Effects of ball milling medium on Cu-Al spinel sustained release catalyst for H2 generation from methanol steam reforming
-
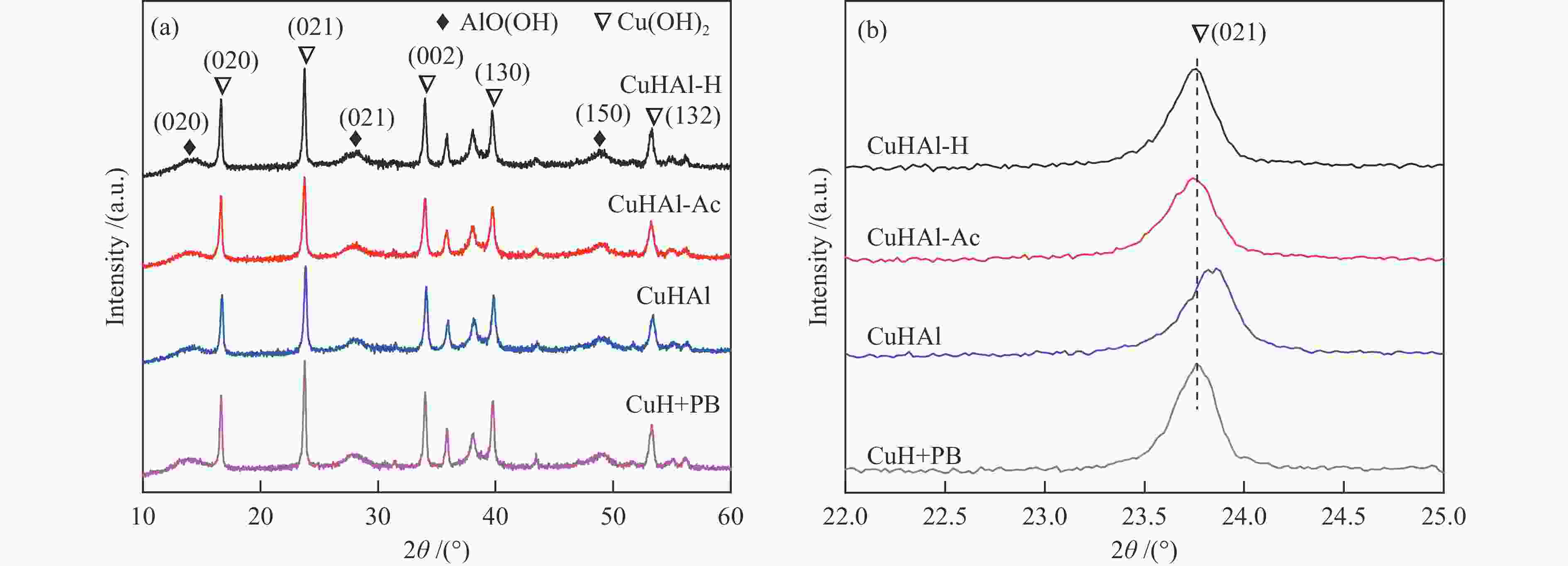
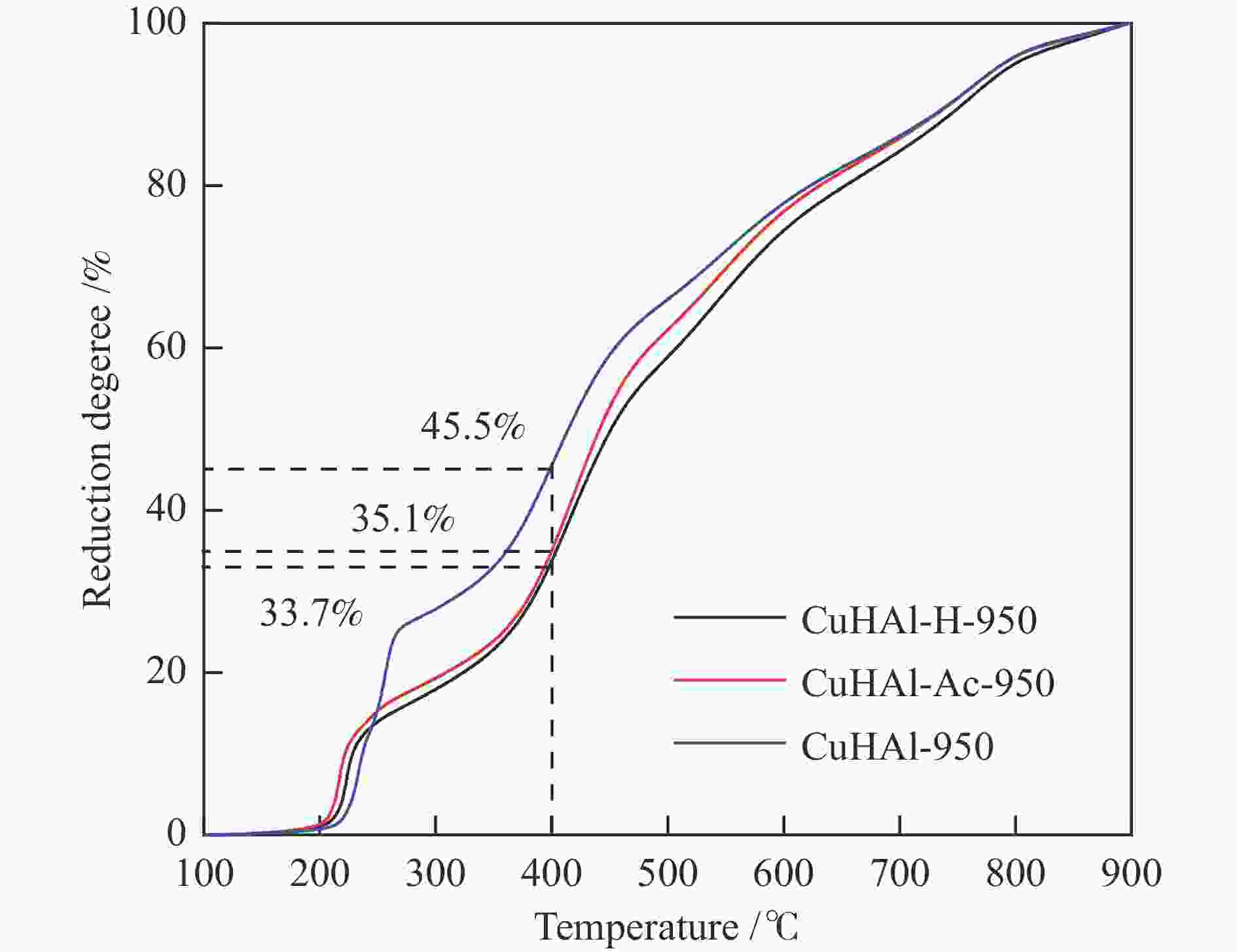
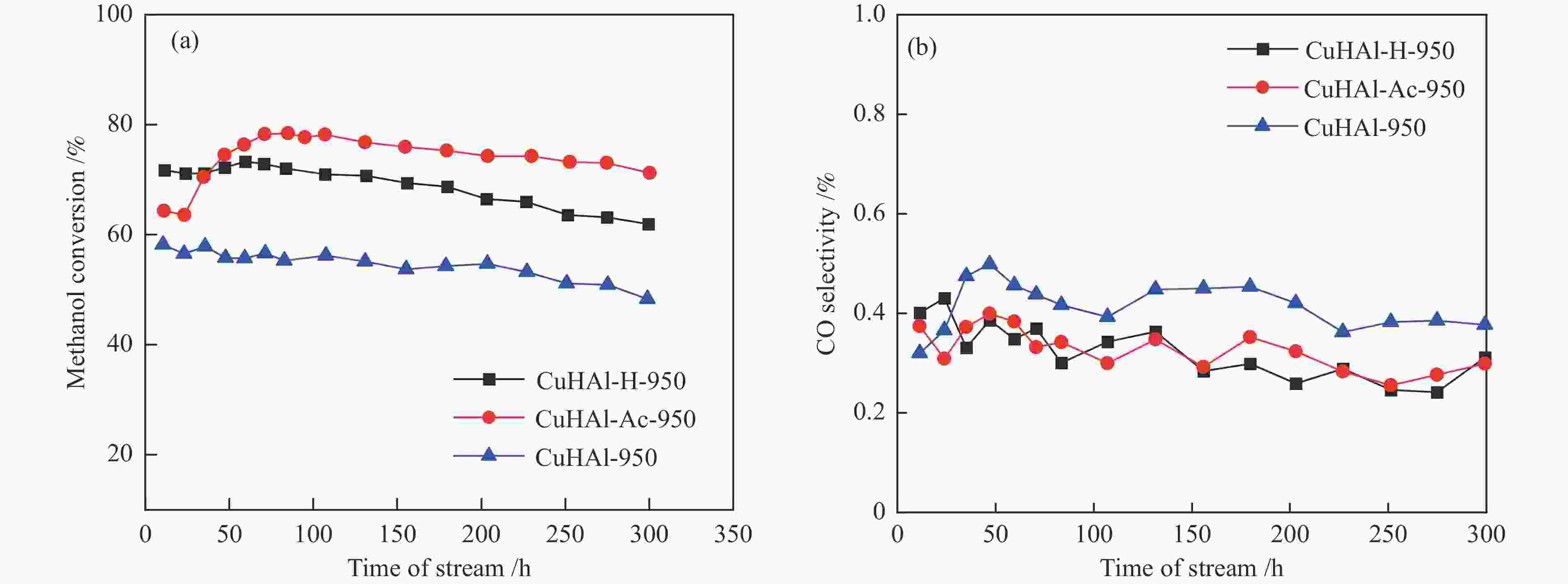
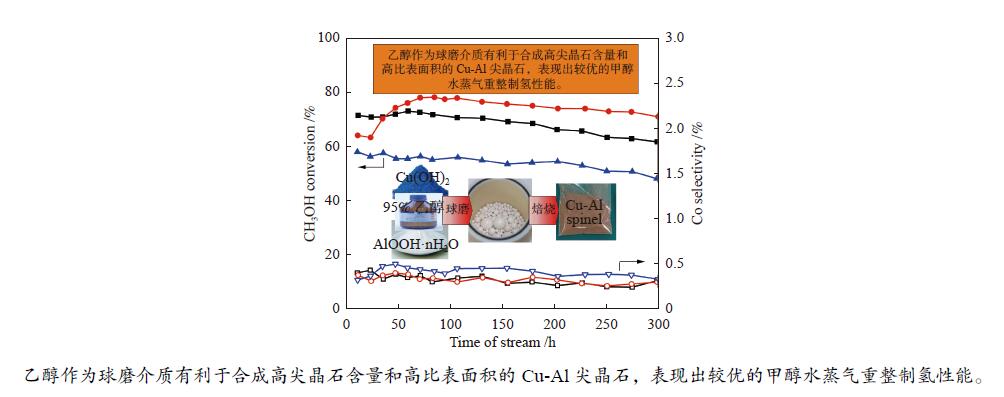
摘要: 以拟薄水铝石和超细氢氧化铜为原料,选择Cu/Al物质的量比为1∶3,考察了不同机械球磨介质对固相法制备Cu-Al尖晶石缓释催化剂的影响。通过XRD、BET和H2-TPR对合成的催化剂进行了表征,并考察了其对甲醇水蒸气重整制氢的催化性能。结果表明,Cu-Al尖晶石固溶体可通过干式或湿式机械球磨法合成,然而,通过湿式球磨法能使较多Cu2 + 进入尖晶石晶体结构。合成的富含Al的尖晶石固溶体在晶粒尺寸上差异不大,但它们的比表面积、孔隙体积和还原性能却明显不同。与干磨法相比,湿磨法有利于促进固相反应,通过湿式球磨法合成的催化剂只有尖晶石晶相,且具有较高的比表面积和较大的孔隙体积。通过湿法研磨得到的Cu-Al尖晶石催化剂样品在甲醇水蒸气重整制氢反应中表现出更好的催化性能,用乙醇(95%)作为球磨介质制备的CuHAl-Ac-950表现出最高的催化活性。Abstract: Using pseudo-boehmite and ultrafine copper hydroxide as the raw materials with n(Cu/Al) = 1∶3, the effects of ball milling medium on the Cu-Al spinel sustained release catalysts prepared via the solid-state reaction method are explored. The obtained catalysts are characterized by XRD, BET, and H2-TPR techniques, and their catalytic properties in methanol steam reforming (MSR) are evaluated. The results demonstrate that Cu-Al spinel solid solution can be synthesized by both dry and wet mechanical ball milling methods, and more Cu2 + ions are found to be incorporated into the spinel lattice through the latter method. The crystalline sizes of as-synthesized spinels are similar; however, the specific surface areas and pore volumes are different as well as their reduction properties. Compared with the dry milling method, the wet ball milling method can facilitate the solid phase reaction, generating catalysts with solely spinel crystalline phase, higher specific surface area, and larger pore volume. Furthermore, catalysts derived from the wet milling method demonstrate improved catalytic activity and stability, and lower CO selectivity in MSR. The highest activity is obtained over CuHAl-Ac-950 prepared using ethanol (95%) as the ball milling medium.
-
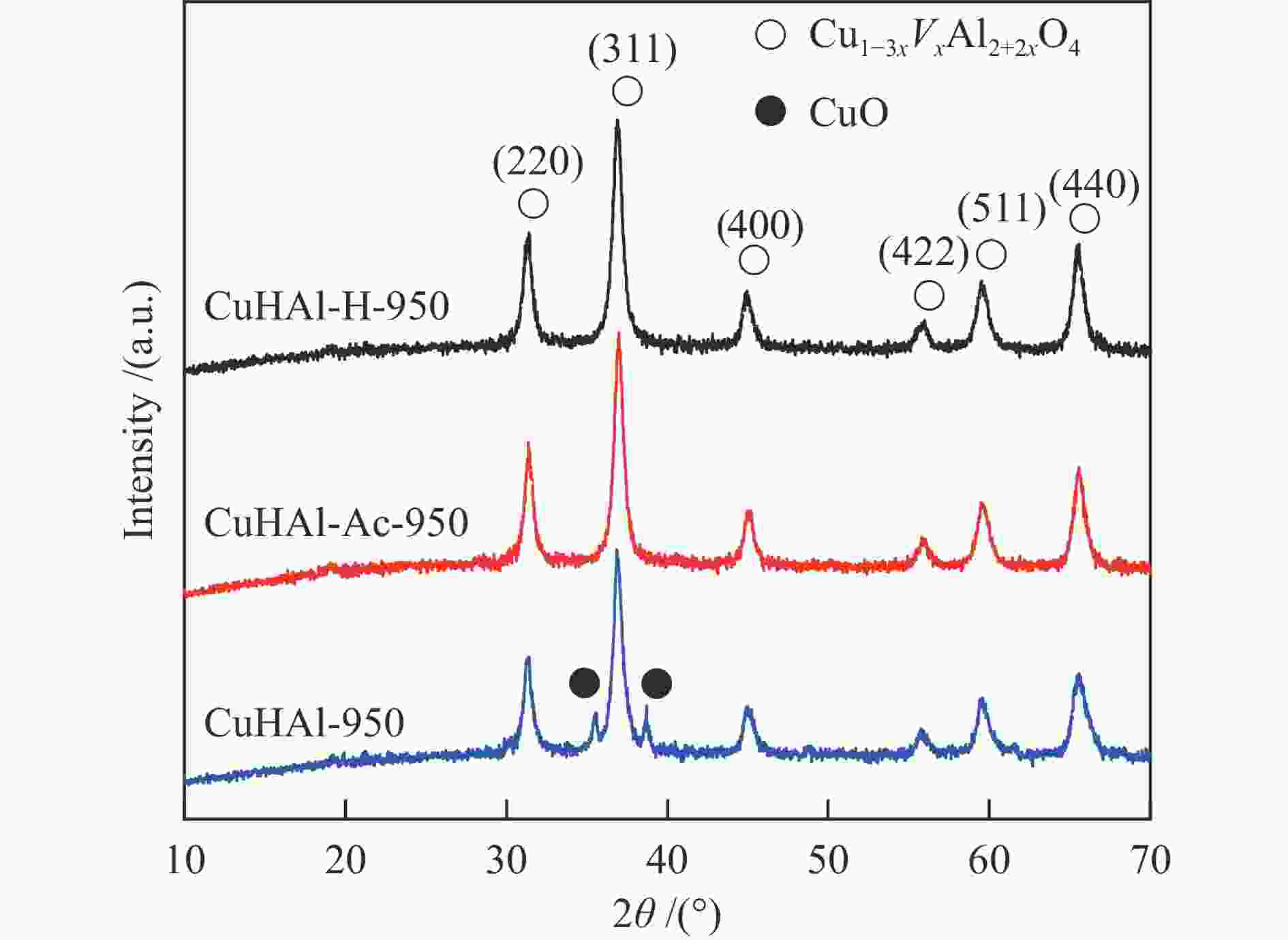
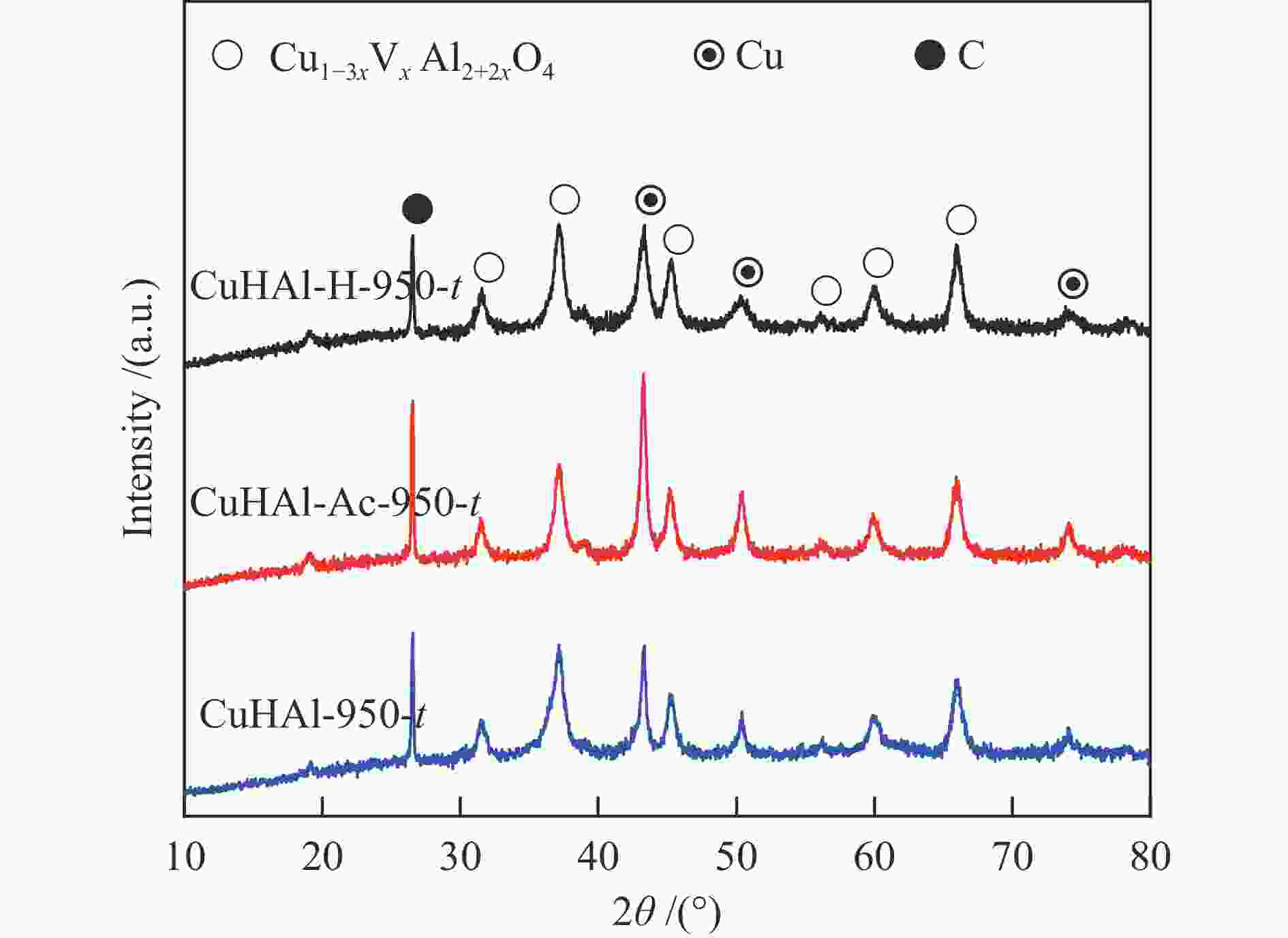
Table 1 Relative intensity of six main diffraction peaks of the spinel phase
Sample (220) (311) (400) (422) (511) (440) PDF#78-1605 50.7 100 16.8 12.3 31.1 38.2 CuHAl-950 48.3 100 25.1 15.3 28.5 42.3 CuHAl-Ac-950 53.4 100 24.3 13.5 26.6 43.8 CuHAl-H-950 50.1 100 25.4 13.2 29.9 46.8 Table 2 Characteristic parameters of the fresh and tested catalysts
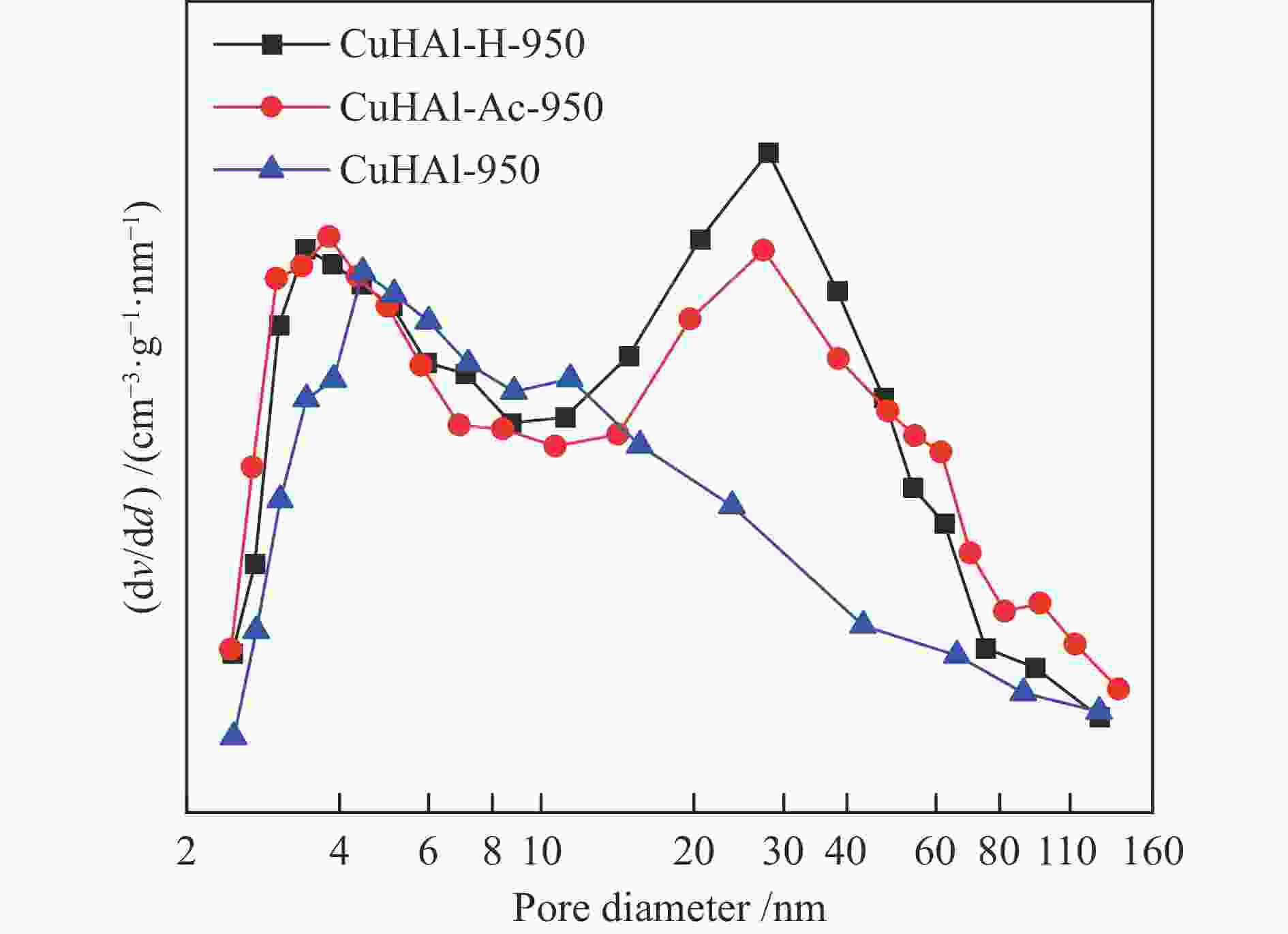
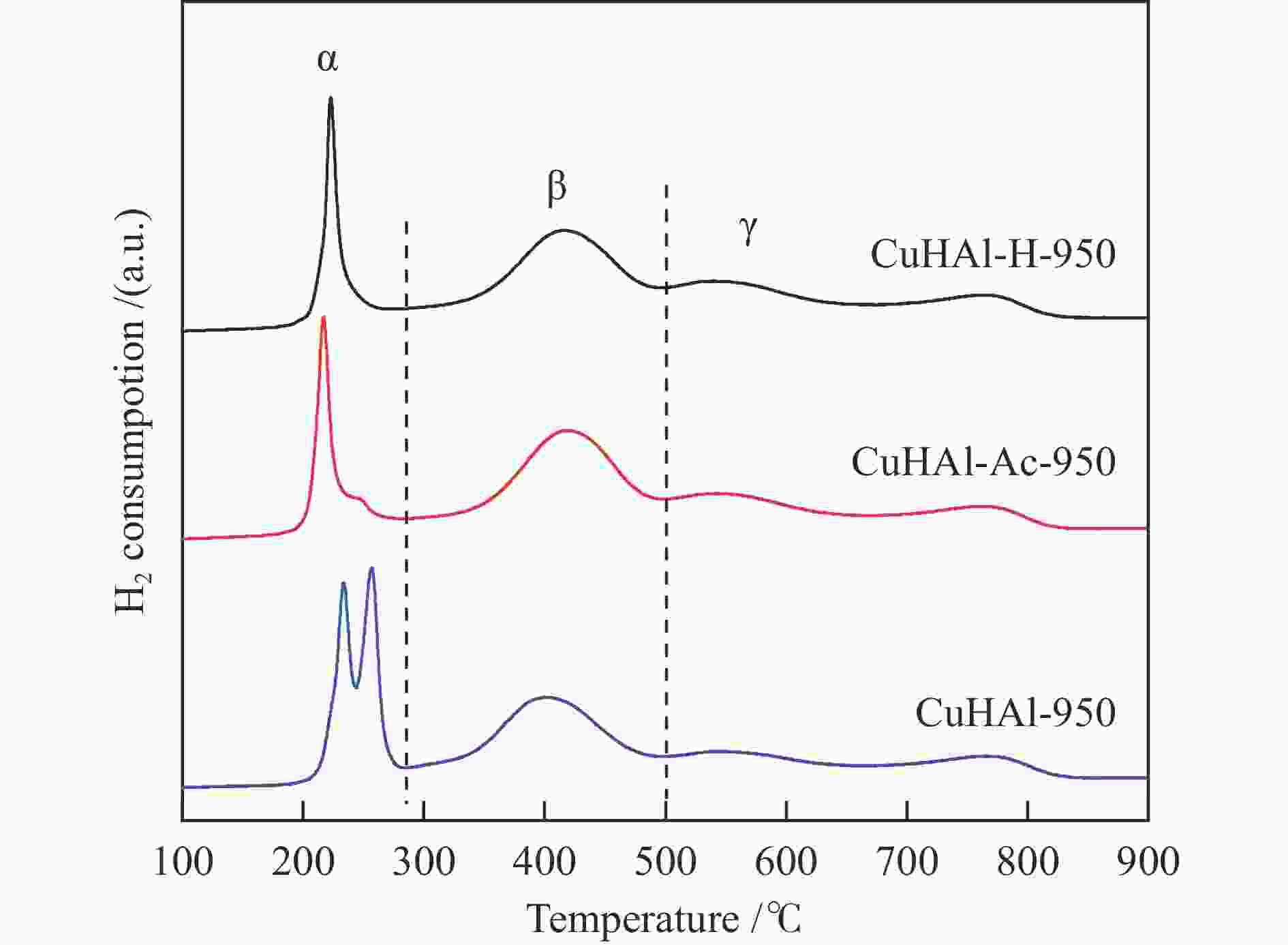
Fresh catalyst CuHAl-950 CuHAl-Ac-950 CuHAl-H-950 d a spinel /nm 12.1 11.9 12.3 SBET /(m2·g−1) 56.1 86.4 89.5 Pore volume /(cm3·g−1) 0.380 0.727 0.663 Y b spinel /% 69.5 79.8 80.7 x in Cu1−3xVxAl2 + 2xO4 0.155 0.133 0.131 Tested catalysts CuHAl-950-t CuHAl-Ac-950-t CuHAl-H-950-t d c Cu /nm 15.7 16.0 11.3 a: Cu-Al spinel crystal size; b: Spinel Cu2 + content; c: Cu crystal size -
[1] MINDRU I, GINGASU D, PATRON L, MARINESCU G, CALDERON-MORENO J M, PREDA S, OPREA O, NITA S. Copper aluminate spinel by soft chemical routes[J]. Ceram Int,2016,42:154−164. doi: 10.1016/j.ceramint.2015.08.058 [2] KWAK B K, PARK D S, YUN Y S, YI J. Preparation and characterization of nanocrystalline CuAl2O4 spinel catalysts by sol-gel method for the hydrogenolysis of glycerol[J]. Catal Commun,2012,24:90−95. doi: 10.1016/j.catcom.2012.03.029 [3] PATEL S, PANT K K. Activity and stability enhancement of copper-alumina catalysts using cerium and zinc promoters for the selective production of hydrogen via steam reforming of methanol[J]. J Power Sources,2006,159:139−143. doi: 10.1016/j.jpowsour.2006.04.008 [4] HUANG Y H, WANG S F, TSAI A P, KAMEOKA S. Reduction behaviors and catalytic properties for methanol steam reforming of Cu-based spinel compounds CuX2O4 (X=Fe, Mn, Al, La)[J]. Ceram Int,2014,40:4541−4551. doi: 10.1016/j.ceramint.2013.08.130 [5] XI H J, LI G J, QING S J, HOU X N, ZHAO J Z, LIU Y J, GAO Z X. Cu-Al spinel catalyst prepared by solid phase method for methanol steam reforming[J]. J Fuel Chem Technol,2013,41:998−1002. [6] QIN F J, LIU Y J, QING S J, HOU X N, GAO Z X. Cu-Al spinel as a sustained release catalyst for H2 production from methanol steam reforming: Effects of different copper sources[J]. J Fuel Chem Technol,2017,45:1481−1488. doi: 10.1016/S1872-5813(17)30065-8 [7] XI H J, HOU X N, LIU Y J, QING S J, GAO Z X. Cu-Al spinel oxide as an efficient catalyst for methanol steam reforming[J]. Angew Chem Int Ed,2014,53:11886−11889. doi: 10.1002/anie.201405213 [8] LIU Y J, QING S J, HOU X N, QIN F J, WANG X, GAO Z X, XIANG H W. Temperature dependence of Cu-Al spinel formation and its catalytic performance in methanol steam reforming[J]. Catal Sci Technol,2017,7:5069−5078. doi: 10.1039/C7CY01236E [9] FAN Y, LU X B, NI Y W, ZHANG H J, ZHU M W, LI Y, CHEN J P. Catalytic destruction of chlorinated aromatic pollutants over mesoporous CuxMg1–xAl2O4 spinel oxides[J]. Appl Catal B: Environ,2011,101:606−612. doi: 10.1016/j.apcatb.2010.11.001 [10] BAYAL N, JEEVANANDAM P. Synthesis of metal aluminate nanoparticles by sol-gel method and studies on their reactivity[J]. J Alloys Compd,2012,516:27−32. doi: 10.1016/j.jallcom.2011.11.080 [11] LV W Z, LUO Z L, YANG H, LIU B, WENG W J, LIU J H. Effect of processing conditions on sonochemical synthesis of nanosized copper aluminate powders[J]. Ultrason Sonochem,2010,17:344−351. doi: 10.1016/j.ultsonch.2009.06.006 [12] RIVAS M E. Ball milling towards green synthesis: applications, projects, challenges[J]. Johnson Matthey Tech Rev,2016,60:148−150. doi: 10.1595/205651316X691375 [13] QIN F J. Synthesis and characterization of Cu-Al spinel sustained release catalysts[D]. Beijing: University of Chinese Academy of Sciences, 2018. [14] BAKKER H, ZHOU G F, YANG H. Mechanically driven disorder and phase transformations in alloys[J]. Prog Mater Sci,1995,39:159−241. doi: 10.1016/0079-6425(95)00001-1 [15] LEE W H, LEE J, BAE J D, BYUN C S, KIM D K. Syntheses of Ni2Si, Ni5Si2, and NiSi by mechanical alloying[J]. Scripta Mater,2001,44:97−103. doi: 10.1016/S1359-6462(00)00547-9 [16] KRYSTNA W C, GAMRAT K, FELA K. Chemical reactions during high-energy ball milling of the Cu2(OH)2CO3–Al0 system[J]. Solid State Ionics,2003,164:193−198. doi: 10.1016/S0167-2738(03)00320-5 [17] AKGUL F A, AKGUL G. Microstructural properties and local atomic structures of cobalt oxide nanoparticles synthesized by mechanical ball-milling process[J]. Kurban M Philos Mag,2016,96:3211−3226. doi: 10.1080/14786435.2016.1232493 [18] MCCORMICK P G, TSUZUKI T, ROBINSON J S, DING J. Nanopowders synthesized by mechanochemical processing[J]. Adv Mater,2001,13:1008−1010. doi: 10.1002/1521-4095(200107)13:12/13<1008::AID-ADMA1008>3.0.CO;2-Q [19] GOYA G F, RECHENBERG H R. Magnetic properties of ZnFe2O4 synthesized by ball milling[J]. J Magn Magn Mater,1999,203:141−142. doi: 10.1016/S0304-8853(99)00250-4 [20] JANOT R, GUÉRARD D. One-step synthesis of maghemite nanometric powders by ball-milling[J]. J Alloys Compd,2002,333:302−307. doi: 10.1016/S0925-8388(01)01737-6 [21] CROCKER M, HEROLD R H M, EMEIS C A, KRIJGER M. Preparation of acidic forms of montmorillonite clay via solid-state ion-exchange reactions[J]. Catal Lett,1992,1:339−345. [22] KECSENOVITY E, FEJES D, RETI B, HERNADI K. Growth and characterization of bamboo-like carbon nanotubes synthesized on Fe-Co-Cu catalysts prepared by high-energy ball milling[J]. Phys Status Solidi B,2013,250:2544−2548. doi: 10.1002/pssb.201300075 [23] HUANG L, KRAMER G J, WIELDRAAIJER W, BRANDS D S, POELS E K, CASTRICUM H L, BAKKER H. Methanol synthesis over Cu/ZnO catalysts prepared by ball milling[J]. Catal Lett,1997,48:55−59. doi: 10.1023/A:1019014701674 [24] GRANDJEAN D, CASTRICUM H L, VAN J C, WECKHUYSEN B M. Highly mixed phases in ball-milled Cu/ZnO catalysts: an EXAFS and XANES study[J]. J Phys Chem B,2006,110:16892−16901. doi: 10.1021/jp055820i [25] MAHMOUD M H, HASSAN A M, SAID E A A, HAMDEH H H. Structural; magnetic and catalytic properties of nanocrystalline Cu0.5Zn0.5Fe2O4 synthesized by microwave combustion and ball milling methods[J]. J Mol Struct,2016,1114:1−6. doi: 10.1016/j.molstruc.2016.02.051 [26] RALPHS K, HARDACRE C, JAMES S L. Application of heterogeneous catalysts prepared by mechanochemical synthesis[J]. Chem Soc Rev,2013,42:7701−7718. doi: 10.1039/c3cs60066a [27] XU J L, GUO Q, GAO W, KANG Z, XI G Q, ZHANG L. Effect of milling mediums on nano-Sb powders prepared by ball milling[J]. J Aeronaut Mater,2013,33:50−55. [28] LIU Y J, QING S J, HOU X N, ZHANG L, GAO Z X, XIANG H W. Synthesis of Cu-Al spinels and its non-isothermal formation kinetics analysis[J]. J Fuel Chem Technol,2020,48:338−348. -




 下载:
下载: