta Phys-Chim Sin,2012,28(10):2315−2356. doi: 10.3866/PKU.WHXB201209146 [23] LIU H C, CHEUNG P, IGLESIA E. Structure and support effects on the selective oxidation of dimethyl ether to formaldehyde catalyzed by MoOx domains[J]. J Catal,2003,217(1):222−232. [24] GONCALVES F, MEDEIROS P R S, EON J G, APPEL L G. Active sites for ethanol oxidation over SnO2-supported molybdenum oxides[J]. Appl Catal A: Gen,2000,193(1/2):195−202. [25] 杨奇. 钼锡催化剂上二甲醚低温氧化机理研究[D]. 北京: 中国科学院大学, 2019.YANG Qi. Study on the mechanism of the low-temperature oxidation of dimethyl ether over MoO3-SnO2 catalyst[D]. Beijing: University of Chinese Academy of Sciences, 2019. [26] HANG Z Z, ZHANG Q D, JIA L Y, WANG W F, ZHANG T, HAN Y Z, TSUBAKI N, TAN Y S. Effects f tetrahedral molybdenum oxide species and MoOx domains on the selective oxidation of dimethyl ther under mild conditions[J]. Catal Sci Technol,2016,6(9):2975−2983. doi: 10.1039/C5CY01569C [27] YANG J, XIAO X, CHEN P, ZHU K, CHENG K, YE K, WANG G L, CAO D X, YAN J. Creating oxygen-vacancies in MoO3−x nanobelts toward high volumetric energy-density asymmetric superercapacitors with long lifespan[J]. Nano Energy,2019,58(1):455−465. -





 下载:
下载: